No products in the cart.
Forlax, 4 g 20 pcs.
€7.71 €6.75
Out of stock
(E-mail when Stock is available)
Description
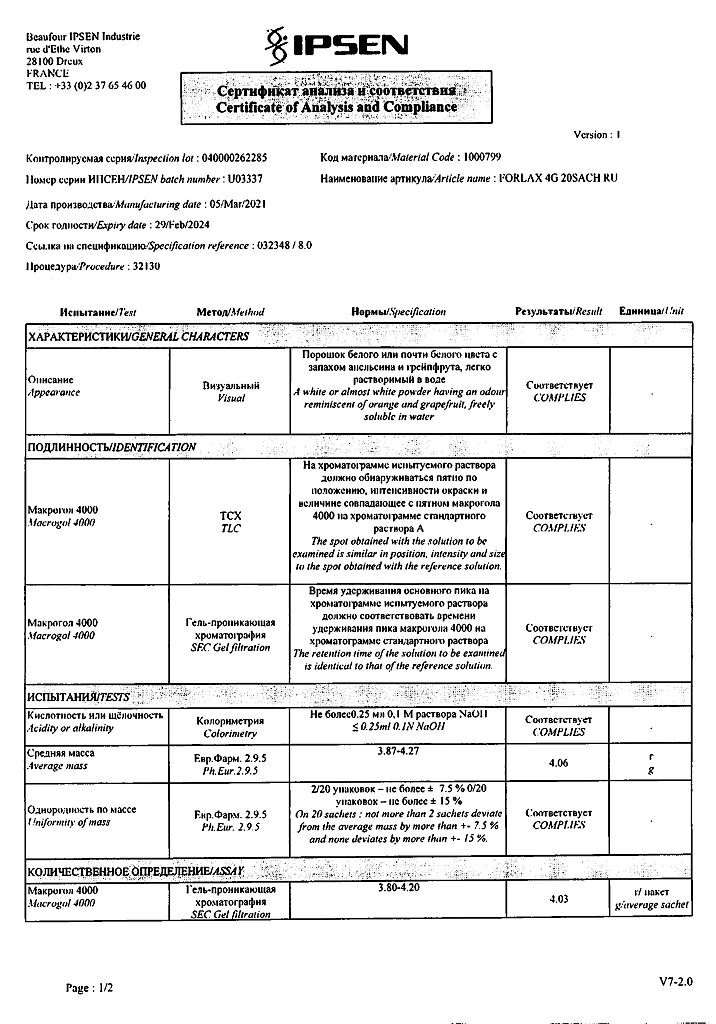
Forlax is a laxative.
Pharmacodynamics
The high molecular weight compound macrogoal 4000 is a linear polymer that retains water molecules through hydrogen bonds. This increases the volume of intestinal contents after oral administration.
The volume of unabsorbed fluid in the intestinal lumen supports the laxative effect of the solution.
Pharmacokinetics
Pharmacokinetic data confirm that macrogoal 4000 undergoes neither gastrointestinal resorption nor biotransformation when taken orally.
Indications
Indications
Constipation, Preparation for Diagnostic ProceduresSymptomatic treatment of constipation in children aged 6 months to 8 years.
Active ingredient
Active ingredient
Macrogol
Composition
Composition
1 bag of powder for preparation of oral solution contains:
active ingredient:
macrogol 4000 – 4 g,
Auxiliary substances:
Orange and Grapefruit Fragrance* – 60.0
Sodium saccharinate (E954) – 6.8
* Orange oil, grapefruit oil, concentrated orange juice, citral, acetic aldehyde, linalool, ethyl butyrate, alpha terpineol, octanal, cis-3-hexenol, maltodextrin, acacia gum, sorbitol (E420), sulfur dioxide (E220), butylhydroxyanisole (E320).
How to take, the dosage
How to take, the dosage
Inside.
– from 6 months to 1 year: 1 sachet per day.
– from 1 to 4 years: 1 to 2 sachets per day.
– from 4 to 8 years: 2 to 4 sachets per day.
The daily dose should be adjusted according to the clinical response. The action of Forlax® is observed 24 to 48 hours after intake.
The duration of treatment is not more than 3 months. The effect after the restoration of normal bowel function should be maintained by means of an active lifestyle and a diet rich in vegetable fiber.
The contents of one sachet should be dissolved in about 50 ml of water and taken in the morning (if the dosage is 1 sachet per day) or in the evening (if the dosage is more than 1 sachet per day). The prepared solution should be a clear liquid like water.
Interaction
Interaction
Delays absorption of concomitantly taken drugs. Therefore, it is recommended to prescribe Forlax® at least 2 hours after other drugs.
Special Instructions
Special Instructions
The data on the effectiveness of therapy in children under 2 years of age are limited.
The treatment of constipation with medication is recommended only as an adjunct to a healthy lifestyle and diet, such as:
– increasing fluid and fiber intake,
– adequate physical activity that helps restore the motility of the digestive tract.
Organic gastrointestinal disorders should be ruled out prior to initiation of therapy.
The drug contains macrogol (polyethylene glycol). Cases of hypersensitivity (anaphylactic shock, angioneurotic edema, urticaria, rash, pruritus, erythema) have been reported when taking drugs containing macrogoal (polyethylene glycol).
Because of the presence of sulfur dioxide in rare cases severe hypersensitivity reactions and bronchospasm may occur.
Patients with hereditary fructose intolerance should not take this drug.
Patients with a predisposition to water-electrolyte imbalance (e.g., patients with impaired hepatic or renal function or patients taking diuretics) should be treated with particular caution if diarrhea develops and electrolyte control should be performed.
In cases of aspiration have been reported when large volumes of macrogoal and electrolytes have been administered using a nasogastric tube. Children with neurological disorders who have a swallowing disorder are at risk for aspiration.
Caution when administering.
Forlax® 4 g does not contain significant amounts of sugar or polyols and may be used in children with diabetes or in children whose diet is free of galactose.
Contraindications
Contraindications
- Serious inflammatory bowel disease (ulcerative colitis, Crohn’s disease) or toxic megacolon;
- perforation or risk of perforation of the gastrointestinal tract;
- abdominal pain of unclear etiology;
- hypersensitivity to the active substance or to any other excipients of the drug.
.
Side effects
Side effects
The adverse reactions listed in the table below have been reported in clinical trials involving 147 children aged 6 months to 15 years and during post-registration use.
In general, adverse reactions were mild and transient and mostly involved the gastrointestinal systems.
The frequency of adverse drug reactions was determined according to the following grading: very common (≥1/10); common (≥1/100, < 1/10); infrequent (≥1/1000, < 1/100), rare (≥1/10000, < 1/1000), very rare (< 1/10000), frequency not determined (cannot be estimated from available data).
Gastrointestinal disorders:
Often – Abdominal pain, Diarrhea*
Infrequent – Vomiting , Bloating, Nausea
Immune system disorders
/p>
Frequency not established – Hypersensitivity reactions (anaphylactic shock, angioedema, urticaria, rash, itching).
* Diarrhea may cause perianal irritation.
Overdose
Overdose
Diarrhea may occur, which stops when the dose of the drug is reduced or cancelled.
Intensive diarrhea or vomiting may cause an imbalance of the electrolyte balance, which will require correction.
There have been reports of perianal inflammation and irritation and fecal incontinence when large volumes (4-11 liters) of polyethylene glycol have been used for colon cleansing prior to colonoscopy.
Pregnancy use
Pregnancy use
Pregnancy Experiments in rats and rabbits have shown that Macrogol 4000 has no teratogenic effect.
Pregnant women should exercise caution when prescribing Forlax to pregnant women because of the paucity of data on the use of Forlax in pregnant women.
Lactation It is not known whether the drug passes into the mother’s milk. Because macrogol 4000 is not absorbed, Forlax can be taken during lactation.
Similarities
Similarities
Fortrans, Lavacol
Additional information
| Weight | 0.120 kg |
|---|---|
| Shelf life | 3 years |
| Conditions of storage | At a temperature not exceeding 30 °C |
| Manufacturer | Bofour Ipsen Industries, France |
| Medication form | Powder for preparation of solution for oral administration |
| Brand | Bofour Ipsen Industries |
Other forms…
Related products
Buy Forlax, 4 g 20 pcs. with delivery to USA, UK, Europe and over 120 other countries.