No products in the cart.
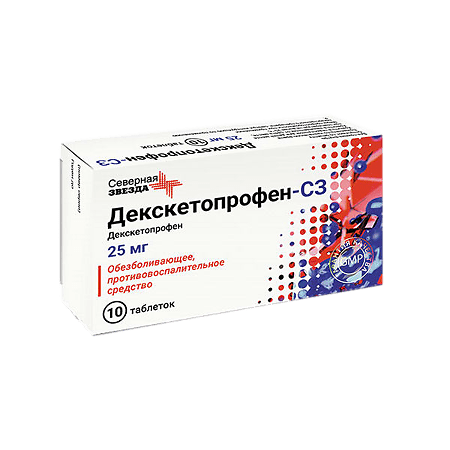
Dexketoprofen-SZ, 25 mg 10 pcs
€6.00 €5.42
Description
It has analgesic, anti-inflammatory and antipyretic effects. The mechanism of action of the drug is associated with inhibition of proslaglandin synthesis at the level of cyclooxygenases (COX-1 and COX-2).
The analgesic effect comes within 30 minutes after oral administration; therapeutic effect lasts for 4-6 hours.
In combined therapy with opioid analgesics, the drug significantly (up to 30-45%) reduces the need for opioids.
Indications
Indications
– relief of pain syndrome of various origins (including postoperative pain, pain with bone metastases, post-traumatic pain, pain with renal colic, algodismenorrhea, sciatica, sciatica, neuralgia, toothache, etc.);
– symptomatic treatment of acute and chronic inflammatory, inflammatory-degenerative and metabolic diseases of the musculoskeletal system (including rheumatoid arthritis, spondyloarthritis, osteoarthritis, osteochondrosis).
The tablets are intended for symptomatic therapy, reducing pain and inflammation at the time of use.
Pharmacological effect
Pharmacological effect
It has analgesic, anti-inflammatory and antipyretic effects. The mechanism of action of the drug is associated with inhibition of the synthesis of prostaglandins at the level of cyclooxygenases (COX-1 and COX-2).
The analgesic effect occurs 30 minutes after oral administration, the duration of the therapeutic effect reaches 4-6 hours.
When combined with opioid analgesics, the drug significantly (up to 30-45%) reduces the need for opioids.
Special instructions
Special instructions
During treatment with the drug, caution should be exercised when driving vehicles and engaging in potentially hazardous activities that require increased concentration and speed of psychomotor reactions.
Patients with gastrointestinal disorders or a history of gastrointestinal diseases require careful monitoring. If gastrointestinal bleeding or ulceration occurs, therapy should be discontinued.
It has been clinically proven that simultaneous use of low molecular weight heparin with drugs in prophylactic doses in the postoperative period does not change coagulation parameters. However, when used simultaneously with other drugs that affect blood clotting, careful medical monitoring of the blood clotting system is necessary.
Like other NSAIDs, the drug can lead to increased concentrations of creatinine and nitrogen in the blood plasma. Like other prostaglandin synthesis inhibitors, it may have side effects on the urinary system, which can lead to the development of glomerulonephritis. interstitial nephritis, papillary necrosis, nephrotic syndrome and acute renal failure.
Active ingredient
Active ingredient
Dexketoprofen
Composition
Composition
1 film-coated tablet contains:
active ingredient:
dexketoprofen trometamol – 36.9 mg, in terms of dexketoprofen – 25 mg,
excipients (core):
microcrystalline cellulose -135.2 mg;
pregelatinized starch (Starch 1500) – 50.0 mg,
sodium carboxymethyl starch – 27.5 mg,
magnesium stearate – 2.6 mg;
talc -7.8 mg.
excipients (shell): 8 mg (hypromellose – 4.3 mg; polysorbate-80 (Tween-80) – 1.2 mg; talc – 1.2 mg; titanium dioxide E 171 – 0.9 mg; aluminum varnish based on quinoline yellow dye – 0.4 mg).
Pregnancy
Pregnancy
Use during pregnancy and lactation is contraindicated.
Contraindications
Contraindications
– hypersensitivity to any of the components of the drug;
– peptic ulcer of the stomach and duodenum in the acute phase;
– history of gastrointestinal bleeding, other active bleeding (including suspected intracranial bleeding), anticoagulant therapy;
– inflammatory bowel diseases (Crohn’s disease, ulcerative colitis) in the acute phase;
– severe liver dysfunction (10-15 points on the Child-Pugh scale);
– moderate or severe renal impairment (creatinine clearance <50 ml/min);
– complete or incomplete combination of bronchial asthma, recurrent polyposis of the nose and paranasal sinuses and intolerance to acetylsalicylic acid or other non-steroidal anti-inflammatory drugs (including a history);
– severe heart failure;
– the period after coronary artery bypass surgery;
– hemorrhagic diathesis or other coagulation disorders;
– age under 18 years (there are no data on the effectiveness and safety of the drug).
With caution
– allergic reactions;
– violation of the blood coagulation system;
– systemic lupus erythematosus or mixed connective tissue diseases;
simultaneous therapy with other drugs (see section ‘Interaction with other drugs’);
– state of severe hypovolemia;
– coronary heart disease;
– cerebrovascular diseases;
– diabetes mellitus, hyperlipidemia;
– diseases of peripheral arteries;
– anamnestic data on the development of ulcerative lesions of the gastrointestinal tract;
– long-term use of NSAIDs;
– alcoholism, heavy smoking;
– old age (over 65 years).
Use during pregnancy and breastfeeding is contraindicated.
Side Effects
Side Effects
Disorders of the blood and lymphatic system: very rarely – neutropenia, thrombocytopenia.
Nervous system disorders: infrequently – headache, dizziness, insomnia, drowsiness, rarely – paresthesia.
Visual disturbances: very rarely – blurred vision.
Hearing and labyrinthine disorders: rarely – tinnitus. Disorders of the cardiovascular system: infrequently – feeling of heat, hyperemia of the skin, rarely – extrasystole. increased blood pressure, very rarely – tachycardia, decreased blood pressure.
Disorders of the respiratory system, thoracic and mediastinal organs: rarely – bradypnea, very rarely – bronchospasm, shortness of breath.
Gastrointestinal disorders: often – nausea, vomiting, abdominal pain, dyspepsia, diarrhea, infrequently – constipation, dry mouth. flatulence, rarely – erosive and ulcerative lesions of the gastrointestinal tract, bleeding from an ulcer or its perforation, anorexia, very rarely – damage to the pancreas.
Disorders of the liver and biliary tract: rarely – increased activity of liver enzymes, including aspartate aminotransferase and alanine aminotransferase (AST and ALT), jaundice, very rarely – liver damage. Renal and urinary tract disorders: rarely – polyuria, very rarely – nephritis or nephrotic syndrome.
Reproductive system disorders: rarely – in women – menstrual irregularities, in men – transient dysfunction of the prostate gland with long-term use.
Musculoskeletal and connective tissue disorders: rarely – back pain, muscle spasm, difficulty moving the joints.
Disorders of the skin and subcutaneous tissues: infrequently – dermatitis, rash, rarely – urticaria, acne, sweating, very rarely – severe skin reactions (Stevens-Johnson syndrome, Lyell’s syndrome), angioedema, allergic dermatitis, photosensitivity.
Metabolic disorders: rarely – hyperglycemia, hypoglycemia, hypertriglyceridemia.
Laboratory data and instrumental data: rarely – ketonuria, proteinuria. General disorders and disorders at the injection site: infrequently – fever, fatigue, very rarely – anaphylactic shock, facial swelling.
Other disorders: infrequently – aseptic meningitis, occurring mainly in patients with systemic lupus erythematosus or mixed connective tissue diseases, hematological disorders (purpura, aplastic and hemolytic anemia), rarely – agranulocytosis and bone marrow hypoplasia.
Interaction
Interaction
Undesirable drug combinations.
With other NSAIDs, including salicylates in high doses (more than 3 g per day):
simultaneous use of several NSAIDs due to the synergistic effect increases the risk of gastrointestinal bleeding and ulcers.
With oral anticoagulants, heparin in doses exceeding prophylactic doses, and ticlopidine: increased risk of bleeding due to inhibition of platelet aggregation and damage to the mucous membrane of the gastrointestinal tract.
With lithium preparations: NSAIDs increase the concentration of lithium in the blood, up to toxic levels, and therefore this indicator must be monitored during use, dose changes and after discontinuation of NG1VP.
With methotrexate in high doses (15 mg per week or more): increased hematological toxicity of methotrexate due to a decrease in its renal clearance during NSAID therapy.
With hydantoins and sulfonamides: the risk of increased toxic effects of these drugs.
Combinations requiring caution.
With diuretics, angiotensin-converting enzyme inhibitors: NSAID therapy is associated with a risk of acute renal failure in dehydrated patients (decreased glomerular filtration rate due to decreased prostaglandin synthesis). Taking the drug may reduce the antihypertensive effect of some drugs.
Overdose
Overdose
Symptoms: nausea, anorexia, abdominal pain, headache, dizziness, disorientation, insomnia.
Treatment: symptomatic therapy, if necessary – gastric lavage, hemodialysis.
Manufacturer
Manufacturer
North Star NAO, Russia
Additional information
| Manufacturer | North Star NAO, Russia |
|---|---|
| Medication form | pills |
| Brand | North Star NAO |
Related products
Buy Dexketoprofen-SZ, 25 mg 10 pcs with delivery to USA, UK, Europe and over 120 other countries.