No products in the cart.
Escapel, 1.5 mg tablets
€23.00 €19.17
EAN: 5997001360484
SKU: 207825
Categories: Contraceptive, Gynecology and Obstetrics, Hormonal, Medicine
Description
Pharmacotherapeutic group:Emergency contraceptive.
ATX code: G03AD01
Pharmacodynamics
Levonorgestrel is a synthetic gestagen with contraceptive action, pronounced gestagenic and anti-estrogenic properties. The main mechanism of action is inhibition and/or delay of ovulation as a result of suppression of peak luteinizing hormone. Under the recommended dosing regimen, levonorgestrel suppresses ovulation and fertilization if sexual intercourse occurred in the pre-ovulatory phase, when
the possibility of fertilization is greatest. Levonorgestrel is not effective if implantation of a fertilized egg has already occurred.
Effectiveness: According to a previous clinical study, two doses of levonorgestrel 0.75 mg at 12-hour intervals prevented pregnancy in 85% of cases. Its efficacy decreases with time following intercourse (95% if used within 24 hours, 85% between 24 and 48 hours, and 58% between 48 and 72 hours).
The results of another clinical study showed that a single levonorgestrel dose of 1.5 mg (within 72 hours of unprotected intercourse) prevents pregnancy in 84% of cases.
There is limited data, requiring further confirmation, on the effect of excess body weight/high body mass index (BMI) on contraceptive efficacy. Two clinical trials (CIs) found decreased efficacy of levonorgestrel and increased pregnancy rates in women with BMI ≥30 kg/m2 compared with women with normal BMI (5.19% and 0.96%, respectively). However, no decrease in the contraceptive efficacy of levonorgestrel was confirmed in other studies (pregnancy rates were 1.17% in obese women and
0.99% in women with normal BMI).
With the recommended dosing regimen, levonorgestrel has no significant effect on clotting factors, lipid and carbohydrate metabolism.
Adolescent girls under 18 years
In a prospective observational study, it was shown that of 305 cases of levonorgestrel as an emergency contraceptive, seven resulted in pregnancy. Thus, the overall failure rate was 2.3%.
The failure rate in adolescent girls younger than 18 years (2.6% or 4/153) was comparable to the failure rate in women 18 years and older (2.0% or 3/152).
Pharmacokinetics
Assimilation
Levonorgestrel is rapidly and almost completely absorbed when taken orally. After taking levonorgestrel at a dose of 1.5 mg, the maximum plasma concentration (Cmax) is 18.5 ng/ml and is reached after 2 hours. After reaching the maximum values, the concentration of levonorgestrel decreases. Absolute bioavailability is 100%.
Distribution
Levonorgestrel binds to plasma albumin and sex hormone-binding globulin (hGBS). Only 1.5% of the total dose is in free form, 65% is bound to HSPH. Passes into breast milk.
Metabolism
Metabolism of levonorgestrel corresponds to that of sex hormones. Levonorgestrel is hydrolysed in the liver and the metabolites are excreted as conjugated glucuronides. Pharmacologically active metabolites of levonorgestrel are unknown.
Elimation
Excreted exclusively as metabolites, approximately equally by the kidneys and through the intestine. The elimination half-life (T1/2) is about 26 hours.
Pharmacokinetics in special groups of patients
Children and adolescents under 18 years:The pharmacokinetics of levonorgestrel have been studied exclusively in adult women; there are limited data on the use of levonorgestrel in adolescent girls under 16 years of age.
Patients with renal and hepatic impairment: Pharmacokinetics of levonorgestrel in patients with hepatic or renal impairment have not been studied.
Patients with obesity
. A pharmacokinetics study showed that levonorgestrel concentrations were significantly reduced in obese women (BMI ≥30 kg/m²) (approximately 50% reduction in Cmax and AUC0-24 was observed) compared with those in women with normal BMI (< 25 kg/m²).
Another study also reported a reduction in levonorgestrel Cmax of approximately 50% in obese women compared with that in women with normal BMI, while doubling the levonorgestrel dose to 3 mg in obese women provided plasma concentrations similar to those observed in women with normal BMI who received 1.5 mg of levonorgestrel. The clinical significance of these
data is unclear.
Indications
Indications
Contraception
Emergency (postcoital) contraception (after unprotected intercourse or when the contraceptive method used is unreliable).
Active ingredient
Active ingredient
Levonorgestrel
Composition
Composition
1 tablet contains:
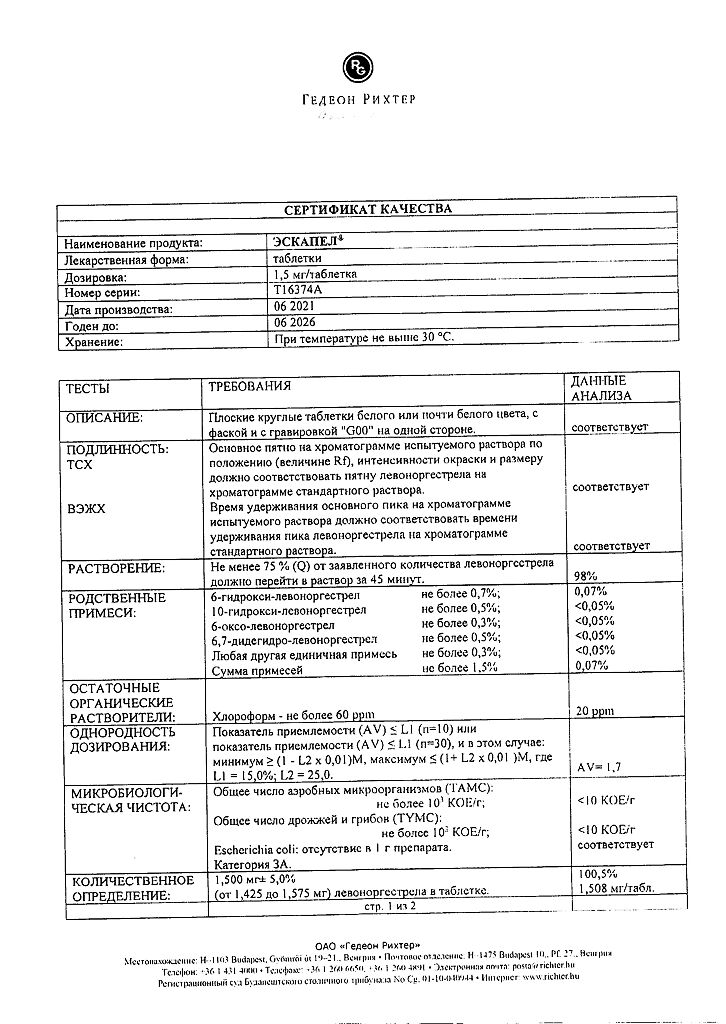
Active substance: levonorgestrel 1.5 mg.
Ancillary substances: colloidal silica; potato starch; magnesium stearate; talc; corn starch; lactose monohydrate.
How to take, the dosage
How to take, the dosage
Ingestion.
The drug can be used at any time of the menstrual cycle. In case of irregular menstrual cycle it is necessary to exclude pregnancy in advance.
To obtain a stronger contraceptive effect, 1 tablet of Escapel® should be taken as soon as possible, preferably within 12 hours (but not later than 72 hours after unprotected intercourse). If within 3 hours after taking the tablet vomiting occurred, you should take 1 more tablet of Escapel®. After taking Escapel® until the next menstruation you should use non-hormonal methods of contraception (condom, spermicide + cervical cap, diaphragm or contraceptive sponge). The use of levonorgestrel is not a contraindication for continuation of planned hormonal contraception. The use of the drug in repeated unprotected intercourse within the same menstrual cycle is not recommended because of the increased risk of the development of cycle disorders.
Particular groups of patients
Children and adolescents under 16 years of age
The use of Escapel® is contraindicated before the age of 16 years (due to limited data on the safety and effectiveness of levonorgestrel in this age group).
Patients with renal impairment
There are no data on the use of levonorgestrel in patients with renal impairment.
Patients with hepatic impairment
There are no data on the use of levonorgestrel in patients with hepatic impairment. Its use in patients with severe hepatic impairment is contraindicated.
Interaction
Interaction
Concomitant use with drugs inducers of microsomal liver enzymes (mainly inducers of CYP3A4 isoenzyme) accelerates the metabolism of levonorgestrel.
The concomitant use of efavirenz decreases plasma concentrations of levonorgestrel by approximately 50%.
The following liver enzyme-inducing drugs may decrease the effectiveness of levonorgestrel: barbiturates (including primidone), phenytoin and carbamazepine, preparations containing Hypericum perforatum, and rifampicin, ritonavir, rifabutin and griseofulvin.
The drugs containing levonorgestrel may increase the risk of cyclosporine toxicity by inhibiting its metabolism.
Levonorgestrel may decrease the efficacy of ulipristal acetate through competitive effects on the progesterone receptor, and therefore concomitant use is not recommended.
Special Instructions
Special Instructions
Emergency contraception is a method that can be used occasionally. It should not replace routine contraception.
Emergency contraception does not prevent pregnancy in all cases.
If there are doubts concerning the time of unprotected sexual intercourse, or if unprotected intercourse took place more than 72 hours during the same menstrual cycle, there is a possibility that conception has already occurred. In this connection the use of Escapel® at the second sexual intercourse can be ineffective with respect to prevention of pregnancy. If menstrual bleeding is delayed for more than 5-7 days and its character has changed (scarce or abundant bloody discharge) it is necessary to exclude pregnancy.
If pregnancy occurs after the use of the drug Escapel® it is necessary to consider the possibility of ectopic pregnancy. The appearance of pain in the lower abdomen, fainting may indicate an ectopic pregnancy. The absolute risk of ectopic pregnancy seems to be low, since levonorgestrel prevents ovulation and fertilization. Ectopic pregnancy may develop despite the occurrence of uterine bleeding. In this regard, the use of the drug Escapel® is not recommended in women with the risk of developing
ectopic pregnancy (salpingitis or ectopic pregnancy in the anamnesis).
The use of the drug Escapel® is not recommended in patients with severe hepatic impairment.
Severe malabsorption syndromes, such as Crohn’s disease, may reduce the effectiveness of the levonorgestrel.
After taking Escapel® the menstrual cycle is usually unchanged and menstrual bleeding occurs on time. Sometimes menstrual bleeding may start a few days earlier or later. Women should be advised to see a doctor to choose and start using one of the methods of planned contraception. If Escapel® was taken against a background of regular hormonal contraception, but
the expected bleeding “cancellation” in the next 7-day period “without the pill” has not occurred, pregnancy should be excluded.
Repeated use of the drug during one menstrual cycle is not recommended due to the possibility of cycle disorders.
There is limited data requiring further confirmation that the contraceptive efficacy of Escapel® may decrease with increasing body weight or BMI. In all women, regardless of body weight and BMI, emergency contraception should be taken as soon as possible after unprotected intercourse.
Escapel® is ineffective as a permanent method of contraception and can only be used as an emergency measure. Levonorgestrel in a dose of 1.5 mg should be used only for emergency contraception!
Women who seek repeated courses of emergency contraception should be recommended to use methods of planned contraception.
Use of the drug in adolescent girls under 16 years is possible only in exceptional cases (including rape) and only after consulting a gynecologist. After emergency contraception, consultation with a gynecologist is recommended.
Fertility
Levonorgestrel increases the risk of menstrual irregularities, which in some cases may lead to earlier or later ovulation. These changes may affect fertility, but there are no data on the effects of levonorgestrel on fertility with long-term use.
The use of emergency contraception does not replace necessary precautions related to protection against sexually transmitted diseases.
Influence on driving and operating machinery
The effect of the drug on driving and operating machinery has not been studied.
Synopsis
Synopsis
Flat round tablets, white or almost white, beveled and engraved “G00” on one side.
Contraindications
Contraindications
– Hypersensitivity to levonorgestrel and/or to any of the excipients in the drug.
– Age under 16 years (due to the limited data on safety and efficacy of levonorgestrel in this age group).
– Severe hepatic failure.
– Known or suspected pregnancy.
– Breast-feeding for at least 8 hours after taking the drug.
– Lactose intolerance, lactase deficiency, glucose-galactose malabsorption syndrome.
With caution
Liver disease (mild to moderate severity) or biliary tract; jaundice (including anamnesis); severe malabsorption syndromes, such as Crohn’s disease; presence of hereditary or acquired predisposition to thrombosis.
Side effects
Side effects
The most common adverse reaction (AR) with levonorgestrel was nausea.
HDs are presented by systemic organ class according to the MedDRA classification and with frequency of occurrence: very common (≥1/10), common (≥1/100, < 1/10), infrequent (≥1/1000, < 1/100), rare (≥1/10000, < 1/1000), very rare (< 1/10000), including individual reports. Within each group, HPs are distributed in decreasing order of importance.
Nervous system disorders
Very common: headache.
Often: dizziness.
Gastrointestinal tract disorders
Very common: nausea, pain in the lower abdomen.
Often: diarrhea, vomiting.
Genital and mammary disorders
Very common: bleeding not associated with menstruation.
Often: menstrual delays of more than 7 days, irregular menstrual bleeding, breast engorgement.
General disorders and disorders at the site of administration
Very common: increased fatigue.
The nature of menstrual bleeding may change slightly, but in most women the next menstruation begins within 5 days of the expected due date.
If your next period is more than 5 days late, pregnancy should be ruled out.
The following adverse events have been observed in clinical practice during the post-registration period:
Gastrointestinal disorders
Very rare: abdominal pain.
Skin and subcutaneous tissue disorders
Very rare: skin rash, urticaria, itching.
Gender and mammary gland disorders
Very rare: pelvic pain, dysmenorrhea.
General disorders and disorders at the site of administration
Very rare: facial swelling.
Overdose
Overdose
No serious adverse reactions have been reported in acute overdose with high doses of oral contraceptives.
Symptoms: nausea and bleeding “withdrawal”.
Treatment: symptomatic. There is no specific antidote.
Pregnancy use
Pregnancy use
Pregnancy
Administration of the drug during known or suspected pregnancy is contraindicated. Based on available data, no adverse effects on the fetus have been identified in the case of continued pregnancy with levonorgestrel-containing emergency contraception.
Breastfeeding
Levonorgestrel penetrates into breast milk. The potential effects of levonorgestrel on the baby can be reduced if the woman takes the drug immediately after breastfeeding. After taking the drug, breastfeeding should be stopped for at least 8 hours.
Similarities
Similarities
Postinor
Additional information
| Weight | 0.010 kg |
|---|---|
| Shelf life | 5 years. Do not use after the expiration date stated on the package. |
| Conditions of storage | At a temperature not exceeding 30 ° C. Keep out of reach of children. |
| Manufacturer | Gedeon Richter, Hungary |
| Medication form | pills |
| Brand | Gedeon Richter |
Related products
Gynecology and Obstetrics
Gynecology and Obstetrics
Prepidil, intracervical gel 0.5 mg/3 g syringes with catheter
Buy Escapel, 1.5 mg tablets with delivery to USA, UK, Europe and over 120 other countries.