No products in the cart.
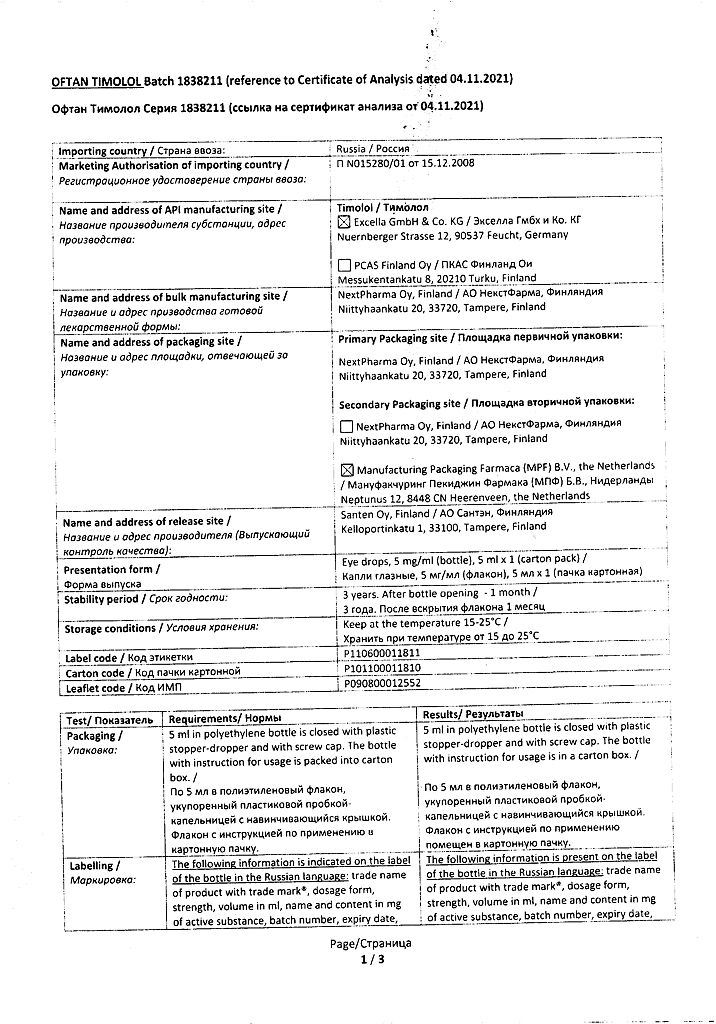
Oftan Timolol, eye drops 5 mg/ml 5 ml
€2.04 €1.00
Out of stock
(E-mail when Stock is available)
Description
The main active ingredient is timolol, a substance from the group of non-selective beta-adrenoblockers. Timolol blocks the beta-adrenoceptors without a parallel pronounced sympathomimetic effect. The mechanism of hypotensive action of Timolol eye drops is associated with inhibition of intraocular fluid (aqueous humor) production in the anterior chamber of the eye, as well as facilitation of outflow of aqueous humor from the eye chambers. The drug has no effect on the size of the pupil and accommodation capacity of the eye, therefore it does not affect visual acuity and does not worsen the quality of twilight vision.
The drug reduces both elevated and normal intraocular pressure regardless of whether the patient has glaucoma or not.
A pronounced hypotensive effect develops on the average 30 minutes after a single use of the eye drops and its maximum effect is 1-2 hours after instillation of the solution into the conjunctival sac; the pressure remains reduced for 24 hours after a single use of the medicine.
Timolol is well tolerated and causes a small number of side effects and when used as directed has almost no effect on heart rate.
Indications
Indications
Elevated intraocular pressure, Glaucoma
For reduction of intraocular pressure and prevention of complications in patients with elevated ophthalmotonus and primary open angle glaucoma as well as secondary glaucoma (including posttraumatic, uveal and aphakic).
The drug may be administered in the complex therapy of closed glaucoma (when prescribed with mitotics), as an adjunctive drug in congenital glaucoma or in cases of acute increase in intraocular pressure.
Active ingredient
Active ingredient
Timolol
How to take, the dosage
How to take, the dosage
At the beginning of treatment, 1-2 drops in the affected eye 2 times a day.
If intraocular pressure normalizes with regular use, the dose should be reduced to 1 drop once daily in the morning.
Treatment with Oftan Timolol is usually prolonged. Interruption of treatment or changes in the dosage of the drug are made only on the prescription of the attending physician.
Interaction
Interaction
The concomitant use of Oftan Timolol with eye drops containing adrenaline may cause dilation of the pupil.
The simultaneous use of eye drops containing epinephrine and pilocarpine may increase the decrease in intraocular pressure.
Arterial hypotension and bradycardia may worsen when concomitant use of Oftan Timolol with calcium antagonists, reserpine and systemic beta-adrenoblockers.
CYP2D6 isoenzyme inhibitors, such as quinidine and cimetidine, may increase the plasma concentration of Timolol.
Simultaneous use with insulin or oral antidiabetic agents may lead to hypoglycemia.
Timolol increases the effects of muscle relaxants, therefore it is necessary to cancel the drug 48 hours prior to the planned surgery under general anesthesia. This information may also apply to medications used shortly before.
.
Special Instructions
Special Instructions
Visual disturbances, dizziness and fatigue may occasionally be observed with Oftan Timolol Eye Drops.
The efficacy of the drug is recommended to be monitored approximately 3-4 weeks after the start of therapy (not earlier than 1-2 weeks). Long-term use of timolol may result in a weakening of the effect.
When co-administered with beta-blockers, calcium channel blockers, excessive pressure decrease may occur. When using the drug Oftan Timolol at least once every 6 months the lacrimal function, corneal integrity and visual field should be monitored.
Oftan Timolol contains benzalkonium chloride which may irritate eyes, be absorbed by soft contact lenses causing discoloration and have adverse effect on ocular tissue. Contact lenses should be removed before application and inserted again not earlier than 15 min after instillation, if necessary.
The change in refraction caused by previously used miotics may need to be corrected when patients are switched to timolol treatment.
Oftan Timolol is used with caution in patients with impaired liver function, renal function and diabetes mellitus.
In case of upcoming surgery under general anesthesia the drug should be withdrawn 48 hours prior to surgery as Timolol increases the effect of muscle relaxants and general anesthetics.
Two different beta-adrenoblockers should not be injected into the same eye.
Impact on driving and operating machinery
At the time of treatment, caution should be exercised while driving motor transport and operating complex equipment requiring high concentration, rapid psychomotor reaction and good vision (within 30 min after eye injection) as the drug may decrease BP and cause fatigue and dizziness.
Contraindications
Contraindications
Timolol should not be administered for the treatment of patients under 18 years of age, with bronchial asthma, chronic obstructive pulmonary disease, with sinus bradycardia, atrioventricular block of II – III degree, chronic heart failure in decompensation stage, cardiogenic shock, corneal dystrophic changes. Do not use if hypersensitivity to any of the ingredients of the drug has occurred.
Particular caution is required when using Timolol in patients with arterial hypotension, sinoatrial blockade of II-III degree, atrophic rhinitis, pulmonary insufficiency, severe disturbances of cerebral circulation, diabetes, myasthenia, thyrotoxic goiter, pheochromocytoma, Raynaud’s syndrome, and during pregnancy or breast-feeding.
The concomitant administration of other beta-adrenoblockers and psychoactive drugs that increase epinephrine release.
Side effects
Side effects
Visually: blurred vision, conjunctival irritation and hyperemia, burning and itching eyes, lacrimation, corneal epithelial edema, pitting superficial keratopathy, corneal hypersthesia, dry eye syndrome, blepharitis, conjunctivitis and keratitis. Prolonged use may cause ptosis and rarely diplopia. During fistulizing (penetrating) antiglaucoma operations vascular detachment may develop in the postoperative period.
Cardiovascular system disorders: bradycardia, bradyarrhythmia, BP decrease, collapse, heart block, transient cerebral circulatory disorders, exacerbation of chronic heart failure, chest pain.
Digestive system disorders: nausea, diarrhea.
Respiratory system: nasal congestion, shortness of breath, bronchospasm, pulmonary failure.
CNS and peripheral nervous system disorders: headache, dizziness, weakness, confusion, hallucinations, insomnia, onyrodynia, anxiety, mood changes, paresthesias.
Skin disorders: alopecia, psoriasis-like rashes and exacerbation of psoriasis.
Urogenital system disorders: Peyronie’s disease, decreased potency.
Allergic reactions: generalized or localized rash, itching.
Others: myasthenia gravis, tinnitus.
.
Overdose
Overdose
Symptoms: possible development of systemic effects typical for beta-adrenoblockers (dizziness, headache, arrhythmia, bradycardia, bronchospasm, nausea and vomiting).
Treatment: immediately flush eyes with water or saline solution, symptomatic therapy.
.
Pregnancy use
Pregnancy use
There is no sufficient experience in the use of the drug during pregnancy and lactation, but it has been found that timolol penetrates the placental barrier and is excreted with breast milk. On prescription of a physician Oftan® Timolol can be used during pregnancy and lactation in cases when the expected medical effect for a mother justifies the potential risk for a fetus and a child.
If the drug was used immediately before delivery or during breastfeeding, neonates should be closely monitored for several days after birth and during the entire period of treatment of nursing mothers with Oftan® Timolol.
Periatric use
In the absence of data on efficacy and safety, the drug is contraindicated in children and adolescents under 18 years of age.
Similarities
Similarities
Arutimol, Okupres-E, Okumed, Timolol-Akos, Timolol Renewal, Timolol-SOLofarm
Additional information
| Weight | 0.017 kg |
|---|---|
| Shelf life | 3 years |
| Conditions of storage | At 15-25 °C |
| Manufacturer | Santen AO, Finland |
| Medication form | eye drops |
| Brand | Santen AO |
Related products
Buy Oftan Timolol, eye drops 5 mg/ml 5 ml with delivery to USA, UK, Europe and over 120 other countries.