No products in the cart.
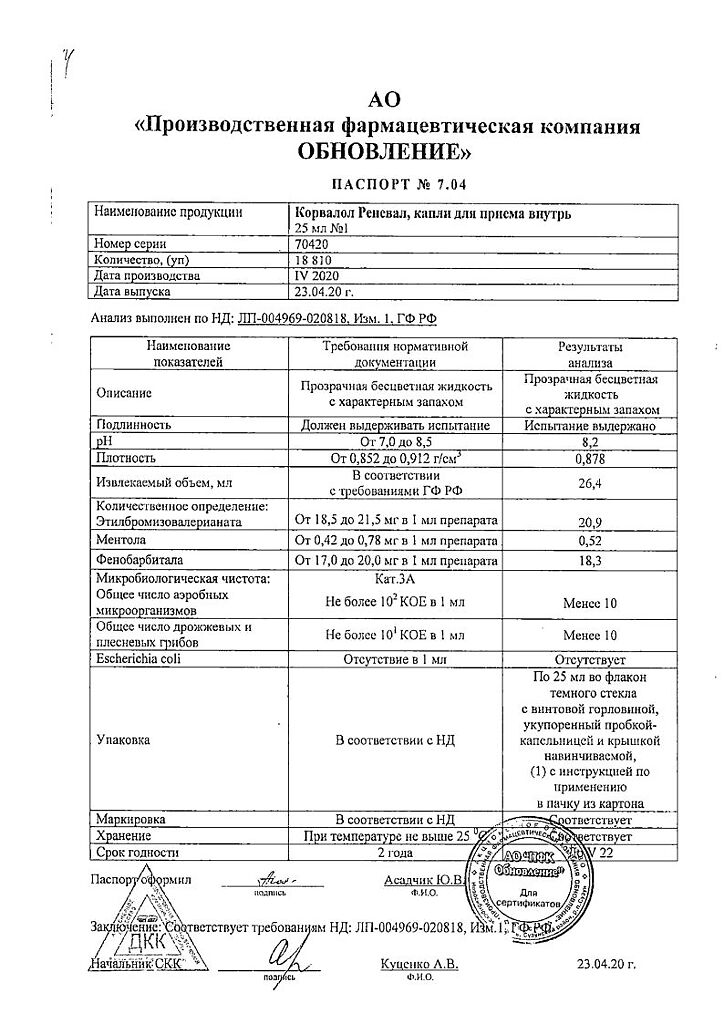
Corvalol Reneval, drops 25 ml
€6.25 €5.47
Out of stock
(E-mail when Stock is available)
EAN: 4603988016207
SKU: 306083
Categories: Medicine, Neurology and Psychiatry, Sedatives and hypnotics
Description
Pharmacotherapeutic group:
sedative.
ATX code: N05CM
Pharmacological properties
Pharmacodynamics
Combined drug, has sedative and antispasmodic effects, facilitates the onset of natural sleep.
Ethylbromisovalerianate has sedative and antispasmodic effect caused by irritation, mainly of receptors in the oral cavity and nasopharynx, reduction of reflex excitability in the central nervous system and increased inhibition in the cortex and subcortical structures of the brain, as well as reduced activity of central vasomotor centers and a direct local spasmolytic effect on smooth muscles.
Phenobarbital has sedative (in small doses), hypnotic, muscle relaxant and antispasmodic effect, helps to reduce agitation of the central nervous system (CNS) and facilitates sleep, increases sedative effects of other components.
Pearmint oil has a reflex vasodilatory, antispasmodic, mild choleretic, antiseptic action. The mechanism of action is related to the ability to irritate the “cold” receptors of the oral mucosa and reflexively dilate mainly vessels of the heart and brain. It eliminates the phenomena of flatulence by irritating the mucosal receptors of the gastrointestinal tract (GIT), increasing intestinal peristalsis.
Pharmacokinetics
There are no data on the pharmacokinetics of ethylbromisovalerianate and peppermint components.
Phenobarbital is slowly, completely absorbed when taken orally. Maximal concentration in blood plasma is determined after 1-2 hours. Its binding to plasma proteins is 50%, in newborns – 30-40%. It is metabolized in liver, induces microsomal liver enzymes by CYP3A4, CYP3A5, CYP3A7 isoenzymes (enzymatic reaction rate increases by 10-12 times). It accumulates in the body. Period of half-life is 2-4 days. It is excreted by kidneys as glucuronide, about 25% – unchanged. It penetrates into the breast milk and through the placental barrier.
Indications
Indications
As a symptomatic (sedative and vasodilator) agent for functional disorders of the cardiovascular system, for neurosis-like conditions accompanied by increased irritability, for difficulty falling asleep, tachycardia, a state of agitation with pronounced vegetative manifestations; as an antispasmodic – for intestinal spasms.
Pharmacological effect
Pharmacological effect
Pharmacotherapeutic group:
sedative.
ATX code: N05CM
Pharmacological properties
Pharmacodynamics
A combined drug that has a sedative and antispasmodic effect, facilitating the onset of natural sleep.
Ethyl bromizovalerianate has a sedative and antispasmodic effect due to irritation, mainly of the receptors of the oral cavity and nasopharynx, a decrease in reflex excitability in the central parts of the nervous system and increased inhibition in the neurons of the cortex and subcortical structures of the brain, as well as a decrease in the activity of the central vasomotor centers and a direct local antispasmodic effect on smooth muscles.
Phenobarbital has a sedative (in small doses), hypnotic, muscle relaxant and antispasmodic effects, helps reduce excitation of the central nervous system (CNS) and facilitates the onset of sleep, enhances the sedative effect of other components.
Peppermint oil has a reflex vasodilating, antispasmodic, mild choleretic, antiseptic effect. The mechanism of action is associated with the ability to irritate the “cold” receptors of the oral mucosa and reflexively dilate mainly the vessels of the heart and brain. Eliminates flatulence by irritating receptors in the mucous membrane of the gastrointestinal tract (GIT), enhancing intestinal motility.
Pharmacokinetics
Data on the pharmacokinetics of ethyl bromoisovalerate and peppermint components are not available.
When taken orally, phenobarbital is absorbed slowly and completely. The maximum concentration in blood plasma is determined after 1-2 hours, the connection with plasma proteins is 50%, in newborns – 30-40%. Metabolized in the liver, induces microsomal liver enzymes with isoenzymes CYP3A4, CYP3A5, CYP3A7 (the rate of enzymatic reactions increases 10-12 times). Cumulates in the body. The half-life is 2-4 days. It is excreted by the kidneys in the form of glucuronide, about 25% unchanged. Penetrates into breast milk and through the placental barrier.
Special instructions
Special instructions
1 drop of the drug contains 0.022 g of absolute ethyl alcohol.
The maximum single dose of the drug (50 drops) contains 1.1 g of absolute ethyl alcohol, the maximum daily dose (150 drops) contains 3.3 g of absolute ethyl alcohol.
Influence on the ability to drive vehicles and machinery
The drug contains at least 70 volume percent ethanol and phenobarbital, therefore, during the period of taking the drug Corvalol, it is necessary to refrain from driving vehicles and engaging in other potentially hazardous activities that require increased concentration and speed of psychomotor reactions.
Active ingredient
Active ingredient
Peppermint Leaf Oil, Phenobarbital, Ethyl Bromoisovalerianate
Composition
Composition
Active ingredients:
Ethyl bromoisovalerianate (ethyl ester of alpha-bromoisovaleric
acid) – 2000 mg
Phenobarbital – 1826 mg
Peppermint Leaf Oil
(peppermint oil) – 142 mg
Excipients:
ethanol (ethyl alcohol) 95% – 79 ml
sodium hydroxide
1 M solution – up to pH 8.0-8.5
purified water – up to 100 ml
Pregnancy
Pregnancy
The use of the drug Corvalol Renewal during pregnancy and breastfeeding is contraindicated, since the drug contains phenobarbital, which penetrates the placenta and has a teratogenic effect, has a negative effect on the formation and further functioning of the central nervous system of the fetus and newborn, penetrates into breast milk, and the development of physical dependence in the newborn is possible. If it is necessary to use it during breastfeeding, the issue of stopping breastfeeding should be decided.
Contraindications
Contraindications
Hypersensitivity to the components of the drug; severe renal and/or liver dysfunction; alcoholism; traumatic brain injury, brain diseases; pregnancy, breastfeeding period; children up to 3 years old.
With caution
Children’s age from 3 years.
Impaired liver and kidney function.
Side Effects
Side Effects
Drowsiness, dizziness, slow heart rate, decreased ability to concentrate, allergic reactions. Gastrointestinal disorders may occur. These phenomena disappear when the dose of the drug is reduced or the drug is stopped.
With long-term use of the drug, drug dependence, addiction, withdrawal syndrome may occur, as well as the accumulation of bromine in the body and the development of bromism phenomena (depressive mood, apathy, rhinitis, conjunctivitis, hemorrhagic diathesis, impaired coordination of movements).
If any of the side effects indicated in the instructions get worse, or you notice any other side effects not listed in the instructions, tell your doctor.
Interaction
Interaction
Medicines that depress the central nervous system enhance the effect of the drug.
Phenobarbital (an inducer of microsomal oxidation) may reduce the effectiveness of drugs metabolized in the liver (including coumarin derivatives, griseofulvin, glucocorticosteroids, oral contraceptives); enhances the effect of local anesthetics, analgesics and hypnotics.
The drug increases the toxicity of methotrexate.
The effect of the drug is enhanced by the use of valproic acid drugs.
Overdose
Overdose
Symptoms: depression of the central nervous system (CNS), nystagmus, ataxia, decreased blood pressure, agitation, dizziness, weakness, chronic bromine intoxication (depression, apathy, rhinitis, conjunctivitis, hemorrhagic diathesis, impaired coordination of movements).
Treatment: discontinuation of the drug, gastric lavage and symptomatic therapy; for central nervous system depression – caffeine, niketamide.
Storage conditions
Storage conditions
At a temperature not exceeding 25 °C.
Keep out of the reach of children.
Shelf life
Shelf life
2 years.
Do not use after expiration date.
Manufacturer
Manufacturer
Update of PFC JSC, Russia
Additional information
| Shelf life | 2 years. Do not use after the expiration date. |
|---|---|
| Conditions of storage | At a temperature not higher than 25 ° C. Keep out of reach of children. |
| Manufacturer | Update PFC AO, Russia |
| Medication form | oral drops |
| Brand | Update PFC AO |
Other forms…
Related products
Buy Corvalol Reneval, drops 25 ml with delivery to USA, UK, Europe and over 120 other countries.