No products in the cart.
Glukonorm plus, 2.5mg+500 mg 30 pcs
€8.35 €7.31
Description
Oral hypoglycemic agent (second generation sulfonylurea derivative + biguanide)
Indications
Indications
Type 2 diabetes mellitus in adults:
– when diet therapy, exercise, and prior monotherapy with metformin or a sulfonylurea derivative are ineffective;
– to replace prior therapy with two drugs (metformin and a sulfonylurea derivative) in patients with stable and well-controlled glycemia levels.
Active ingredient
Active ingredient
Glibenclamide, Metformin
Composition
Composition
Active substances:
Glibenclamide – 2.50 mg,
metformin hydrochloride – 500 mg.
Auxiliary substances:
Microcrystalline cellulose – 61.50 mg,
Hyprolose (hydroxypropyl cellulose) – 12.00 mg,
croscarmellose sodium – 18.00 mg,
magnesium stearate – 6.00 mg.
Shell for dosage of 2.5 mg + 500mg:
. OPADRY® 20A230018 Orange [hypromellose (hydroxypropyl methylcellulose 6 cPs) – 7.92 mg, hyprolose (hydroxypropyl cellulose) – 4.62 mg, talc – 3.78 mg, titanium dioxide – 1.65 mg, sunset yellow dye (E110) – 0.03 mg] – 18.00 mg
How to take, the dosage
How to take, the dosage
The dose of the drug is determined by the doctor individually for each patient depending on the level of glycemia. The initial dose is 1 tablet of Glukonorm® Plus 2.5 mg + 500 mg once a day. To avoid hypoglycemia, the initial dose should not exceed the daily dose of glibenclamide (or the equivalent dose of another previously taken sulfonylurea drug) or metformin, if they were used as first-line therapy. It is recommended that the dose be increased by no more than 5 mg of glibenclamide + 500 mg of metformin per day every 2 or more weeks to achieve adequate blood glucose control.
Replacement of prior combination therapy with metformin and glibenclamide: The starting dose should not exceed the daily dose of glibenclamide (or the equivalent dose of another sulfonylurea drug) and metformin taken previously. Every 2 or more weeks after the start of treatment, the dose of the drug is adjusted according to the level of glycemia.
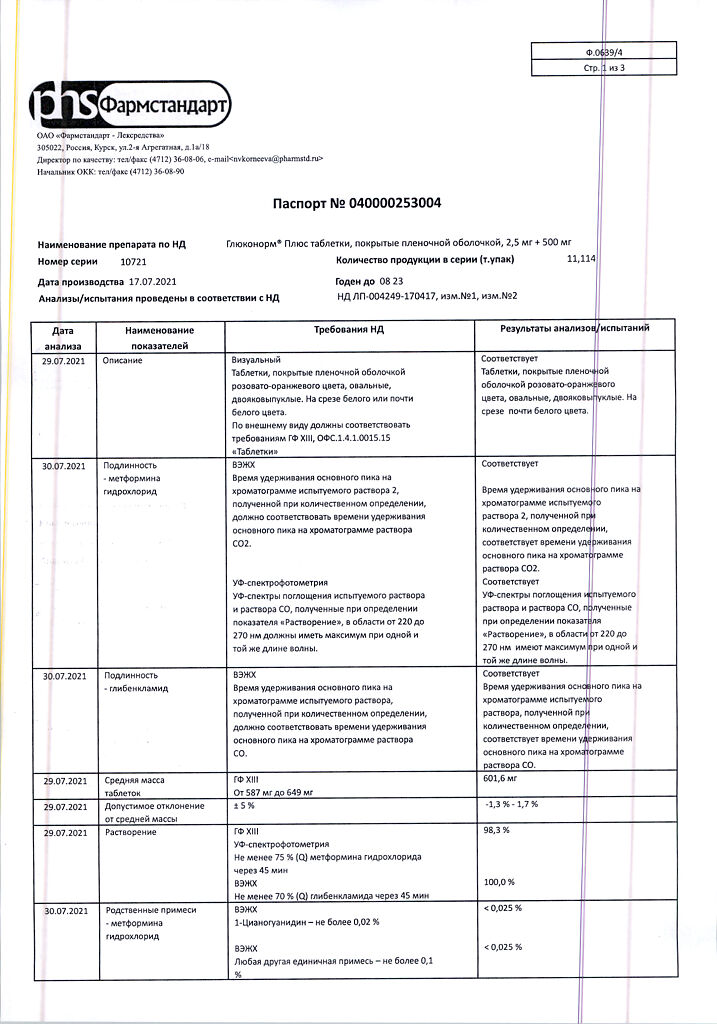
The maximum daily dose is 6 tablets of Gluconorm® Plus 2.5 mg + 500 mg.
The dosing regimen
The dosing regimen depends on the individual prescription:
For doses of 2.5 mg + 500 mg
– once daily, in the morning at breakfast, when prescribed 1 tablet per day;
– twice daily, in the morning and evening, when prescribed 2 or 4 tablets per day.
For doses of 2.5 mg + 500 mg
Three times daily, morning, afternoon, and evening, when prescribing 3, 5, or 6 pills per day.
The tablets should be taken with meals. Each ingestion of the drug should be accompanied by a meal that is high enough in carbohydrates to prevent hypoglycemia.
Elderly patients
The dose of the drug is adjusted according to the state of renal function. The initial dose should not exceed 1 tablet of Gluconorm® Plus 2.5 mg + 500 mg. Regular evaluation of renal function should be carried out.
Children
The drug Glukonorm® Plus is not recommended for use in children.
Special Instructions
Gluconorm® Plus treatment should be regularly monitored on an empty stomach and after meals. Impact on ability to drive vehicles and machinery:
Special Instructions
Lactoacidosis
Lactoacidosis is an extremely rare but serious (high mortality in the absence of emergency treatment) complication that can occur due to metformin cumulation. Cases of lactoacidosis in patients treated with metformin occurred mainly in patients with diabetes mellitus with severe renal failure. Other associated risk factors, such as poorly controlled diabetes, ketosis, prolonged fasting, excessive alcohol consumption, liver failure, and any condition associated with severe hypoxia should be considered. The risk of lactoacidosis should be considered if nonspecific signs occur, such as muscle cramps accompanied by dyspeptic disorders, abdominal pain, and severe malaise. In severe cases, acidotic dyspnea, hypoxia, hypothermia, and coma may be noted. Diagnostic laboratory indicators are: low blood pH, plasma lactate concentration above 5 mmol/L, elevated anion interval and lactate/pyruvate ratio.
Hypoglycemia
Since Glukonorm® Plus contains glibenclamide, the drug administration is accompanied by the risk of hypoglycemia in the patient. Gradual titration of the dose after the start of treatment can prevent the occurrence of hypoglycemia. This treatment can be administered only to a patient who adheres to a regular meal regimen (including breakfast). It is important that carbohydrate intake be regular, since the risk of hypoglycemia increases with late meals, insufficient or unbalanced carbohydrate intake. The development of hypoglycemia is most likely in case of a hypocaloric diet, after intense or prolonged physical activity, when drinking alcohol or when taking a combination of hypoglycemic agents.
Due to compensatory reactions caused by hypoglycemia, sweating, anxiety, tachycardia, hypertension, palpitations, angina pectoris and arrhythmia may occur. The latter symptoms may be absent if hypoglycemia develops slowly, in cases of autonomic neuropathy or concomitant administration of β-adrenoblockers, clonidine, reserpine, guanethidine or sympathomimetics.
Other symptoms of hypoglycemia in diabetic patients may include headache, hunger, nausea, vomiting, marked fatigue, sleep disturbance, agitation, aggression, impaired concentration and psychomotor reactions, depression, confusion, speech disturbance, visual disturbance, tremor, paralysis and paresthesia, dizziness, delirium, seizures, somnolence, unconsciousness, shallow breathing and bradycardia.
Careful prescribing of the drug, dosage selection, and proper patient instructions are important to reduce the risk of hypoglycemia. If the patient has repeated episodes of hypoglycemia that are either severe or related to ignorance of symptoms, treatment with other hypoglycemic agents should be considered.
Factors contributing to hypoglycemia:
– concomitant alcohol consumption, especially when fasting;
– refusal or inability of the patient to interact with the physician and follow the recommendations set forth in the instructions for use (especially in elderly patients);
– Poor nutrition, irregular meal intake, starvation or change in diet;
– imbalance between physical activity and carbohydrate intake;
– renal insufficiency;
– severe hepatic insufficiency;
– overdose of Gluconorm® Plus;
– certain endocrine disorders: insufficiency of thyroid, pituitary gland and adrenal gland function;
– simultaneous reception of certain medicines.
Renal and hepatic insufficiency
Pharmacokinetics and/or pharmacodynamics may vary in patients with hepatic insufficiency or severe renal failure.
Hypoglycemia occurring in such patients may be prolonged, in which case appropriate treatment should be initiated.
Unstable blood glucose
In the case of surgery or other cause of decompensation of diabetes, it is recommended to consider a temporary transition to insulin therapy. Symptoms of hypoglycemia are frequent urination, pronounced thirst, dry skin.
Glukonorm® Plus should be discontinued 48 hours before elective surgery or intravenous administration of iodine containing radiopaque contrast media. It is recommended to restart the treatment in 48 hours and only after renal function has been evaluated and found to be normal.
Renal function
Because metformin is excreted by the kidneys, creatinine clearance and/or serum creatinine content should be determined before starting treatment, and regularly thereafter: at least once a year in patients with normal renal function, and 2-4 times a year in older patients, and in patients with creatinine clearance at the upper limit of normal.
Particular caution is recommended in cases where renal function may be impaired, such as in elderly patients, or if antihypertensive therapy, diuretics or non-steroidal anti-inflammatory drugs (NSAIDs) are started.
Other precautions
The patient should inform the physician of any bronchopulmonary infection or urinary tract infection.
Patients should be informed of the risk of hypoglycemia and should observe precautions when driving vehicles and operating machinery that require increased concentration and rapid psychomotor reactions.
.
Contraindications Cautions:
Contraindications
– Hypersensitivity to metformin, glibenclamide or other sulfonylurea derivatives or excipients;
– type 1 diabetes mellitus;
– diabetic ketoacidosis, diabetic precoma, diabetic coma;
– renal insufficiency or impaired renal function (creatinine clearance less than 60 ml/min);
– acute conditions that may lead to changes in renal function: dehydration, severe infection, shock, intravascular administration of iodine-containing contrast agents (see “
– acute or chronic diseases that are accompanied by tissue hypoxia: cardiac or respiratory failure, recent myocardial infarction, shock;
– hepatic failure;
– porphyria;
– pregnancy, period of breastfeeding;
– concomitant use of miconazole;
– major surgical operations;
– chronic alcoholism, acute alcohol intoxication;
– lactoacidosis (including a history of
– adherence to a hypocaloric diet (less than 1000 calories per day).
The drug is not recommended for persons older than 60 years of age who perform heavy physical work due to the increased risk of lactoacidosis in them.
Fever syndrome, adrenal insufficiency, hypothyroidism, thyroid disease with uncompensated thyroid dysfunction.
.
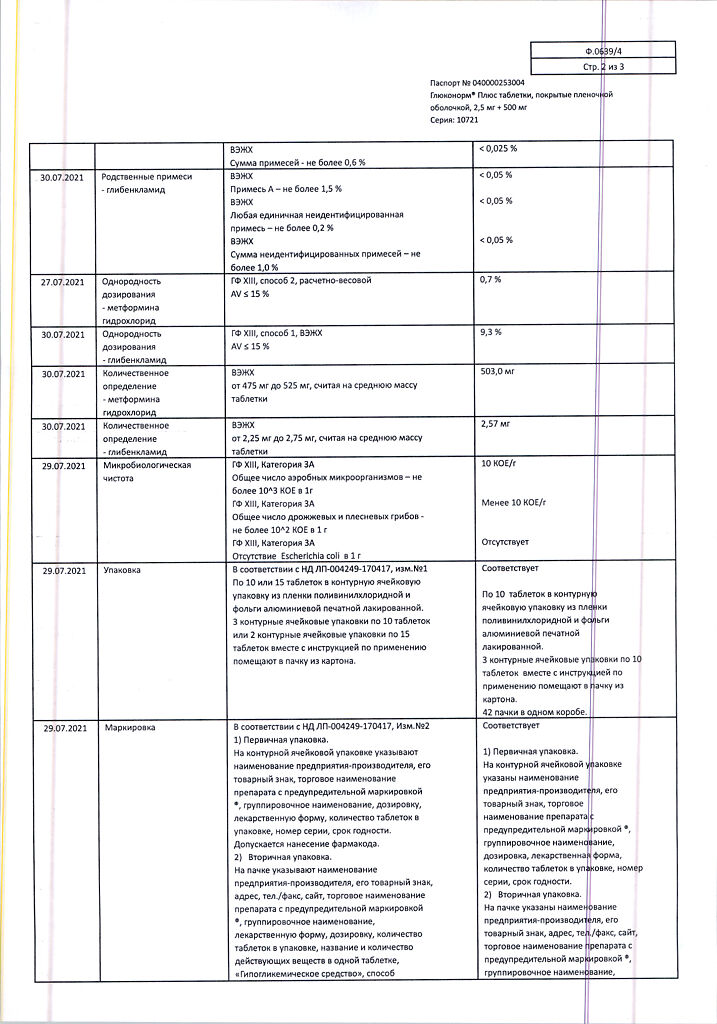
Side effects
Side effects
The following side effects may occur during treatment with Gluconorm® Plus.
The side effects are presented according to the effect on organs and organ systems.
The following adverse events noted with Glukonorm® Plus are distributed by frequency of occurrence according to the following gradation: Very common (≥ 1/10), common (≥ 1/100 to 1/10), infrequent (≥ 1/1000 to < 1/100), rare (≥ 1/10000 to < 1/1000), very rare (< 1/10000).
In each group, adverse effects are presented in decreasing order of severity.
In metabolic and nutritional aspects:
Hypoglycemia (see “Overdose,” “Special Precautions”).
Rarely: episodes of hepatic porphyria and cutaneous porphyria.
Very rare: lactoacidosis (see “Special Precautions”).
A decrease in absorption of vitamin B12, accompanied by a decrease in its concentration in the blood serum with long-term use of metformin. If megaloblastic anemia is detected, the possibility of this etiology must be considered. Disulfiram-like reaction with alcohol use.
Laboratory findings:
Infrequent: moderate to moderate increases in serum urea and creatinine concentrations.
Very rare: hyponatremia.
The blood and lymphatic system:
Rarely: leukopenia, thrombocytopenia.
very rarely: agranulocytosis, hemolytic anemia, bone marrow aplasia and pancytopenia.
These adverse events disappear after discontinuation of the drug.
Nervous system disorders:
Often: taste disorder (metallic taste in the mouth).
Visually: at the beginning of treatment there may be temporary visual impairment due to decreased blood glucose levels.
Gastrointestinal tract:
Very common: nausea, vomiting, diarrhea, abdominal pain and lack of appetite. These symptoms are more common at the beginning of treatment and in most cases go away on their own. To prevent these symptoms, it is recommended that the drug be taken in 2 or 3 doses; increasing the dose slowly also improves tolerability.
Some skin reactions, such as itching, urticaria, maculopapular rash.
Very rare: cutaneous or visceral allergic vasculitis, erythema polymorphicum, exfoliative dermatitis, photosensitization.
The immune system:
Very rare: anaphylactic shock.
Cross-sensitivity reactions to sulfonamides and their derivatives may occur.
Hepatic and biliary tract disorders:
Very rare: impaired liver function or hepatitis requiring discontinuation of treatment.
Overdose
Overdose
In case of overdose, hypoglycemia may occur due to the presence of sulfonylurea derivative in the drug (see “Cautions”).
Pregnancy use
Pregnancy use
Use of the drug is contraindicated during pregnancy.
Similarities
Similarities
Glybomet, Glucovance, Gluconorm, Metglyb Force
Additional information
| Weight | 0.035 kg |
|---|---|
| Shelf life | 2 years. Do not use after the expiration date stated on the package. |
| Conditions of storage | At a temperature not exceeding 25 ° C. Store out of the reach of children. |
| Manufacturer | Pharmstandard-Leksredstva, Russia |
| Medication form | pills |
| Brand | Pharmstandard-Leksredstva |
Related products
Buy Glukonorm plus, 2.5mg+500 mg 30 pcs with delivery to USA, UK, Europe and over 120 other countries.