No products in the cart.
Encorate chrono, 500 mg 30 pcs
€6.81 €5.96
Description
Sodium valproate increases the content of GABA (gamma-aminobutyric acid) in the brain, which in turn increases the content of GABA in postsynaptic neurons. In addition, sodium valproate affects the transport of potassium ions across neuronal membranes. The result of this effect is the suppression of the emergence as well as the spread of epileptic excitation through neurons.
Valproate has anticonvulsant activity for various degrees of epilepsy in humans. In addition, the drug has no pronounced hypnotic and sedative effects and does not have a depressant effect on the respiratory center. Encorate Chrono has no negative effect on blood pressure, heart rate, renal function and body temperature.
Pharmacokinetics
Absorption of the drug is fast and complete. Cmax in plasma are reached within 2-8 hours after oral administration of controlled-release tablets.
80-90% of the drug is bound by plasma proteins, T1/2 is 8-22 hours. Therapeutic efficacy is seen at plasma concentrations of valproic acid of 40-100 mg/L. At the same time, the pharmacological and therapeutic effects of this controlled-release drug are not always dependent on its plasma concentration. Vd is 0.2 L/kg body weight.
The drug is metabolized in the liver and excreted in the urine. No presystemic metabolism has been noted for this drug.
Indications
Indications
Treatment of generalized or partial epilepsy, especially for the following types of seizures:
absence seizures;
myoclonic;
tonic-clonic;
atonic.
Partial epilepsy:
simple or combined seizures;
secondary generalized seizures.
Specific syndromes (Vest, Lennox-Gastaut)
Pharmacological effect
Pharmacological effect
Sodium valproate increases GABA (gamma-aminobutyric acid) content in the brain, which in turn increases GABA content in postsynaptic neurons. In addition, sodium valproate affects the transport of potassium ions across neuronal membranes. The result of this influence is the suppression of the occurrence and spread of epileptic excitation along neurons.
Valproate has anticonvulsant activity in various degrees of epilepsy in humans. In addition, the drug does not have pronounced hypnotic or sedative effects and does not depress the respiratory center. Encorat Chrono does not have a negative effect on blood pressure, heart rate, kidney function and body temperature.
Pharmacokinetics
Absorption of the drug is fast and complete. Cmax in plasma is achieved within 2-8 hours after oral administration of controlled-release tablets.
80-90% of the drug is bound by blood plasma proteins, T1/2 is 8-22 hours. Therapeutic effectiveness is manifested when the concentration of valproic acid in the blood plasma is 40-100 mg/l. At the same time, the pharmacological and therapeutic effects of this controlled-release drug do not always depend on its plasma concentration. Vd – 0.2 l/kg body weight.
The drug is metabolized in the liver and excreted in the urine. First-pass metabolism was not observed for this drug.
Special instructions
Special instructions
Care should be taken when working with machinery, as the drug impairs attention. It is necessary to regularly monitor the level of transaminases, bilirubin, blood platelets, amylase (every 3 months).
Valproic acid inhibits platelet aggregation, which increases the risk of increased clotting time during bleeding. The possibility of complications associated with bleeding in surgical patients receiving valproate should be taken into account. Patients taking valproate for a long time may develop spontaneous hematomas and bleeding. In this case, you should immediately stop taking the drug.
Valproate may cause drug-induced pancreatitis and liver dysfunction (usually in the first 6 months of use). In this regard, during the first 6 months of treatment it is necessary to monitor the condition of the pancreas, perform liver tests, and monitor the level of prothrombin. Liver dysfunction and liver failure during valproate therapy are sometimes observed in children with epilepsy and concomitant metabolic and degenerative diseases, organic pathology of brain tissue and delayed mental development. If symptoms such as severe weakness, lethargy, swelling, vomiting and jaundice occur, you should immediately stop using the drug; severe weakness, lethargy, swelling, vomiting and jaundice should immediately stop using the drug. Use during pregnancy and lactation:
Prescribing the drug to pregnant women is possible only if the expected benefit to the mother outweighs the potential risk to the fetus. If you take the drug during pregnancy, specialized prenatal monitoring of the fetus is mandatory. The use of valproate during breastfeeding is not recommended.
Active ingredient
Active ingredient
Valproic acid
Composition
Composition
1 tab.
sodium valproate
333 mg
valproic acid
145 mg,
which corresponds to the content of sodium valproate
167 mg
Excipients:
hypromellose-K100M (methocel-K100M),
hypromellose-K15M (methocel-K15M),
starch (dried),
purified talc,
magnesium stearate,
colloidal silicon dioxide.
Shell composition:
copolymer of methacrylate, dimethylaminoethyl methacrylate and butyl methacrylate (eudragit E100), titanium dioxide, polyethylene glycol 6000, sodium lauryl sulfate, sunset yellow dye.
Pregnancy
Pregnancy
Prescribing the drug to pregnant women is possible only when the expected benefit to the mother outweighs the potential risk to the fetus. If you take the drug during pregnancy, specialized prenatal monitoring of the fetus is mandatory.
The use of valproate during breastfeeding is not recommended.
Use in children
Encorat Chrono should not be used in children weighing up to 20 kg.
Contraindications
Contraindications
individual intolerance to the drug;
severe dysfunction of the pancreas and liver;
acute hepatitis;
pregnancy, lactation;
porphyria;
thrombocytopenia;
children up to 3 years old.
Use for liver dysfunction
Contraindicated in cases of severe liver dysfunction and acute hepatitis.
Use in elderly patients
Elderly patients: it is typical that the volume of distribution increases and plasma volume decreases. However, with normal liver and kidney function, this increase in the amount of free drug is not clinically significant. The dosage regimen of Encorat Chrono in elderly patients is similar to the dosage regimen in adults and is selected individually. In patients in whom the necessary therapeutic effect has been achieved during therapy with Encorat tablets, it is possible to use Encorat Chrono in the appropriate daily dosage.
Side Effects
Side Effects
In rare cases, the following side effects have been reported:
nausea, vomiting, diarrhea and/or constipation
hepatitis
pancreatitis
weight gain
skin rash, itching, photosensitivity, erythema multiforme, Steven-Johnson syndrome
hair loss (2-12% of patients)
ataxia, tremor, disturbance of consciousness, coma
menstrual irregularities, secondary amenorrhea
anemia, hyperammonemia, hypercreatinemia, thrombocytopenia, neutropenia, leukopenia, decreased fibrinogen content, inhibition of platelet aggregation.
Interaction
Interaction
Valproate enhances the effect of antipsychotic drugs, anticonvulsants and antidepressants.
Unlike other anticonvulsants, valproate does not induce liver enzymes and therefore does not reduce the effectiveness of oral contraceptives.
When valproate and warfarin are used together, the percentage of plasma protein binding of the latter decreases.
Valproate alters plasma concentrations of phenytoin and lamotrigine.
In combination with anticoagulants and acetylsalicylic acid derivatives, inhibition of platelet aggregation increases.
Overdose
Overdose
Rare cases of accidental or intentional overdose of the drug have been reported. In this case, nausea, vomiting, dizziness, diarrhea, respiratory depression, hyporeflexia, and coma were observed.
First aid: gastric lavage, taking activated carbon. If necessary, inpatient treatment using symptomatic therapy and hemodialysis.
Storage conditions
Storage conditions
In a dry place, protected from light, at a temperature not exceeding 25 °C
Shelf life
Shelf life
3 years
Manufacturer
Manufacturer
Sun Pharmaceutical Industries Ltd, India
Additional information
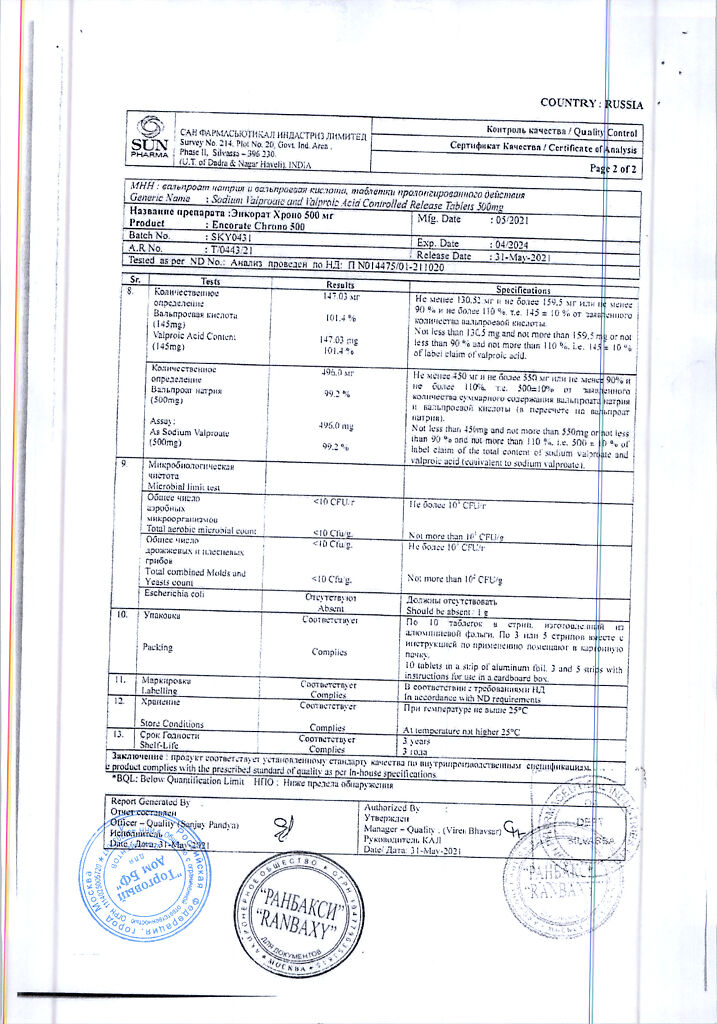
| Shelf life | 3 years |
|---|---|
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25 °C |
| Manufacturer | Sun Pharmaceutical Industries Ltd, India |
| Medication form | sustained release tablets |
| Brand | Sun Pharmaceutical Industries Ltd |
Other forms…
Related products
Buy Encorate chrono, 500 mg 30 pcs with delivery to USA, UK, Europe and over 120 other countries.