No products in the cart.
Novigan, 20 pcs.
€5.99 €5.24
Description
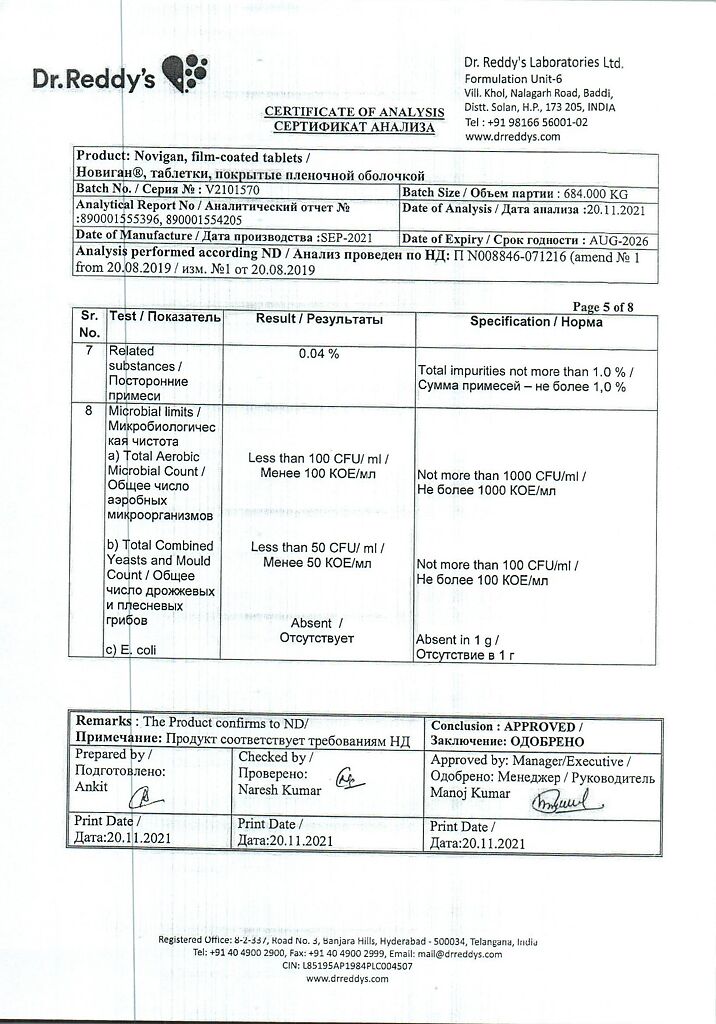
Neuralgia, Arthritis, Bursitis, Colic, Biliary dyskinesia, Flu, Toothache, Inflamed tendons, Neuralgia (nerve pain), Gallic Colic, Migraine, Myalgia (muscle pain), Colitis, Headache, Arthritis
Mild to moderate pain syndrome with spasm of smooth muscles of internal organs: Renal and biliary colic, biliary dyskinesia, intestinal colic.
Active ingredient
Active ingredient
Ibuprofen, Pitofenone, Phenpiveryinia bromide
How to take, the dosage
How to take, the dosage
Orally, 1 hour before or 3 hours after the meal. To avoid irritant effect on the stomach, the drug may be taken immediately after a meal or with milk.
In the absence of special doctor’s prescriptions it is recommended to take Novigan® in spastic pain 1 tablet up to 3 times a day. The maximum daily dose is 3 tablets. Do not exceed the stated dose!
The course of treatment with Novigan® without physician’s consultation should not exceed 5 days. Longer use is possible under the supervision of a physician with control of peripheral blood parameters and functional status of the liver.
Interaction
Interaction
In therapeutic doses Novigan® has no significant interactions with commonly used drugs.
The inducers of microsomal oxidation enzymes in the liver (phenytoin, ethanol, barbiturates, flumecinol, rifampicin, phenylbutazone, tricyclic antidepressants) increase the production of hydroxylated active metabolites, increasing the risk of severe intoxication.
Microsomal oxidation inhibitors reduce the risk of hepatotoxic effects.
Decreases the hypotensive activity of vasodilators and the natriuretic effect of furosemide and hydrochlorothiazide.
Decreases the effectiveness of uricosuric drugs.
Accelerates the effect of indirect anticoagulants, antiaggregants, fibrinolytics (which increases the risk of bleeding).
Accelerates the side effects of mineralocorticosteroids, GCS (increased risk of gastrointestinal bleeding), estrogen, ethanol; increases the hypoglycemic effect of sulfonylurea derivatives.
Antacids and colestiramine decrease absorption of ibuprofen.
Enhances blood concentrations of digoxin, lithium drugs and methotrexate.
It increases the effect of m-cholinoblockers, H1-histamine receptor blockers, butyrophenones, phenothiazines, amantadine and quinidine.
The concomitant administration of other NSAIDs increases the frequency of side effects.
Caffeine increases the analgesic (analgesic) effect.
When taken concomitantly it reduces anti-inflammatory and antiaggregant effect of acetylsalicylic acid (increased incidence of acute coronary failure is possible in patients receiving low doses of acetylsalicylic acid as an antiaggregant after initiation of Novigan® administration).
Cefamandol, cefoperazone, cefotetan, valproic acid, plikamycin increase the incidence of hypoprothrombinemia when administered simultaneously.
Myelotoxic drugs increase the manifestation of hematotoxicity of the drug.
Cyclosporine and gold drugs increase the effect of ibuprofen on PG synthesis in kidneys, which is manifested by increased nephrotoxicity. Ibuprofen increases the plasma concentration of cyclosporine and the likelihood of its hepatotoxic effects.
Like drugs that block tubular secretion decrease excretion and increase the plasma concentration of ibuprofen.
Special Instructions
Special Instructions
In case of long-term use it is necessary to monitor peripheral blood picture and functional state of the liver and kidneys.
To reduce the risk of gastrointestinal tract adverse events, the lowest effective dose should be used. In case of gastropathy symptoms it is required thorough control including esophagogastroduodenoscopy, blood test with hemoglobin and hematocrit determination, fecal occult blood test.
If 17-ketosteroids determination is required the preparation should be discontinued 48 hours prior to the test.
During the treatment the patient should refrain from taking alcohol.
During the treatment the patient should refrain from potentially dangerous activities that require high concentration and rapid psychomotor reactions.
Synopsis
Synopsis
Round biconvex white film-coated tablets with NOVIGAN embossing on one side.
Appearance on cross section: white to almost white.
Contraindications
Contraindications
Hypersensitivity to any of the ingredients included in the drug; erosive-ulcerative changes of the mucous membrane of the stomach or duodenum, active gastrointestinal bleeding; inflammatory bowel disease in the acute phase, including ulcerative colitis; anamnestic evidence of an attack of bronchoobstruction, rhinitis, urticaria, after taking acetylsalicylic acid or other non-steroidal anti-inflammatory drug – NSAIDs (complete or incomplete acetylsalicylic acid intolerance syndrome – rhinosinusitis, urticaria, nasal polyps, bronchial asthma); hepatic insufficiency or active liver disease; renal insufficiency (creatinine clearance less than 30 ml/min).), advanced renal disease; confirmed hyperkalemia; hemophilia and other disorders of blood clotting (including hypocoagulation), hemorrhagic diathesis; period after coronary artery bypass grafting; acute “intermittent” porphyria; Granulocytopenia; hematopoiesis disorders; glucose-6-phosphate dehydrogenase deficiency; tachyarrhythmia; closed-angle glaucoma; optic nerve disease; prostatic hyperplasia; intestinal obstruction; pregnancy and lactation, age under 16 years old.
With caution:
Side effects
Side effects
Novigan® does not cause side effects in the recommended doses.
Gastrointestinal tract: NSAID gastropathy (abdominal pain, nausea, vomiting, heartburn, decreased appetite, diarrhea, flatulence, constipation; gastrointestinal mucosal ulcerations, which in some cases are complicated by perforation and bleeding; irritation or dryness of oral mucosa, mouth pain, gum mucosal ulceration, aphthous stomatitis, pancreatitis.
Hepatobiliary system: hepatitis.
Respiratory system: shortness of breath, bronchospasm.
Senses: hearing disorders: decreased hearing, ringing or tinnitus; vision disorders: toxic optic nerve injury, blurred vision, scotoma, dry and irritated eyes, conjunctival and eyelid edema (allergic genesis), accommodation paresis.
Central and peripheral nervous system disorders: headache, dizziness, insomnia, anxiety, nervousness and irritability, psychomotor agitation, drowsiness, depression, confusion, hallucinations, aseptic meningitis (more often in patients with autoimmune diseases).
Cardiovascular system: heart failure, tachycardia, increased blood pressure.
Urinary system: acute renal failure, allergic nephritis, nephrotic syndrome (edema), oliguria, anuria, polyuria, proteinuria, cystitis, urine staining red.
Allergic reactions: Skin rash (usually erythematous or urticaria), skin itching, Quincke’s edema, anaphylactoid reactions, anaphylactoid shock, bronchospasm or dyspnea, fever, erythema multiforme exudative (including Stevens-Johnson syndrome), toxic epidermal necrolysis (Lyell syndrome), eosinophilia, allergic rhinitis.
Blood organs: anemia (including hemolytic, aplastic), thrombocytopenia and thrombocytopenic purpura, agranulocytosis, leukopenia.
Other: increased or decreased sweating.
Laboratory parameters: bleeding time (may increase), serum glucose concentration (may decrease), creatinine clearance (may decrease), hematocrit or hemoglobin (may decrease), serum creatinine concentration (may increase), liver transaminase activity (may increase).
If taking the drug has caused a change in your usual condition, stop taking it and see your doctor immediately.
Overdose
Overdose
Symptoms: abdominal pain, nausea, vomiting, lethargy, drowsiness, depression, headache, tinnitus, metabolic acidosis, coma, acute renal failure, decreased BP, bradycardia, tachycardia, atrial fibrillation, respiratory arrest.
The treatment: gastric lavage (only within one hour after ingestion), prescription of activated charcoal, alkaline drinking, forced diuresis, symptomatic therapy (correction of acid-base status, BP). There is no specific antidote to the drug.
Do not exceed the recommended doses. If the recommended doses are exceeded, seek immediate medical attention or contact the nearest medical facility (bring the drug package with you).
Similarities
Similarities
Spazgan Neo
Additional information
| Weight | 0.022 kg |
|---|---|
| Shelf life | 5 years |
| Conditions of storage | At a temperature not exceeding 25 C. Keep out of reach of children! |
| Manufacturer | Dr. Reddy's, India |
| Medication form | pills |
| Brand | Dr. Reddy's |
Other forms…
Related products
Buy Novigan, 20 pcs. with delivery to USA, UK, Europe and over 120 other countries.