No products in the cart.
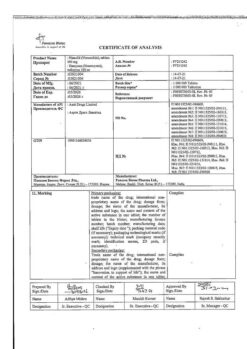
Ketorol, 30 mg/ml 1 ml 10 pcs
€4.25 €3.78
Description
Pharmacodynamics
Non-steroidal anti-inflammatory drug (NSAID), has a pronounced analgesic effect, anti-inflammatory and moderate antipyretic effect.The mechanism of action is related to non-selective inhibition of cyclooxygenase (COX) – COX-1 and COX-2, catalyzing formation of prostaglandins from arachidonic acid, which play an important role in the pathogenesis of pain, inflammation and fever.
Ketorolac is a racemic mixture of [-]S and [+]R enantiomers, with analgesic effect due to the [-]S form. By the analgesic effect it is comparable with morphine and is superior to other NSAIDs.
The drug does not affect the opioid receptors, does not inhibit respiration, does not cause drug addiction, has no sedative and anxiolytic effects.
Pharmacokinetics
The pharmacokinetics of ketorolac after single and multiple intravenous and intramuscular administration is linear.
When administered intramuscularly absorption is complete and rapid. Maximal concentration of the preparation (Cmax) after intramuscular introduction of 30 mg is 1.74-3.1 µg/ml, after 60 mg – 3.23-5.77 µg/ml; time of reaching the maximum concentration (TCmax) is 15-73 and 30-60 minutes, respectively. Cmax after intravenous administration of 15 mg – 1.96-2.98 mcg/ml, 30 mg – 3.69-5.61 mcg/ml, TCmax – 0.4-1.8 min and 1.1-4.7 min, respectively.
The binding to plasma proteins is 99%. Time of reaching of equilibrium concentration (Css) in parenteral administration of 30 mg 4 times per day – 24 hours; in intramuscular administration of 15 mg – 0.65-1.13 mkg/ml, of 30 mg – 1.29-2.47 mkg/ml.
Distribution volume (Vd) in intramuscular administration – 0.136-0.214 l/kg, in intravenous – 0.166-0.254 l/kg. In patients with renal insufficiency the drug distribution volume may increase twice, and the distribution volume of its R-enantiomer – by 20%.
Indications
Indications
Pain syndrome of strong and moderate severity of different origin in trauma, toothache, pain in the postoperative period, in cancer and rheumatic diseases, myalgia, arthralgia, neuralgia, radiculitis.
Preferred to symptomatic therapy, to reduce pain and inflammation at the time of use. It has no effect on the progression of the disease.
Active ingredient
Active ingredient
Ketorolac
Composition
Composition
1 ml of the solution contains:
the active ingredient:.
How to take, the dosage
How to take, the dosage
For direct use, put 1-2 drops in each eye and blink several times.
For indirect use, put one or two drops on the inner surface of each contact lens.
Avizor MOISTURE DROPS can be used as needed. Wash your hands thoroughly with soap and water before use.
Interaction
Interaction
Concomitant use of ketorolac with acetylsalicylic acid or other NSAIDs, calcium preparations, GCS, ethanol, corticotropin may lead to the formation of GI ulcers and development of gastrointestinal bleeding.
The drug should not be used simultaneously with other NSAIDs.
Concomitant administration with paracetamol increases nephrotoxicity, with methotrexate – hepatotoxicity and nephrotoxicity.
Simultaneous administration of ketorolac and methotrexate is only possible with low doses of the latter (methotrexate plasma concentration must be monitored).
Probenecid decreases plasma clearance and Vd of ketorolac, increases its plasma concentration and increases T1/2.
Ketorolac can decrease clearance of methotrexate and lithium and increase toxicity of these substances.
Simultaneous administration with indirect anticoagulants, heparin, thrombolytics, antiaggregants, cefoperazone, cefotetan and pentoxifylline increases the risk of bleeding.
Reduces the effect of hypotensive and diuretic drugs (reduces the synthesis of PG in the kidneys).
When combined with narcotic analgesics the doses of the latter may be significantly reduced.
Antacids do not affect the completeness of drug absorption.
Increases hypoglycemic effect of insulin and oral hypoglycemic drugs (requires dose recalculation).
Concomitant administration with valproic acid causes platelet aggregation disorders.
Increases plasma concentration of verapamil and nifedipine.
When prescribed with other nephrotoxic drugs (including gold drugs) the risk of nephrotoxicity increases. Drugs that block tubular secretion reduce clearance of ketorolac and increase its concentration in blood plasma.
Special Instructions
Special Instructions
Ketorol® has two dosage forms (film-coated tablets and solution for intravenous and intramuscular administration). The choice of the method of drug administration depends on the severity of the pain syndrome and the condition of the patient.
Before prescribing the drug it is necessary to find out about the previous allergic reaction to the drug or NSAIDs. Due to the risk of allergic reactions the first dose is administered under the close supervision of a physician.
Hypovolemia increases the risk of nephrotoxic adverse reactions.
If necessary the drug may be administered in combination with narcotic analgesics.
It is not recommended to use the drug as a prophylactic analgesic before and during major surgical interventions due to the high risk of bleeding. The drug should not be used together with NSAIDs (including cyclooxygenase-2 inhibitors), because when combined with other NSAIDs fluid retention, decompensation of cardiac activity, increased blood pressure may be observed. The effect on platelet aggregation ceases after 24-48 hours.
The drug may alter the properties of platelets.
Patients with blood clotting disorders are prescribed only with continuous monitoring of platelet count, especially important for post-surgical patients who require close monitoring of hemostasis.
The risk of drug complications increases with prolongation of treatment (in patients with chronic pain) and increasing the drug dose more than 90 mg/day.
To decrease the risk of adverse events the lowest effective dose should be used for the shortest possible course.
To decrease the risk of NSAID gastropathy misoprostol, omeprazole are prescribed.
During treatment, caution should be exercised when driving motor transport and engaging in other potentially hazardous activities that require increased concentration and rapid psychomotor reactions.
Contraindications
Contraindications
- High sensitivity to ketorolac;
- complete or incomplete combination of bronchial asthma, recurrent nasal or paranasal sinus polyposis and intolerance to acetylsalicylic acid and other NSAIDs (including history);
- erosive ulcerative changes of the mucous membrane of the stomach and duodenum, active gastrointestinal bleeding; cerebrovascular or other bleeding;
- inflammatory bowel disease (Crohn’s disease, ulcerative colitis) in the acute phase;
- hemophilia and other blood clotting disorders;
- decompensated heart failure;
- hepatic failure or active liver disease;
- explicit renal insufficiency (creatinine clearance less than 30 ml/min), progressive renal disease, confirmed hyperkalemia;
- postoperative period after aortocoronary bypass;
- Simultaneous use with probenecid, pentoxifylline, acetylsalicylic acid and other NSAIDs (including cyclooxygenase-2 inhibitors), lithium salts, anticoagulants (including warfarin and heparin);
- The drug is not used for prophylactic analgesia before and during major surgical procedures because of the high risk of bleeding;
- pregnancy, childbirth, lactation;
- children under 16 years (safety and effectiveness not established).
With caution:
Bronchial asthma, coronary heart disease, congestive heart failure, edema syndrome, arterial hypertension, cerebrovascular disease, abnormal dyslipidemia or hyperlipidemia, renal function disorders (creatinine clearance 30-60 ml/l), diabetes, cholestasis, sepsis, systemic lupus erythematosus,
peripheral arterial disease, smoking, older age (over 65 years), anamnestic data on the development of gastrointestinal ulcers, alcohol abuse, severe somatic diseases, concomitant therapy with the following drugs antiaggregants (e.g., clopidogrel), oral glucocorticosteroids (e.g., prednisolone), selective serotonin reuptake inhibitors (e.g., citalopram, fluoxetine, paroxetine, sertraline).
Side effects
Side effects
The frequency of side effects is classified according to the frequency of the case: often (1-10%), sometimes (0.1-1%), rarely (0.01-0.1%), very rarely (less than 0.01%), including individual reports.
Digestive system disorders: Frequent (especially in elderly patients older than 65 years with a history of gastrointestinal erosive ulcerative lesions) – gastralgia, diarrhea; less frequent – stomatitis, flatulence, constipation, vomiting, feeling of stomach fullness; rarely – nausea, erosive-ulcerative lesions of the gastrointestinal tract (including with perforation and/or bleeding – abdominal pain, epigastric spasm or burning, melena, “coffee grounds” type vomiting, nausea, heartburn and others), cholestatic jaundice, hepatitis, hepatomegaly, acute pancreatitis.
Urinary system disorders: rare – acute renal failure, low back pain with or without hematuria and/or azotemia, hemolytic-uremic syndrome (hemolytic anemia, renal failure, thrombocytopenia, purpura), frequent urination, increased or decreased urine volume, nephritis, edema of renal genesis.
Senses: rare – hearing loss, tinnitus, visual impairment (including blurred vision).
Respiratory system: rare – bronchospasm, rhinitis, laryngeal edema (shortness of breath, difficulty in breathing).
Central nervous system: often – headache, dizziness, somnolence; rarely – aseptic meningitis (fever, severe headache, cramps, neck and/or back stiffness), hyperactivity (mood changes, anxiety), hallucinations, depression, psychosis.
Cardiovascular system: less frequently – increase of blood pressure; rarely – pulmonary edema, fainting.
Blood: rare – anemia, eosinophilia, leukopenia.
Hemostasis system: rare – bleeding from the post-operative wound, nasal bleeding, rectal bleeding.
Skin side: less frequent – skin rash (including maculopapular rash), purpura; rarely – exfoliative dermatitis (fever with or without chills, redness, thickening or peeling of the skin, swelling and/or painful palatine tonsils), urticaria, Stevens-Johnson syndrome, Lyell syndrome.
Local reactions: less frequent – burning or pain at the injection site.
Allergic reactions: rare – anaphylaxis or anaphylactoid reactions (changes in complexion, skin rash, urticaria, skin itching, shortness of breath, edema of the eyelids, periorbital edema, difficulty in breathing, heaviness in the chest, wheezing).
Other: often – edema (face, shins, ankles, fingers, feet, weight gain); less frequently – increased sweating; rarely – tongue swelling, fever.
Overdose
Overdose
Symptoms: abdominal pain, nausea, vomiting, gastrointestinal erosive and ulcerative lesions, renal dysfunction, metabolic acidosis.
Treatment: gastric lavage, administration of adsorbents (activated charcoal) and symptomatic therapy (maintenance of vital body functions). Not eliminated sufficiently by dialysis.
Similarities
Similarities
Dolac, Ketorol, Ketonov, Ketorolac, Ketorolac Rompharm
Additional information
| Weight | 0.029 kg |
|---|---|
| Shelf life | 3 years |
| Conditions of storage | In the dark place at a temperature not exceeding 25 ° C. Do not freeze. Keep out of reach of children! |
| Manufacturer | Dr. Reddy's, India |
| Medication form | solution |
| Brand | Dr. Reddy's |
Other forms…
Related products
Buy Ketorol, 30 mg/ml 1 ml 10 pcs with delivery to USA, UK, Europe and over 120 other countries.