No products in the cart.
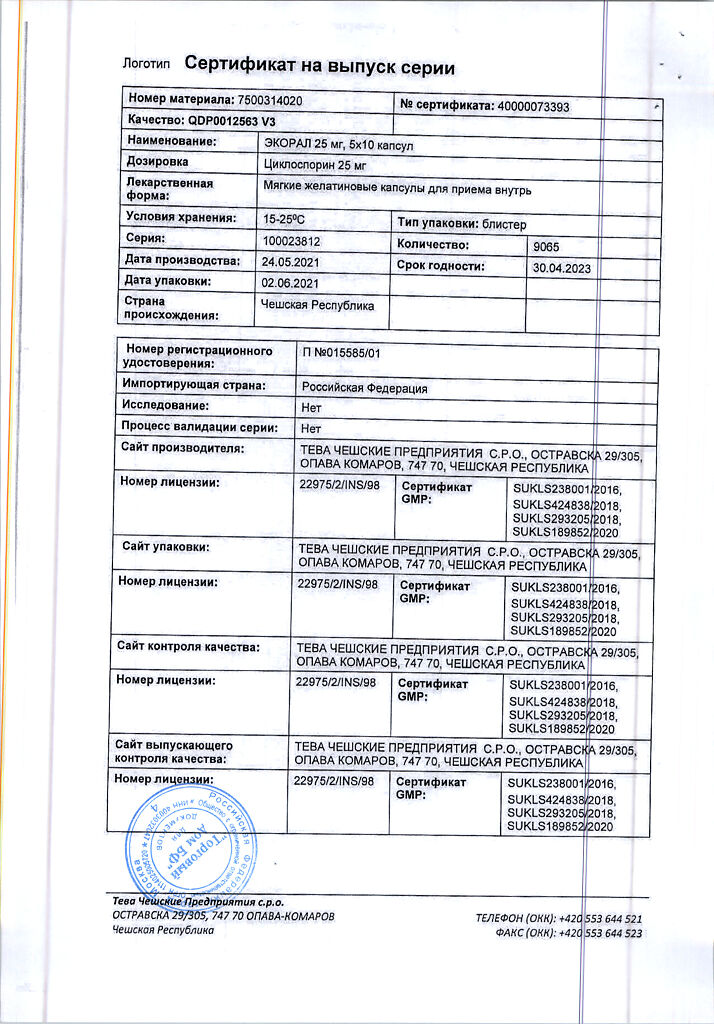
Ecoral, capsules 25 mg 50 pcs
€27.26 €22.71
Out of stock
(E-mail when Stock is available)
Description
Ecoral has an immunosuppressive effect.
At the cellular level it suppresses formation and release of lymphokines, including IL-2 (T-lymphocyte growth factor). It blocks lymphocytes in resting state in G0 or G1 phase of cell cycle and inhibits antigen-dependent release of lymphokines by activated T-lymphocytes. All these data indicate that cyclosporine acts on lymphocytes specifically and reversibly.
It does not inhibit hematopoiesis and does not affect the function of phagocytic cells.
Pharmacokinetics
After oral administration, Cmax in blood is noted between 1 and 6 h, with bioavailability averaging 30% (20-50%) and increasing with increasing dose and duration of treatment. Absorption decreases after liver transplantation, liver disease or gastrointestinal pathology (diarrhea, vomiting, intestinal obstruction).
It is intensively bound to proteins and blood-forming elements (concentration in whole blood is 2-9 times higher than in plasma). Protein binding is 90% (mainly with lipoproteins). It is distributed mainly outside the bloodstream: 33-47% in plasma, 4-9% in lymphocytes, 5-12% in granulocytes, 41-58% in erythrocytes. After oral administration Tmax in plasma is 1.5-3.5 hours. Intensively metabolized in liver by cytochrome P4503A, to a lesser extent in gastrointestinal tract and kidneys with formation of 15 identified metabolites. It is excreted with the bile; 6% of the administered dose is excreted with the urine. It is excreted with breast milk.
The T1/2 in adults is 19 h, in children 7 h, regardless of dose or route of administration.
Indications
Indications
Graft-versus-host disease (GVHD) – prevention and treatment; autoimmune diseases.
Indications related to transplantation: solid organ transplantation (prevention of graft rejection after allogeneic transplantation of the kidney, liver, heart, combined heart-lung, lung or pancreas; treatment of graft rejection in patients previously treated with other immunosuppressants; bone marrow transplantation (prevention of graft rejection after bone marrow transplantation, prevention and treatment of GVHD.
Indications not related to transplantation: endogenous uveitis (active, sight-threatening uveitis of the middle and posterior part of the eye of non-infectious etiology, if conventional therapy is unsuccessful or leads to severe adverse reactions), Behcet’s uveitis with recurrent attacks of inflammation affecting the retina; nephrotic syndrome (steroid-dependent and steroid-resistant forms in remission); rheumatoid arthritis (severe forms), psoriasis (severe forms when systemic therapy is required).
Pharmacological effect
Pharmacological effect
Ecoral has an immunosuppressive effect.
At the cellular level, it inhibits the formation and release of lymphokines, including IL-2 (T-lymphocyte growth factor). Blocks resting lymphocytes in the G0 or G1 phase of the cell cycle and suppresses antigen-dependent release of lymphokines by activated T lymphocytes. All data obtained indicate that cyclosporine acts specifically and reversibly on lymphocytes.
Does not inhibit hematopoiesis and does not affect the function of phagocytic cells.
Pharmacokinetics
After oral administration, Cmax in the blood is observed in the range from 1 to 6 hours, with bioavailability averaging 30% (20–50%) and increasing with increasing dose and duration of treatment. Absorption decreases after liver transplantation, with liver disease or gastrointestinal pathology (diarrhea, vomiting, intestinal obstruction).
Intensively binds to proteins and formed elements of blood (concentration in whole blood is 2–9 times higher than in plasma). Protein binding – 90% (mainly with lipoproteins). Distributed mainly outside the bloodstream: 33–47% is present in plasma, 4–9% in lymphocytes, 5–12% in granulocytes, 41–58% in erythrocytes. After oral administration, Tmax in plasma is 1.5–3.5 hours. It is intensively metabolized in the liver by cytochrome P4503A, to a lesser extent in the gastrointestinal tract and kidneys with the formation of 15 identified metabolites. Excreted in bile; kidneys with urine – 6% of the dose administered orally. Excreted in breast milk.
T1/2 in adults – 19 hours, in children – 7 hours, regardless of dose or route of administration.
Special instructions
Special instructions
Data on the experience of using the drug in elderly people are quite limited, but to date no deviations have been recorded in the condition of patients taking the drug at the recommended dose.
If a patient experiences an increase in blood pressure during treatment with cyclosporine, an increase in creatinine levels (more than 30% of the initial value), a dose reduction of 25–50% is necessary.
If it is impossible to control the side effect or in case of severe renal impairment, the drug is discontinued.
Active ingredient
Active ingredient
Cyclosporine
Composition
Composition
1 capsule contains:
Active substances:
cyclosporine 25 mg,
excipients:
ethyl alcohol;
macrogol glyceryl hydroxystearate;
polyglyceryl (3) oleate;
polyglyceryl (10) oleate;
D,L-alpha tocopherol.
Pregnancy
Pregnancy
Experience with the use of cyclosporine in pregnant women is limited.
Data obtained from patients in the post-transplant period show that treatment with cyclosporine increases the risk of negative effects on the course and outcome of pregnancy.
If it is necessary to prescribe Ecoral, breastfeeding should be stopped.
Contraindications
Contraindications
Hypersensitivity to the components of Ecoral, malignant neoplasms and precancerous skin diseases, chicken pox, Herpes zoster (risk of generalization of the process), severe liver failure, hyperkalemia, hypertension, malabsorption syndrome, infectious diseases in the acute phase.
Side Effects
Side Effects
In the first week of using Ecoral, you may experience a burning sensation on the skin of the extremities.
After organ transplantation, the most common occurrences are: hypertrichosis, tremor, renal dysfunction and liver dysfunction, gum hypertrophy, gastrointestinal disorders (anorexia, nausea, vomiting); dose-dependent and reversible increase in the level of creatinine, urea, bilirubin, liver enzymes in the blood serum (careful monitoring of parameters and adjustment of the dosage of cyclosporine is necessary).
Hypertension most often develops during heart transplantation; this side effect occurs less frequently during kidney transplantation.
The most common symptoms that occur during bone marrow transplantation are: gastrointestinal disorders, tremor, hypertrichosis, and possible facial swelling. Children experience swelling and cramps.
Adverse reactions are moderate and are usually eliminated by reducing the dosage of Ecoral.
Interaction
Interaction
Inducers or inhibitors of cytochrome P450 can reduce or increase the concentration of cyclosporine in the blood.
Drugs that reduce the concentration of cyclosporine: barbiturates, carbamazepine, phenytoin, nafcillin, sulfadimidine (with intravenous administration); rifampicin, octreotide, probucol, orlistat; preparations containing St. John’s wort (Hypericum perforatum); troglitazone.
Drugs that increase the concentration of cyclosporine: some antibiotics – macrolides (mainly erythromycin and clarithromycin); ketoconazole, fluconazole, itraconazole, diltiazem, nicardipine, verapamil, metoclopramide, oral contraceptives, danazol, methylprednisolone (high doses); allopurinol, amiodarone, cholic acid and its derivatives.
Oral erythromycin should be avoided (increases the concentration of cyclosporine in the blood). If, due to the lack of alternative therapy, erythromycin is prescribed, it is recommended to carefully monitor the concentration of cyclosporine in the blood, renal function and the presence of side effects of cyclosporine.
Caution should be exercised when simultaneously prescribing drugs with nephrotoxic effects, for example: aminoglycosides (including gentamicin, tobramycin), amphotericin B, ciprofloxacin, vancomycin, trimethoprim (+ sulfamethoxazole), NSAIDs (including diclofenac, naproxen, sulindac), melphalan.
Vaccinations may be less effective during treatment with cyclosporine; The use of live attenuated vaccines should be avoided.
Combined use with nifedipine may lead to more pronounced gingival hyperplasia than with cyclosporine monotherapy.
May significantly increase the bioavailability of diclofenac (probably due to decreased metabolism) with the possible development of reversible renal impairment.
May reduce the clearance of digoxin, colchicine, lovastatin and prednisolone, leading to increased toxic effects, in particular muscle pain, weakness, myositis and, in rare cases, rhabdomyolysis.
Overdose
Overdose
There are no data on overdose with the drug to date and there is limited experience with overdose of other cyclosporines.
Symptoms: impaired renal function, which is probably reversible and disappears after discontinuation of the drug.
Treatment: according to indications – general supportive measures. The drug can only be eliminated from the body using nonspecific measures, including gastric lavage, since hemodialysis and hemoperfusion using activated charcoal are ineffective.
Storage conditions
Storage conditions
In a place protected from light, at a temperature not exceeding 25 °C
Shelf life
Shelf life
2 years
Manufacturer
Manufacturer
Teva Czech Enterprises s.r.o., Czech Republic
Additional information
| Shelf life | 2 years |
|---|---|
| Conditions of storage | In a light-protected place, at a temperature not exceeding 25 °C |
| Manufacturer | Teva Czech Enterprises s.r.o., Czech Republic |
| Medication form | capsules |
| Brand | Teva Czech Enterprises s.r.o. |
Related products
Buy Ecoral, capsules 25 mg 50 pcs with delivery to USA, UK, Europe and over 120 other countries.