No products in the cart.
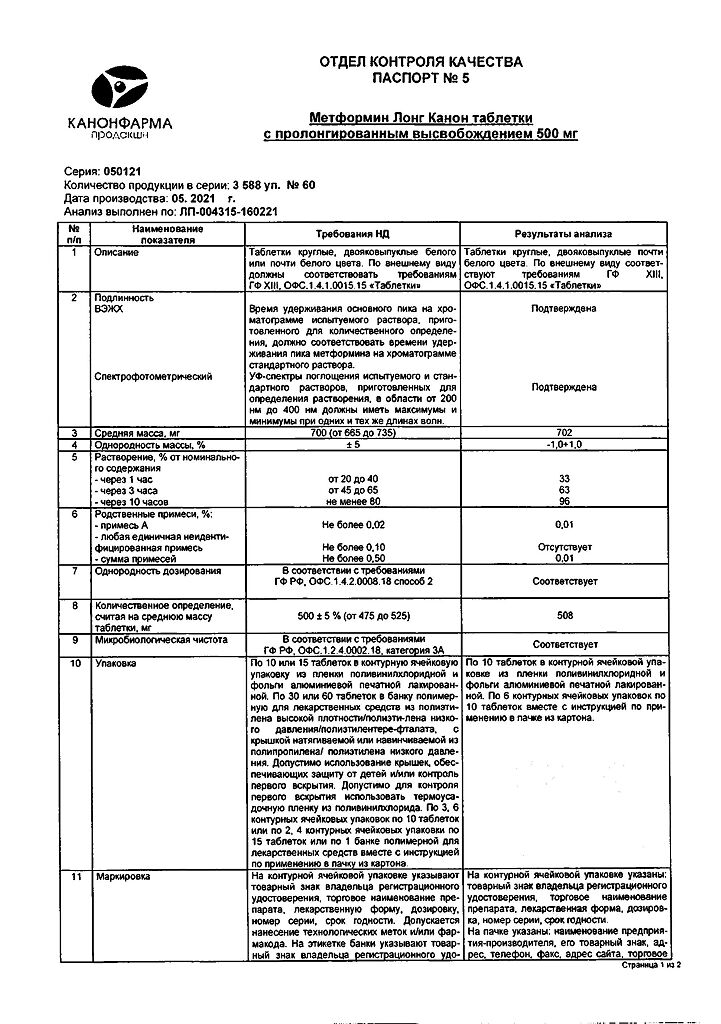
Metformin Long Canon, 500 mg 60 pcs
€8.51 €7.09
Out of stock
(E-mail when Stock is available)
Description
Orally administered hypoglycemic drug from the group of biguanides (dimethylbiguanide). The mechanism of action of metformin is related to its ability to inhibit gluconeogenesis, as well as the formation of free fatty acids and fat oxidation. It increases the sensitivity of peripheral receptors to insulin and glucose utilization by cells. Metformin does not affect the amount of insulin in blood, but changes its pharmacodynamics by reducing the ratio of bound to free insulin and increasing the ratio of insulin to proinsulin.
Metformin stimulates glycogen synthesis by acting on glycogen synthetase. It increases transport capacity of all types of membrane glucose transporters. Delays absorption of glucose in the intestine.
Limits triglycerides, LDL, LDLNP. Metformin improves blood fibrinolytic properties by inhibiting tissue-type plasminogen activator inhibitor.
A patient’s body weight either remains stable or decreases moderately while taking metformin.
Pharmacokinetics
After oral administration metformin is slowly and partially absorbed from the gastrointestinal tract. Cmax in plasma is reached after approximately 2.5 hours. At a single dose of 500 mg absolute bioavailability is 50-60%. Absorption of metformin is decreased and delayed with concomitant food intake.
Metformin is rapidly distributed in body tissues. Practically does not bind with plasma proteins. It accumulates in the salivary glands, liver and kidneys.
Extracted by the kidneys unchanged. T1/2 from plasma is 2-6 hours.
In impaired renal function, metformin may be cumulated.
Indications
Indications
Type 2 diabetes mellitus (insulin-independent) with ineffectiveness of diet therapy and physical activity, in patients with obesity: in adults – as monotherapy or in combination with other oral hypoglycemic agents or with insulin;
Active ingredient
Active ingredient
Metformin
Composition
Composition
Prolonged-release tablets are white or almost white, oval, biconvex.
| 1 tablet | ||
| metformin hydrochloride | 500 mg |
Associates:
Hypromellose 2208 (hydroxypropyl methylcellulose 2208),
hypromellose (hydroxypropyl methylcellulose) 2910,
colloidal silicon dioxide,
magnesium stearate,
microcrystalline cellulose.
How to take, the dosage
How to take, the dosage
It is taken orally, during or after meals.
The dose and frequency of administration depend on the dosage form used.
In monotherapy the initial single dose for adults is 500 mg, depending on the dosage form, the frequency of administration is 1-3 times per day. It is possible to use 850 mg 1-2 times a day. If necessary, the dose is gradually increased at 1 week intervals to 2-3 g/day.
In monotherapy for children aged 10 years and older, the starting dose is 500 mg or 850 once daily or 500 mg twice daily. If necessary, at intervals of at least 1 week, the dose may be increased to a maximum of 2 g/day in 2-3 doses.
In 10-15 days, the dose should be adjusted on the basis of the results of blood glucose determination.
In combination therapy with insulin, the initial dose of metformin is 500-850 mg 2-3 times daily. The dose of insulin is adjusted on the basis of the results of blood glucose determination.
Interaction
Interaction
In concomitant use with sulfonylurea derivatives, acarbose, insulin, salicylates, MAO inhibitors, oxytetracycline, ACE inhibitors, with clofibrate, cyclophosphamide may increase the hypoglycemic effect of metformin.
Concomitant use with GCS, oral hormonal contraceptives, danazolol, epinephrine, glucagon, thyroid hormones, phenothiazine derivatives, thiazide diuretics, nicotinic acid derivatives may decrease the hypoglycemic effects of metformin.
In patients receiving metformin, the use of iodine-containing contrast agents for diagnostic studies (including IV urography, IV cholangiography, angiography, CT) increases the risk of acute renal dysfunction and lactate acidosis. These combinations are contraindicated.
Beta2-adrenomimetics in the form of injections increase blood glucose concentration due to stimulation of β2-adrenoreceptors. In this case it is necessary to control the blood glucose concentration. If necessary, it is recommended to prescribe insulin.
The concomitant use of cimetidine may increase the risk of lactate acidosis.
The concomitant use of “loop” diuretics may lead to the development of lactate acidosis due to possible functional renal failure.
The concomitant use with ethanol increases the risk of lactate acidosis.
Nifedipine increases absorption and Cmax of metformin.
Cationic drugs (amiloride, digoxin, morphine, procainamide, quinidine, quinine, ranitidine, triamterene, trimethoprim and vancomycin) that are secreted in the renal tubules compete with metformin for tubular transport systems and may lead to increased Cmax.
Special Instructions
Special Instructions
The use is not recommended in acute infections, exacerbation of chronic infectious and inflammatory diseases, trauma, acute surgical diseases, risk of dehydration.
Do not use before and within 2 days after surgery.
With caution, metformin should be used in elderly patients and those who perform strenuous physical work due to the increased risk of lactic acidosis. Asymptomatic impairment of renal function is often observed in elderly patients. Particular caution is required if impaired renal function is provoked by taking antihypertensive drugs or diuretics, as well as NSAIDs.
If during treatment the patient develops muscle cramps, digestive disorders (abdominal pain), and severe asthenia, it should be kept in mind that these symptoms may indicate the onset of lactacidosis.
Key renal function should be monitored during treatment; plasma lactate should be determined at least twice a year, and also if myalgia occurs.
Hypoglycemia usually does not occur when metformin is used as monotherapy according to the dosing regimen. However, when combined with insulin or with sulfonylurea derivatives, there is a risk of hypoglycemia. In such cases, especially close control of blood glucose concentration is necessary.
Patients should avoid alcohol consumption during treatment because of the risk of lactate acidosis.
Preclinical studies have shown that metformin has no carcinogenic potential.
Contraindications
Contraindications
Acute or chronic metabolic acidosis, diabetic ketoacidosis, diabetic precoma and coma; renal failure, impaired renal function (CKR less than 60 ml/min);
Side effects
Side effects
Digestive system disorders: nausea, vomiting, diarrhea, flatulence, abdominal discomfort are possible (usually at the beginning of treatment); in single cases – disorders of liver function parameters, hepatitis (disappear after discontinuation of treatment).
Metabolic disorders: very rare – lactate acidosis (discontinuation of treatment is required).
Hematopoietic system disorders: very rare – disorders of vitamin B12 absorption.
The profile of adverse reactions in children aged 10 years and older is the same as in adults.
Similarities
Similarities
Siofor 500, Siofor 850, Metformin-Teva, Siofor 1000, Glucofage Long, Formetin, Metformin, Merifatin
Additional information
| Weight | 0.076 kg |
|---|---|
| Manufacturer | Kanonfarma Production ZAO, Russia |
| Medication form | sustained release tablets |
| Brand | Kanonfarma Production ZAO |
Related products
Buy Metformin Long Canon, 500 mg 60 pcs with delivery to USA, UK, Europe and over 120 other countries.