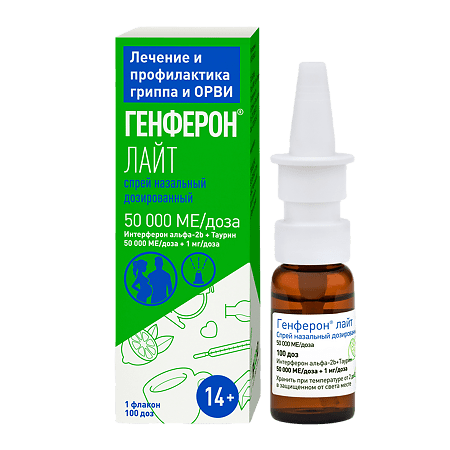
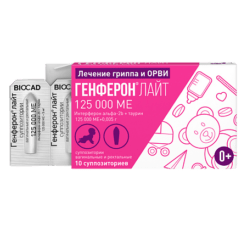
-
×


-
×


-
×


-
×


-
×

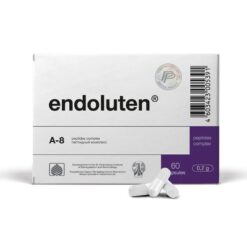
-
×


-
×

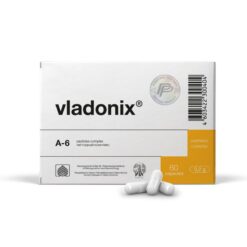
Subtotal: €1179.48