No products in the cart.
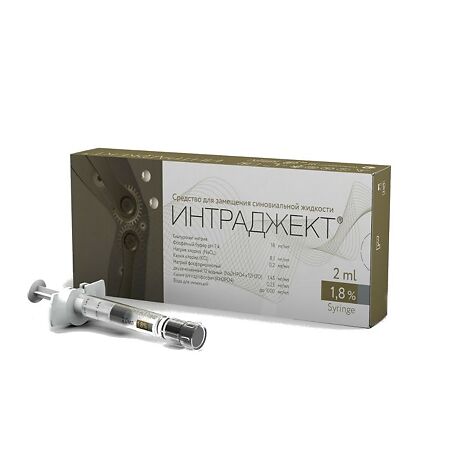
Intraject 1.8% 2 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
Hyaluronic acid is a natural polysaccharide belonging to the class of glycosaminoglycans, which is part of all body tissues and is the most important structural element of synovial fluid. Hyaluronic acid is unmodified.
The product INTRAJECT® contains
Hyaluronic acid sodium salt and water, which molecules form hydrogen bonds due to the presence of
./in the structure of the molecules of hydroxyl groups (OH-groups), as well as due to the presence of a large number of nitrogen atoms in the hyaluronic acid molecule. Due to the formation of hydrogen bonds with water molecules, hyaluronic acid has a high ability to bind and retain water, acquiring a jelly-like structure.
Macromolecules of hyaluronic acid, thanks to the same mechanisms, form intramolecular and intermolecular interactions, giving a fairly stable three-dimensional structure.
The INTRAGEKT® product has an organized gel structure capable of holding hyaluronic acid and water molecules within the synovial sac and providing stability of the INTRAGEKT® product, which guarantees the prolonged action of the product.
The viscoelastic properties of hyaluronic acid in synovial fluid provide lubrication and cushioning of articular surfaces. Synovial fluid in joints with degenerative diseases (osteoarthritis) or traumatic joint changes, has lower viscosity and elasticity than in healthy joints. Hyaluronic acid, a component of INTRAGEKT®, has a molecular weight of 2.2 million daltons, close to natural human hyaluronic acid, and has a positive effect after injection in the diagnosis of osteoarthritis (osteoarthritis). Intra-articular injection of INTRAJECT® restores physiological and rheological properties of synovial fluid of the joint affected by osteoarthritis. As a consequence, it reduces pain and discomfort and improves mobility in the joint.
Indications
Indications
INTRAJECT® is intended for intra-articular administration only.
INTRAJECT® is used:
1. For temporary replacement and replenishment of synovial fluid.
2. To relieve pain and stiffness caused by degenerative-dystrophic (osteoarthritis or osteoarthritis) and traumatic changes in the knee, hip and other synovial joints.
3. For use in patients who lead an active lifestyle and regularly load the damaged joint.
Pharmacological effect
Pharmacological effect
Hyaluronic acid is a natural polysaccharide belonging to the class of glycosaminoglycans, which is part of all tissues of the body and is the most important structural element of synovial fluid. Hyaluronic acid is unmodified.
INTRAJECT® product contains
sodium salt of hyaluronic acid and water, the molecules of which form hydrogen bonds due to the presence
in the structure of molecules of hydroxyl groups (OH-groups), as well as the presence of a large number of oxygen and nitrogen atoms in the hyaluronic acid molecule. Due to the formation of hydrogen bonds with water molecules, hyaluronic acid has a high ability to bind and retain water, acquiring a jelly-like structure.
Macromolecules of hyaluronic acid, thanks to the same mechanisms, form intramolecular and intermolecular interactions, giving a fairly stable three-dimensional structure.
The INTRAJECT® product has an ordered gel structure, which is capable of retaining molecules of hyaluronic acid and water inside the synovial bursa, ensuring the stability of the INTRAJECT® product, which guarantees the prolonged action of the product.
The viscoelastic properties of hyaluronic acid, which is part of the synovial fluid, provide lubrication and shock absorption of the articular surfaces. Synovial fluid in joints with degenerative diseases (osteoarthritis) or traumatic changes in the joints has lower viscosity and elasticity than in healthy joints. Hyaluronic acid, which is part of INTRAJECT®, has a molecular weight of 2.2 million daltons, close to natural human hyaluronic acid, and has a positive effect after administration for a diagnosis of osteoarthritis (osteoarthritis). Intra-articular administration of INTRAJECT® restores the physiological and rheological properties of the synovial fluid of a joint affected by osteoarthritis. As a result, pain and discomfort are reduced and mobility in the joint is improved.
Special instructions
Special instructions
Before using a medical device, consultation with a doctor is required.
Before using INTRAJECT® Products, you must carefully read the instructions for use.
When preparing for use and during the actual use of a medical device, strict adherence to the rules of asepsis is required, because possible risk of infection.
Intravascular administration of INTRAJECT® is unacceptable.
Avoid getting INTRAJECT® into the joint capsule, surrounding tissue or blood vessels.
Within 48-72 hours after the injection of INTRAJECT® into the joint, it is not recommended to carry out physical procedures, taping, compresses with medications, prolonged static stress on the limb, do not take a deep squat position, avoid running, jumping, and carrying heavy objects.
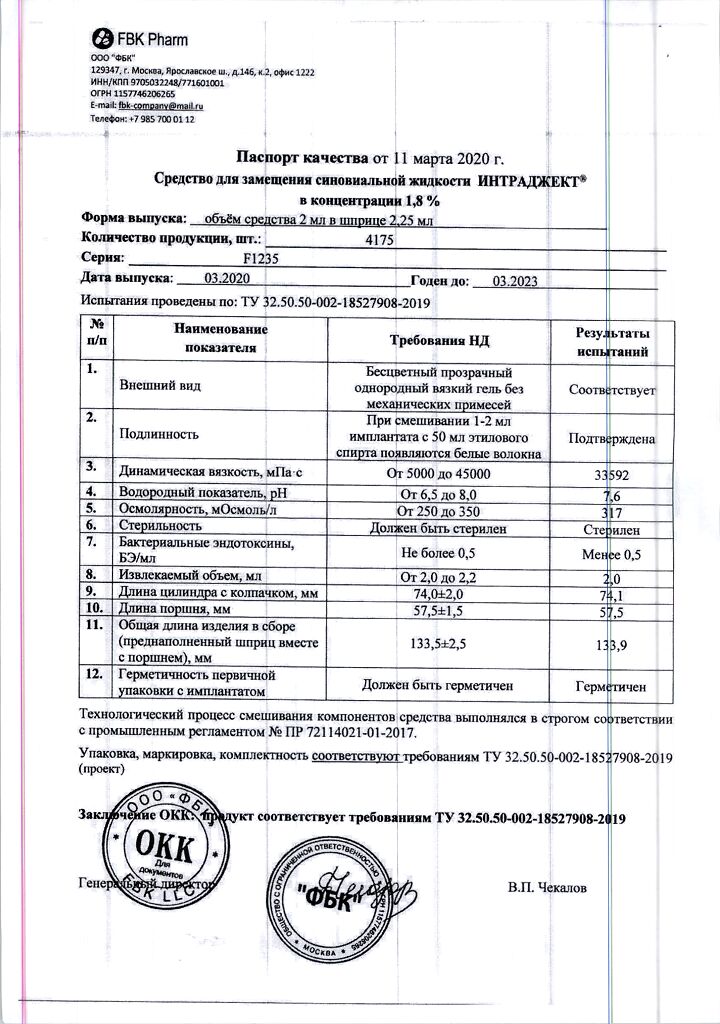
Composition
Composition
1. Sodium hyaluronate
2. Phosphate buffer pH 7.4
Composition of dry matter, mg/ml (%):
– Sodium chloride – 8.1 mg (81.0%) NaCL
– Potassium chloride – 0.2 mg (2.00%) KCl
– Sodium phosphate disubstituted 12 aqueous 1.45 mg (14.50%) Na2HPO4 x 12H2O
– Potassium dihydrogen phosphate – 0.25 mg (2.50%) KH2PO4
3. Water for injections
Pregnancy
Pregnancy
There are no safety data for INTRAJECT® during pregnancy and lactation. Use Tool
INTRAJECT® during pregnancy and lactation – with caution and at the discretion of the attending physician.
Contraindications
Contraindications
1. Hypersensitivity or allergy to the components of INTRAJECT® Products.
2. A history of autoimmune diseases or undergoing autoimmune therapy.
3. Pathological bleeding (endogenous or caused by the use of anticoagulants).
4. Infectious (septic) inflammatory process in the joint or periarticular tissues, intra-articular effusion, general infectious disease.
5. Presence of signs of active skin disease or skin infection in the immediate vicinity of the site
introduction.
6. Pregnancy and lactation.
7. Children under 18 years of age.
Side Effects
Side Effects
Intra-articular administration of INTRAJECT® is well tolerated. In rare cases, local secondary phenomena are possible: pain, sensation of warmth, redness, swelling of the joint, the appearance of intra-articular exudation, and extremely rarely allergic reactions. Side effects may appear
immediately or after some time. These phenomena are short-term and disappear spontaneously. If these symptoms persist for more than one week, the patient should consult a doctor. In exceptional cases, septic arthritis may occur, including that not associated with the use of INTRAJECT®; symptoms of this complication: the appearance of a local inflammatory reaction, increased pain in the joint, increased body temperature.
Interaction
Interaction
Efficiency and safety of use of INTRAJECT® products in combination with simultaneous intra-
joint administration of other medical devices and/or drugs has not been studied, therefore the combined administration of INTRAJECT® with other medical devices and/or drugs is possible only if there is an evidence base for the safety and effectiveness of this method.
Incompatibility has been noted between sodium hyaluronate and quaternary ammonium salts, such as benzalkonium chloride. Therefore, under no circumstances should INTRAJECT® be allowed to come into contact with such drugs or with medical and surgical instruments treated with such antiseptics.
Storage conditions
Storage conditions
Do not use the product with damaged packaging. Do not use after expiration date. Keep out of the reach of children.
Shelf life
Shelf life
3 years from the date of production.
Manufacturer
Manufacturer
FBK LLC, Russia
Additional information
| Shelf life | 3 years from date of manufacture. |
|---|---|
| Conditions of storage | Do not use the product with damaged packaging. Do not use after the expiration date. Keep out of the reach of children. |
| Manufacturer | FBK LLC, Russia |
| Medication form | solution for injection |
| Brand | FBK LLC |
Related products
Buy Intraject 1.8% 2 ml with delivery to USA, UK, Europe and over 120 other countries.