No products in the cart.
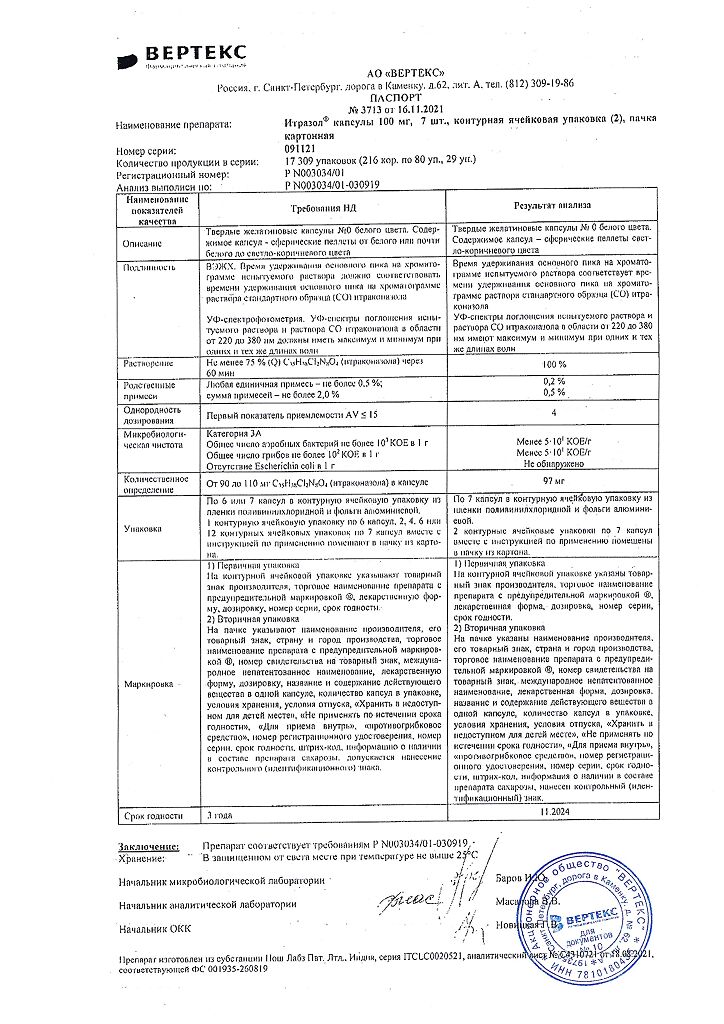
Itrazol, 100 mg capsules 14 pcs
€28.58 €24.77
Description
Itraconazole is an antifungal.
Pharmacodynamics
Itraconazole is a synthetic broad-spectrum antifungal agent, a triazole derivative. It inhibits the synthesis of the cell membrane ergosterol of fungi, which determines the antifungal effect of the drug.
Itraconazole is active against infections caused by dermatophytes (Trichophyton spp.,Microsporum spp., Epidermophyton floccosum), yeast-like fungi and yeasts (Cryptococcus neoformans, Pityrosporum spp., Candida spp. including C albicans, C. glabrata, and C. krusei); Aspergillus spp., Histoplasma spp., Paracoccidioides brasiliensis, Sporothrix schenckii, Fonsecaea spp., Cladosporium spp., Blastomyces dermatidis, and other yeasts and molds.
Pharmacokinetics
It is absorbed from the gastrointestinal tract quite completely. Taking itraconazole in capsules immediately after a meal increases bioavailability. Cmax in plasma is reached within 3-4 hours after oral administration. Css when taking 100 mg of the drug once a day is 0.4 mcg/ml; when taking 200 mg once a day – 1.1 mcg/ml, 200 mg 2 times a day – 2 mcg/ml.
The time of onset of Css in plasma with prolonged use is 1-2 weeks. The binding to plasma proteins is 99.8%.
It penetrates well into tissues and organs (including vaginal mucosa), is contained in the secretion of sebaceous and sweat glands. Concentration of itraconazole in lungs, kidneys, liver, bones, stomach, spleen, skeletal muscles is 2-3 times higher than its concentration in plasma; in tissues containing keratin – 4 times higher.
Therapeutic concentration of itraconazole in skin is maintained for 2-4 weeks after discontinuation of 4-week course of treatment. The therapeutic concentration in the keratin of the nails is reached 1 week after the start of treatment and is maintained for 6 months after the completion of the 3-month course of treatment. Low concentrations are determined in the sebaceous and sweat glands of the skin.
It is metabolized in the liver with the formation of active metabolites, including hydroxyitraconazole. It is an inhibitor of CYP3A4, CYP3A5 and CYP3A7 isoenzymes.
Indications
Indications
Active ingredient
Active ingredient
Composition
Composition
1 capsule contains:
The active ingredient:
itraconazole 100 mg;
Excipients:
sugar pellets (sucrose – 80-91.5%, corn starch – 8.5-20%) – 207.44 mg;
poloxamer 188 (Lutrol) – 25.94 mg;
poloxamer 188 (Lutrol) micronized – 0.51 mg;
hypromellose – 130.11 mg.
How to take, the dosage
How to take, the dosage
Itrazol capsules are taken orally after meals.
Onychomycosis – 200 mg 1 time per day for 3 months or 200 mg 2 times per day for 1 week with a 3 week break; 3 courses of treatment are recommended for feet onychomycosis, 2 courses for hands.
Vulvovaginal candidiasis – 200 mg 2 times per day for 1 day or 200 mg 1 time per day for 3 days; Varicella – 200 mg 1 time per day for 7 days;
Dermatomycoses and oral candidiasis – 100-200 mg 1 time per day for 7-15 days (repeat course if necessary);
Fungal keratitis – 200 mg once daily for 21 days; systemic mycoses – 100-200 mg 1-2 times daily for 2-12 months (depending on the causative agent).
Interaction
Interaction
Drugs affecting the metabolism of itraconazole
The interaction of itraconazole with rifampicin, rifabutin and phenytoin has been studied. Concomitant use of itraconazole with these drugs, which are potential inducers of liver enzymes, is not recommended. Studies of interaction with other inducers of liver enzymes, such as carbamazepine, phenobarbital and isoniazid, have not been conducted, but similar results can be assumed due to the fact that itraconazole is mainly metabolized by CYP3A4 enzyme, the potent inhibitors of this enzyme can increase the bioavailability of itraconazole. Examples are ritonavir, indinavir, clarithromycin and erythromycin.
The effect of itraconazole on the metabolism of other drugs
Itraconazole may inhibit the metabolism of drugs cleaved by CYP3A4 enzyme. This may result in intensification or prolongation of their effects, including side effects.
Drugs that should not be taken concomitantly with itraconazole
– Terfenadine, astemizole, misolastin, cisapride, triazolam and oral midazolam, dofetilide, quinidine, pimozide, HMG-CoA reductase inhibitors such as simvastatin and lovastatin;
– BCAAs may have a negative inotropic effect, which may exacerbate the same effect exhibited by itraconazole. Caution should be exercised when concomitant administration of itraconazole and BCAA, as metabolism of BCAA may be reduced.
Drugs for which plasma concentrations and effects, side effects should be monitored when prescribing
The dose of the following drugs should be decreased, if necessary, when concomitantly prescribed with itraconazole:
– oral anticoagulants;
– HIV protease inhibitors such as ritonavir, indinavir, saquinavir;
– Certain anticancer drugs, such as periwinkle pink alkaloids, busulfan, docetaxel, trimetrexate;
– BCAs that are cleaved by the CYP3A4 enzyme, such as verapamil;
– Certain immunosuppressive drugs: cyclosporine, tacrolimus, sirolimus;
– other drugs: digoxin, carbamazepine, buspirone, alfentanil, alprazolam, brotizolam, rifabutin, methylprednisolone, ebastine, reboxetine.
The interaction between itraconazole and zidovudine and fluvastatin has not been found.
The effect of itraconazole on the metabolism of ethinylestradiol and norethisterone was not observed.
In vitro studies have demonstrated no competition between itraconazole and such drugs as imipramine, propranolol, diazepam, cimetidine, indomethacin, tolbutamide and sulfadimidine in binding to plasma proteins.
Special Instructions
Special Instructions
Women of childbearing age taking Itrazol® should use adequate contraceptive measures throughout the course of treatment until the first menstrual period after completion.
In an IV study of itraconazole, a transient asymptomatic decrease in left ventricular ejection fraction was observed, which normalized before the next infusion of the drug.
Itraconazole has a negative inotropic effect. Cases of heart failure associated with itraconazole administration have been reported. Itraconazole should not be taken in patients with CHF or with a history of this disease, except in cases when the possible benefit significantly exceeds the potential risk.
The BCCs may have a negative inotropic effect, which may enhance the similar effect of itraconazole; itraconazole may decrease metabolism of BCCs. Caution should be exercised when concomitant administration of itraconazole and BCAA.
In reduced gastric acidity, absorption of itraconazole is impaired. Patients who are taking antacids (e.g. aluminum hydroxide), it is recommended to use them not earlier than 2 hours after taking Itrazol® capsules. Patients with achlorhydria or who use H2-histamine receptor blockers or proton pump inhibitors are recommended to take Itrazol® capsules with acidic beverages.
When itraconazole is used for a long time (more than 1 month), when itraconazole is used by patients receiving other drugs with hepatotoxic effect, as well as by patients with liver diseases it is recommended to monitor the liver function regularly. Patients should be warned to contact their physician immediately in case of symptoms suggestive of hepatitis, namely: anorexia, nausea, vomiting, weakness, abdominal pain and darkened urine. If these symptoms occur, therapy should be stopped immediately and liver function tests should be performed.
The bioavailability of itraconazole may be reduced in patients with renal impairment; therefore, the dose should be adjusted.
The treatment should be stopped if neuropathy occurs, which may be associated with the intake of Itrazol® capsules. There are no data on cross-sensitivity to itraconazole and other azole antifungal drugs. Itrazol® capsules should be used with caution in patients with hypersensitivity to other azoles.
In patients with impaired immunity (AIDS, after organ transplantation, neutropenia) the dose of Itrazol® may require increasing.
Contraindications
Contraindications
With caution: severe heart failure; liver disease (including those with hepatic insufficiency). Itrazol® is recommended to be used in children over 3 years old only if possible benefit exceeds potential risk.
Side effects
Side effects
Gastrointestinal disorders: dyspepsia, nausea, abdominal pain and constipation, reversible increase in liver enzyme activity, cholestatic jaundice, hepatitis, anorexia. In very rare cases during the use of Itrazol® preparation severe toxic liver injury occurred, including a case of acute hepatic failure with fatal outcome.
CNS disorders: headache, fatigue, dizziness, peripheral neuropathy.
Systemic diseases: congestive heart failure and pulmonary edema.
Other organs and systems: menstrual disorders, allergic reactions (such as itching, rash, urticaria and angioedema), Stevens-Johnson syndrome, alopecia, hypokalemia, edema, dark urine staining, hypercreatininemia.
Overdose
Overdose
No data available.
Treatment: gastric lavage within the first hour and, if necessary, administration of activated charcoal, symptomatic treatment. Itraconazole is not excreted by hemodialysis. There is no specific antidote to the drug.
Similarities
Similarities
Additional information
| Weight | 0.020 kg |
|---|---|
| Shelf life | 2.5 years |
| Conditions of storage | In a light-protected place, at a temperature not exceeding 25 °C |
| Manufacturer | Vertex, Russia |
| Medication form | capsules |
| Brand | Vertex |
Other forms…
Related products
Buy Itrazol, 100 mg capsules 14 pcs with delivery to USA, UK, Europe and over 120 other countries.