No products in the cart.
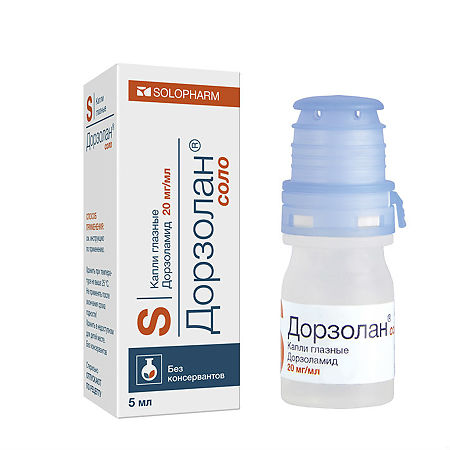
Dorzolan solo, eye drops 20 mg/ml 5 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
The drug is prescribed to adult patients with:
– ophthalmohypertension;
– primary open angle glaucoma;
– pseudoexfoliative glaucoma;
– secondary glaucoma (without anterior chamber angle block).
The drug is prescribed to children:
– For the treatment of glaucoma in children from the 1st week in the monotherapy mode or as an addition to the treatment with beta-adrenoblockers.
Active ingredient
Active ingredient
Dorzolamide
How to take, the dosage
How to take, the dosage
When using Dorzolamide-SOLOfarm, the usual dosage is 1 drop in the affected eye (or both eyes) in the morning, afternoon and evening.
Interaction
Interaction
No special studies on interaction of the drug Dorzolamide-SOLOfarm with other drugs have been conducted. In clinical studies the drug with dorzolamide in the form of eye drops was administered in combination with other drugs without negative manifestations of interdrug interactions, including: with timolol and betaxolol eye drops and systemic drugs: angiotensin-converting enzyme (ACE) inhibitors, calcium channel blockers, diuretics, nonsteroidal anti-inflammatory drugs (including acetylsalicylic acid), hormones (estrogen, insulin, thyroxine).
Special Instructions
Special Instructions
Sensitivity to dorzolamide may increase in the elderly (dose reduction required).
Contraindications
Contraindications
– Age less than 1 week;
– hypersensitivity to the components of the drug;
– chronic renal failure;
– pregnancy;
– lactation period.
With caution:
The drug has not been studied in patients with severe hepatic impairment and, therefore, should be used with caution in this category of patients.
Side effects
Side effects
In clinical trials, dorzolamide in the form of eye drops was administered to 1108 patients as monotherapy or adjunctive therapy to treatment with beta-adrenoblockers. In approximately 3% of patients the drug was discontinued due to local adverse ocular reactions, the most common of which were conjunctivitis and eyelid reactions.
The side effects reported during the studies and during the post-registration period are classified by frequency (very frequently (≥1/10); frequently (≥1/100, <1/10); infrequently (≥1/1000, <1/100); rarely (>1/10000, <1/1000)).
Overdose
Overdose
Symptoms
Possible electrolyte disturbances, the development of metabolic acidosis and the occurrence of drowsiness, nausea, dizziness, headache, weakness, unusual dreams, dysphagia.
Similarities
Similarities
Trusopt, Dorzopt, Dorzial
Additional information
| Weight | 0.020 kg |
|---|---|
| Shelf life | 2 years. Do not use after the expiration date! |
| Conditions of storage | At a temperature not higher than 25 ° C. Keep out of reach of children. |
| Manufacturer | Grotex Ltd, Russia |
| Medication form | eye drops |
| Brand | Grotex Ltd |
Related products
Buy Dorzolan solo, eye drops 20 mg/ml 5 ml with delivery to USA, UK, Europe and over 120 other countries.