No products in the cart.
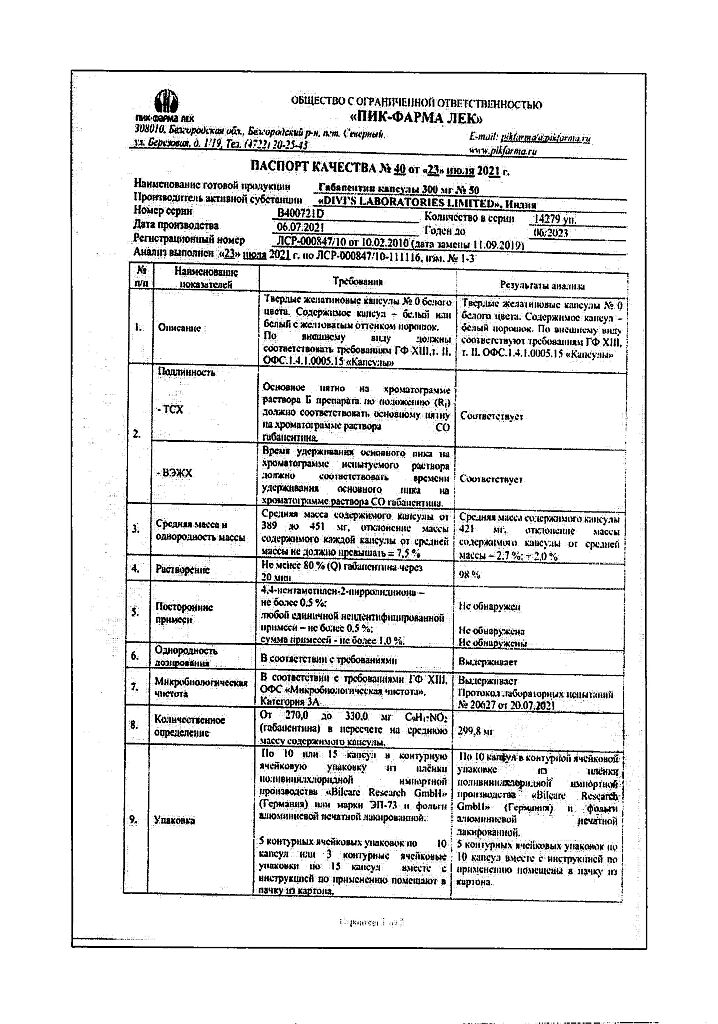
Gabapentin, capsules 300 mg 50 pcs
€14.04 €11.70
Out of stock
(E-mail when Stock is available)
Description
Gabapentin is similar in structure to the neurotransmitter gamma-aminobutyric acid (GABA), but its mechanism of action differs from other drugs that interact with GABA receptors (valproic acid, barbiturates, benzodiazepines, GABA transaminase inhibitors, GABA capture inhibitors, GABA agonists and prodrug forms of GABA).
It has no GABAergic properties and has no effect on GABA capture or metabolism. Preliminary studies have shown that gabapentin binds to the α2-δ-subunit of voltage-dependent calcium channels and reduces calcium ion flux, which plays an important role in the onset of neuropathic pain.
Other mechanisms of action of Gabapentin in neuropathic pain are reduction of glutamate-dependent neuronal death, increase of GABA synthesis, suppression, release of monoamine group neurotransmitters. Gabapentin in clinically relevant, concentrations does not bind to receptors of other common drugs or neurotransmitters, including GABA, GABB, benzodiazepine, glutamate, glycine or N-methyl-D-aspartate receptors.
Gabapentin, unlike phenytoin and carbamazepine, does not interact with sodium channels in vitro. Gabapentin partially attenuated the effects of the glutamate receptor agonist N-methyl-D-aspartate in some in vitro tests, but only at concentrations greater than 100 μmol, which are not achieved in vivo. Gabapentin slightly reduces the release of monoamine neurotransmitters in vitro.
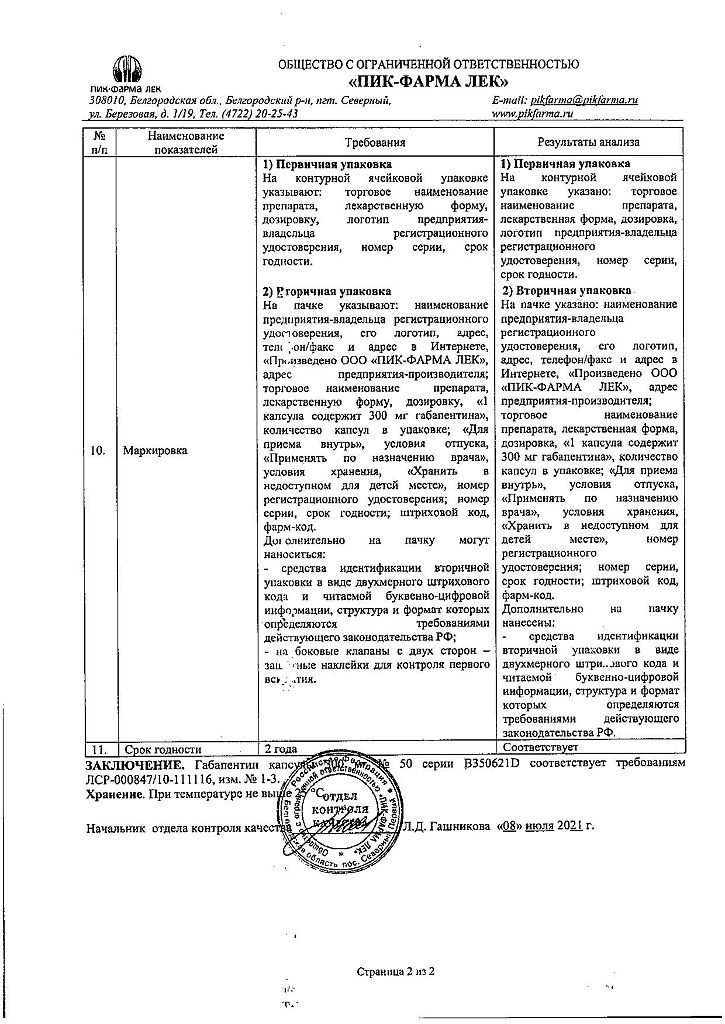
Pharmacokinetics
The bioavailability of Gabapentin is not proportional to the dose. Thus, it decreases with increasing dose. After oral administration Cmax of Gabapentin in plasma is reached after 2-3 hours. The absolute bioavailability of Gabapentin in capsules is about 60%. Food, including high fat content, has no effect on pharmacokinetics.
The plasma excretion of Gabapentin is best described using a linear model. T1/2 from plasma is independent of dose and averages 5-7 h. Pharmacokinetics do not change with repeated administration; equilibrium plasma concentrations can be predicted from a single drug administration.
Gabapentin is almost not bound to plasma proteins (d 57.7 l. It is excreted unchanged by the kidneys only, it is not metabolized.
The drug does not induce hepatic oxidative enzymes with mixed function involved in drug metabolism. Clearance of Gabapentin from plasma is decreased in elderly people and patients with impaired renal function. The elimination rate constant, plasma clearance and renal clearance are directly proportional to creatinine clearance.
Gabapentin is eliminated from plasma by hemodialysis. Dose adjustment is recommended in patients with impaired renal function and patients receiving hemodialysis treatment.
Indications
Indications
Epilepsy, Seizures
- monotherapy or as adjunctive treatment for partial seizures with or without secondary generalization in adults and children from 12 years old;
- neuropathic pain in adults (18 years and older).
Active ingredient
Active ingredient
Gabapentin
Composition
Composition
Active ingredient:
Gabapentin – 300 mg;
Auxiliary substances:
calcium hydrophosphate dihydrate (Emcompress),
potato starch,
Macrogoal (polyethylene glycol 6000),
Magnesium stearate
How to take, the dosage
How to take, the dosage
Over the body, swallowed whole, regardless of meals, with plenty of fluids. If it is necessary to reduce the dose, stop the medicine, or replace it with an alternative medicine, this should be done gradually over at least one week.
Neuropathic pain in adults
The initial daily dose is 900 mg divided into three doses; if necessary, the dose is gradually increased to a maximum of 3600 mg/day. Treatment can begin immediately with a dose of 900 mg/day (300 mg three times a day) or for the first 3 days the dose can be gradually increased to 900 mg/day according to the following schedule:
Day 1: 300 mg once daily;
Day 2: 300 mg 2 times daily;
Day 3: 300 mg 3 times daily.
Partial seizures
Adults and children over 12 years of age: effective dose is 900 to 2400 mg/day. Therapy can be started with a dose of 300 mg 3 times daily on the first day or increased gradually to 900 mg according to the scheme described above.
Thereafter, the dose may be increased to a maximum of 3,600 mg/day (divided into 3 equal doses). The maximum interval between doses in triplicate doses should not exceed 12 hours to avoid recurrence of seizures.
Matching doses in renal failure
Patients with renal impairment are recommended to reduce the dose of Gabapentin according to the table:
Creatinine clearanceDaily dose (mg/day)>80900-240050-79600-1200-1230-49300-60015-29150*-300150*
* administer 300 mg every other day
Recommendations for patients on hemodialysis
Patients on hemodialysis who have not previously taken Gabapentin are advised to prescribe a saturation dose of 300-400 mg and then use 200-300 mg every 4 hours of hemodialysis.
Interaction
Interaction
Morphine: when co-administration of Gabapentin and morphine, when morphine was taken 2 h before Gabapentin administration, a 44% increase in mean area under the pharmacokinetic AUC of Gabapentin was observed compared to Gabapentin monotherapy, which was associated with an increase in pain threshold (cold pressor test).
The clinical significance of this change has not been established, and the pharmacokinetic characteristics of morphine were not altered. Side effects of morphine co-administration with Gabapentin did not differ from those of morphine co-administration with placebo.
There were no interactions between Gabapentin and phenobarbital, phenytoin, valproic acid and carbamazepine. Pharmacokinetics of Gabapentin in equilibrium is similar in healthy subjects and patients receiving other anticonvulsants.
The concomitant use of Gabapentin with oral contraceptives containing norethisterone and/or ethinylestradiol was not accompanied by changes in pharmacokinetics of both components.
The concomitant use of Gabapentin with antacids containing aluminum and magnesium is accompanied by a decrease in the bioavailability of Gabapentin by approximately 20%. It is recommended to take Gabapentin about 2 hours after taking an antacid.
Probenecid has no effect on the renal excretion of Gabapentin.
A slight decrease in renal excretion of Gabapentin when concomitantly taking cimetidine is probably not clinically relevant.
Special Instructions
Special Instructions
While withdrawal syndrome with seizures has not been noted with treatment with Gabapentin, however, abrupt discontinuation of therapy with antiepileptic drugs in patients with partial seizures may provoke the development of seizures.
Gabapentin is not considered an effective treatment for absences epilepsy.
In patients who require concomitant therapy with morphine, it may be necessary to increase the dose of Gabapentin. However, patients should be closely monitored for signs of central nervous system (CNS) depression such as drowsiness. In this case, the dose of Gabapentin or morphine should be adequately reduced.
Laboratory studies
When Gabapentin is added to other anticonvulsants, false positive results have been reported in the determination of protein in urine using Ames N-Multistix SG® test strips. The more specific sulfosalicylic acid precipitation method is recommended for the determination of protein in the urine. Patients should avoid driving or performing work that requires quick psychomotor reactions.
.
Contraindications
Contraindications
- high sensitivity to any of the ingredients of the drug,
- age under 12 years in partial convulsions.
With caution: renal failure.
Preventing renal impairment
Take with caution in renal insufficiency.
In patients with impaired renal function and patients receiving hemodialysis treatment, dose adjustment is recommended.
.
Side effects
Side effects
In the treatment of neuropathic pain
The body in general: accidental injury, asthenia, back pain, flu-like syndrome, headache, infection, pain of various localization, peripheral edema, weight gain;
The digestive tract: constipation, diarrhea, dry mouth, dyspepsia, flatulence, nausea, vomiting, abdominal pain;
Nervous system: gait disturbance, amnesia, ataxia, confusion, dizziness, hypoesthesia, somnolence, impaired thinking, tremor;
Respiratory system: shortness of breath, pharyngitis;
Skin and subcutaneous tissue: skin rash;
Sensory organs: amblyopia.
In treatment of partial seizures
The body as a whole: back pain, fatigue, fever, headache, viral infection, peripheral edema, weight gain, asthenia,-general malaise, facial edema;
The cardiovascular system: symptoms of vasodilation or hypertension;
Digestive tract: constipation, dental disease, diarrhea, dyspepsia, increased appetite, dry mouth or throat, nausea and/or vomiting, abdominal pain, flatulence, anorexia, gingivitis;
The blood, lymphatic system: Leukopenia, purpura (most commonly described as bruising from physical trauma);
Bone and muscular system: fractures, myalgias, arthralgias;
Nervous system: amnesia, ataxia, confusion, impaired coordination, depression, dysarthria, emotional lability, insomnia, nervousness, nystagmus, somnolence, thought disorder, tremor, muscle twitching, vertigo, hyperkinesis; increased, weakened or absent reflexes, paresthesias, anxiety, hostility;
The respiratory system: cough, pharyngitis, rhinitis, pneumonia;
Skin and subcutaneous tissues: abrasions, windows, itchy skin, skin rash;
Sensory organs: amblyopia, diplopia, visual disturbances;
The urinary system: urinary tract infection, impotence.
.
Overdose
Overdose
Symptoms: dizziness, double vision, speech disorders, drowsiness, lethargy, diarrhea.
Treatment: gastric lavage, intake of activated charcoal, symptomatic therapy. Patients with severe renal failure may be indicated for hemodialysis.
.
Pregnancy use
Pregnancy use
There is no data on the use of the drug in pregnant women; therefore, Gabapentin should be used during pregnancy only if the estimated benefit to the mother justifies the possible risk to the fetus.
Gabapentin is excreted with the breast milk, its effect on the breastfed child is unknown, therefore breastfeeding should be avoided during treatment.
Pediatric use
It is contraindicated under 12 years of age for partial seizures. Do not prescribe for the treatment of neuropathic pain in children and adolescents under 18 years of age.
.
Similarities
Similarities
Gabagamma, Catena, Neurontin, Tebantin, Convalis, Gabapentin
Additional information
| Weight | 0.049 kg |
|---|---|
| Shelf life | 2 years. Do not use after the expiration date. |
| Conditions of storage | Store the drug in a dry place protected from light at a temperature not exceeding 25 ° C. |
| Manufacturer | Pik-Pharma, Russia |
| Medication form | capsules |
| Brand | Pik-Pharma |
Other forms…
Related products
Buy Gabapentin, capsules 300 mg 50 pcs with delivery to USA, UK, Europe and over 120 other countries.