No products in the cart.
Glauprost, eye drops 0.005% 2.5ml 3 pcs.
€58.40 €48.67
Description
Antiglaucoma drug.
The prostaglandin F2α analogue and a selective FP-receptor agonist.
Limits intraocular pressure by increasing outflow of aqueous humor, mainly through uveoscleral route and also through trabecular meshwork. It has no significant effect on aqueous humor production and does not affect the blood-ophthalmic barrier.
The decrease of intraocular pressure starts 3-4 hours after administration of the drug, the maximum effect is noted after 8-12 hours, the action lasts for at least 24 hours.
Indications
Indications
Open angle glaucoma.
Increased intraocular pressure.
Pharmacological effect
Pharmacological effect
Antiglaucoma drug.
Prostaglandin F2α analogue and selective FP receptor agonist.
Reduces intraocular pressure by increasing the outflow of aqueous humor, mainly along the uveoscleral pathway, as well as through the trabecular meshwork. It does not have a significant effect on the production of aqueous humor and does not affect the blood-ophthalmic barrier.
The decrease in intraocular pressure begins 3-4 hours after administration of the drug, the maximum effect is observed after 8-12 hours, the effect continues for at least 24 hours.
Special instructions
Special instructions
Causes a gradual change in eye color by increasing the amount of brown pigment in the iris. This effect is detected predominantly in patients with mixed iris color, for example, blue-brown, gray-brown, green-brown or yellow-brown, which is explained by an increase in the melanin content in the stromal melanocytes of the iris. Typically, brown pigmentation extends concentrically around the pupil to the periphery of the iris, and the entire iris or parts of it may become a more intense brown color. In case of intense changes in eye pigmentation, therapy is stopped.
In patients with uniformly colored eyes of blue, grey, green or brown, changes in eye color are very rare after 2 years of use of the drug. Once the color change develops, it may be irreversible. Before starting treatment, patients should be informed about the possibility of changes in eye color.
The drug contains benzalkonium chloride, which can be absorbed by contact lenses. Contact lenses should be removed before instilling eye drops; You can insert lenses only 15 minutes after instillation.
Latanoprost may cause gradual changes in eyelashes and vellus hairs, such as lengthening, thickening, increased pigmentation, increased thickness, and a change in the direction of eyelash growth. Changes in eyelashes are reversible and disappear after cessation of treatment.
Patients using drops in only one eye may develop heterochromia.
Impact on the ability to drive vehicles and operate machinery
Patients who experience transient blurred vision after using eye drops are not recommended to drive vehicles or operate machinery until vision is restored.
Active ingredient
Active ingredient
Latanoprost
Composition
Composition
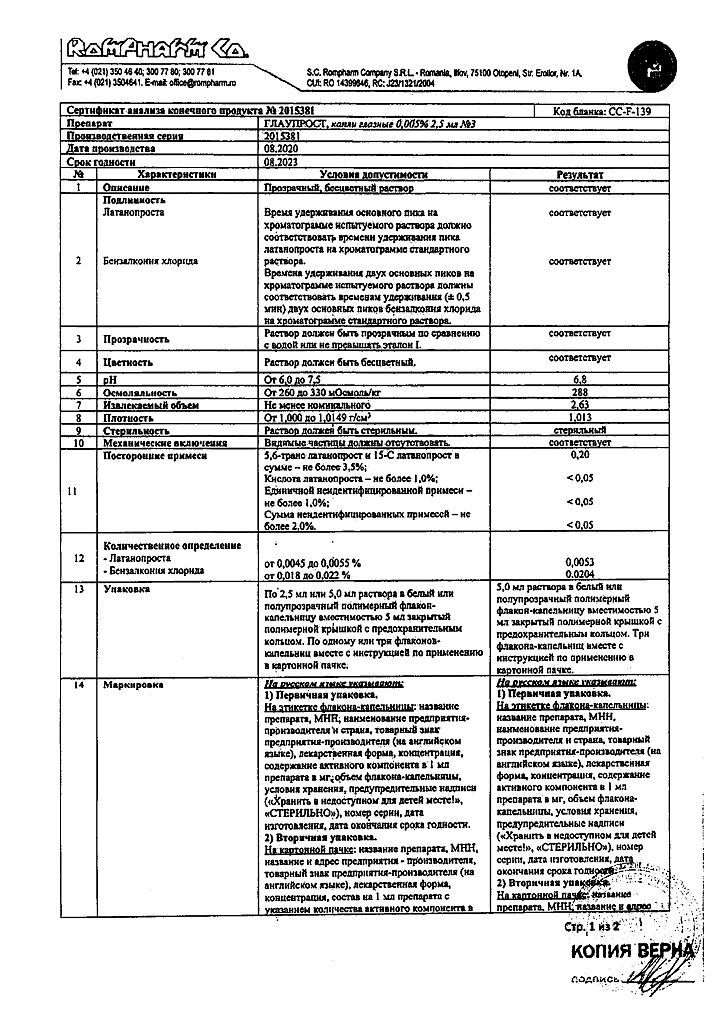
1 ml – latanoprost 50 mcg
Excipients:
disodium hydrogen phosphate dodecahydrate,
sodium dihydrogen phosphate dihydrate,
sodium chloride,
benzalkonium chloride,
purified water.
Pregnancy
Pregnancy
Glauprost is contraindicated in children (under 18 years of age).
The use of Glauprost during pregnancy is possible only under the supervision of a doctor and only if the expected benefit to the mother outweighs the risk of developing possible side effects in the fetus.
Latanoprost and its metabolites may be excreted in breast milk.
If it is necessary to prescribe Glauprost during lactation, breastfeeding should be stopped.
Contraindications
Contraindications
Individual hypersensitivity to latanoprost, benzalkonium chloride or other components of the drug.
Age up to 18 years.
With caution:
Aphakia.
Pseudophakia.
Rupture of the posterior lens capsule.
In patients with known risk factors for macular edema (cases of the development of macular edema, including cystoid, have been described during treatment with latanoprost).
Inflammatory, neovascular or congenital glaucoma (due to lack of sufficient experience with the drug).
Side Effects
Side Effects
From the side of the organ of vision
Eye irritation (burning sensation, feeling of sand in the eyes, itching, tingling and foreign body sensation), blepharitis, conjunctival hyperemia, eye pain, increased pigmentation of the iris, transient pinpoint erosion of the epithelium, swelling of the eyelids, swelling and erosion of the cornea, conjunctivitis; lengthening, thickening, increase in the number and intensification of pigmentation of eyelashes and vellus hair, change in the direction of eyelash growth, sometimes causing irritation of the eye, iritis/uveitis, keratitis, macular edema (including cystoid), blurred vision.
Dermatological reactions
Rash, darkening of the skin of the eyelids and local skin reactions of the eyelids.
From the nervous system
Dizziness, headache.
From the respiratory system
Bronchial asthma (including acute attacks or exacerbation of the disease in patients with a history of bronchial asthma), shortness of breath.
From the musculoskeletal system
Muscle pain, joint pain.
Others
Nonspecific chest pain.
Interaction
Interaction
Pharmaceutically incompatible with eye drops containing thiomersal – precipitation.
When prescribing combination therapy, eye drops of various drugs should be administered at intervals of at least 5 minutes.
Simultaneous instillation of two prostaglandin analogues can cause a paradoxical increase in intraocular pressure.
Overdose
Overdose
Symptoms: irritation of the mucous membrane of the eye, hyperemia of the conjunctiva or episclera.
Treatment: symptomatic.
Storage conditions
Storage conditions
Store in a place protected from light at a temperature of 2° to 8°C.
Shelf life
Shelf life
3 years. After opening the bottle – 4 weeks.
Manufacturer
Manufacturer
K.O.Rompharm Company S.R.L., Romania
Additional information
| Shelf life | 3 years. After opening the bottle – 4 weeks. |
|---|---|
| Conditions of storage | Store in a light-protected place at 2° to 8°C. |
| Manufacturer | C.O.Rompharm Company S.R.L., Romania |
| Medication form | eye drops |
| Brand | C.O.Rompharm Company S.R.L. |
Other forms…
Related products
Buy Glauprost, eye drops 0.005% 2.5ml 3 pcs. with delivery to USA, UK, Europe and over 120 other countries.