No products in the cart.
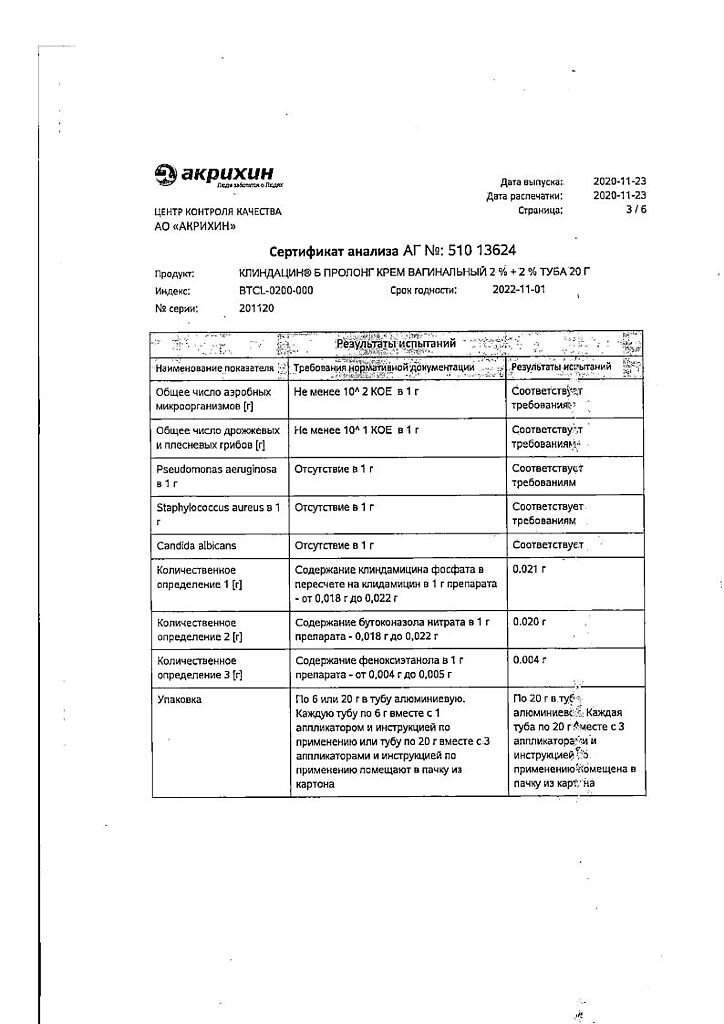
Clindacin B prolong, vaginal cream 2%+2% 20 g
€17.87 €14.89
Description
Pharmacotherapeutic group: Antimicrobial and antifungal agent.
Pharmacological action
Pharmacodynamics
Butoconazole – Imidazole derivative, has fungicidal activity against Candida fungi, Trichophyton, Microsporum, Epidermaphyton and some Gram-positive bacteria. It is most effective against candidiasis. By blocking in the cell membrane formation of ergosterol from lanosterol it increases membrane permeability, which leads to lysis of the fungus cell.
Clindamycin is a bacteriostatic antibiotic of the lincosamide group, it has a broad spectrum of action, binds to 50S subunit of ribosomal membrane and inhibits protein synthesis in a microbial cell. Bactericidal action is possible against a number of Gram-positive cocci. Under in vitro conditions the following microorganisms causing bacterial vaginosis are sensitive to clindamycin: Gardnerella vaginalis, Mobiluncus spp., Mycoplasma hominis, Bacteroides spp., Peptostreptococcus spp.
The hydrophilic cream base provides a gel-like consistency of the drug at 35-40 ° C. The cream does not melt when applied intravaginally, which ensures that the active substances remain on the vaginal mucosa for 1-3 days. There is a cross-resistance between clindamycin and lincomycin.
Pharmacokinetics
Butoconazole
In intravaginal administration, approximately 1.7% of the administered dose of butoconazole. Cmax in plasma butoconazole is reached after 13 hours and is 2-18.6 ng/ml. Butoconazole is extensively metabolized, partially excreted by the kidneys and intestine.
Clindamycin
absorption
. After use of clindamycin intravaginally at a dose of 100 mg/day once (as 2% clindamycin phosphate cream) for 7 days, the Cmax of clindamycin in plasma is reached after 10 h (4-24 h) and averages 18 ng/mL (4-47 ng/mL) on day 1 and 25 ng/mL (6-61 ng/mL) on day 7, with systemic absorption of about 4% (06-11%) of the administered dose.
In women with bacterial vaginosis, approximately 4% of clindamycin undergoes systemic absorption (with a smaller variation of 2-8%) with a similar dosing regimen, Cmax is reached 14 h (4-24 h) after administration and
averages 13 ng/mL (6-34 ng/mL) on day 1 and 16 ng/mL (7-26 ng/mL) on day 7. The systemic effects of clindamycin are less pronounced with intravaginal administration than with oral or IV administration.
Evolution
The T1/2 is 1.5-2.6 h. Clindamycin almost does not cumulate after repeated intravaginal administration.
Pharmacokinetics in Special Patient Groups
Not enough patients aged 65 years or older have participated in clinical trials of clindamycin 2% vaginal cream to allow assessment of the difference in clinical response to therapy between this age group and younger patients. In the available reports from clinical experience, no difference in response between older and younger patients was observed.
Indications
Indications
Bacterial vaginosis, fungal and mixed vaginitis, caused by microorganisms sensitive to the drug.
Active ingredient
Active ingredient
Butoconazole, Clindamycin
Composition
Composition
Vaginal cream from almost white to white with a grayish tint, with a specific smell.
| 100 g | ||
| Butoconazole nitrate | 2 g | |
| clindamycin phosphate | 2.376 mg, | |
| which corresponds to the content of clindamycin | 2 g |
Associates: Preservative Euxyl PE 9010 (phenoxyethanol 90%, ethylhexylglycerol 10%) – 0.5 g, which is equivalent to 0.45 g of phenoxyethanol, propylene glycol – 5 g, isopropyl myristate – 8 g, macrogoal cetostearate (macrogoal-20 cetostearyl ether) – 2 g, cetostearyl alcohol (cetyl alcohol 60%, stearyl alcohol 40%) – 6 g, hydroxypropyl diacrystalline phosphate – 8 g, sodium hydroxide – 0.26 g, purified water – up to 100 g.
How to take, the dosage
How to take, the dosage
It is applied intravaginally with an applicator once a day, preferably before bedtime.
Recommended dose is 1 full applicator (5 g of cream, corresponding to 100 mg of butoconazole nitrate and 100 mg of clindamycin). The course of treatment is daily for 3 days.
Instructions for use
Disposable applicators included in the package are designed for precise dosing and introduction of the cream into the vagina.
1. Open the tube of cream by removing the cap. Pierce the membrane of the tube with the back side of the cap. Screw the plastic applicator to the threaded neck of the tube.
2. gently press the tube from the opposite end, gently squeeze the cream into the applicator. The applicator piston moves independently as it fills the required volume. The applicator is full when its piston reaches the stop. Unscrew the applicator and close the tube.
3. In the supine position, pull your knees up to your chest. Holding the applicator horizontally, gently insert it into the vagina as deep as possible, taking care not to cause discomfort.
4. Slowly pressing the piston to the stop, inject the entire volume of cream into the vagina.
5. Carefully remove the applicator from the vagina and throw it in a trash container.
Interaction
Interaction
There is a cross-resistance between clindamycin and lincomycin. Under in vitro conditions anatagonism between clindamycin and erythromycin has been demonstrated.
It has been found that clindamycin in systemic use impairs neuromuscular transmission and, therefore, may enhance the effect of peripheral muscle relaxants, therefore the drug should be used with caution in patients receiving this group of drugs.
It is not recommended to combine with other drugs for intravaginal administration. It is incompatible with solutions containing vitamin B complex, aminoglycosides, ampicillin, calcium gluconate and magnesium sulfate.
Special Instructions
Special Instructions
Trichomonas vaginalis, Chlamidia trachomatis, Neisseria gonorrhoeae and Herpes simplex virus, which often cause vulvovaginitis, should be excluded by appropriate laboratory methods before prescription. Intravaginal use of clindamycin may lead to increased growth of insensitive microorganisms, especially yeast-like fungi.
Use of clindamycin (like virtually all antibiotics) orally or parenterally is associated with the development of severe diarrhea and in some cases pseudomembranous colitis. If severe or prolonged diarrhea develops, the drug should be discontinued and appropriate diagnostic and therapeutic measures should be taken if necessary. Patients should be warned not to have sexual intercourse during therapy with the drug.
Vaginal cream is not recommended for simultaneous use with other intravaginal drugs. It is not recommended to use the drug during menstruation.
The drug contains components that may reduce the strength of latex or rubber products, therefore the use of condoms, vaginal birth control diaphragms and other latex products for intravaginal use during therapy with the drug and for 72 hours after application is not recommended. If clinical signs of infection persist after the completion of treatment, repeat microbiological examination to identify the causative agent and confirm the diagnosis.
The cetostearyl alcohol contained in the drug may cause local skin reactions (rash, contact dermatitis). The appearance of irritation of the vaginal mucosa or painful sensations is an indication to discontinue treatment.
Impact on ability to drive motor transport and operating machinery
There is no reason to believe that the use of the drugi>Clindacin® B Prololng vaginal cream may affect the ability to drive and operate machinery.
Contraindications
Contraindications
– Hypersensitivity to clindamycin, lincomycin, butoconazole or any component of the drug;
– Crohn’s disease;
– inflammatory bowel diseases (Crohn’s disease, ulcerative, pseudomembranous and antibiotic-associated colitis (including history).
– age under 18 years.
With caution:allergic diseases, concomitant use of muscle relaxants.
Side effects
Side effects
The drug is usually well tolerated, but the following adverse effects may occur:
Butoconazole
Genital and mammary glands: irritation at the injection site, burning, itching, painfulness and swelling of the vaginal mucosa.
From the digestive system: pain/spasms in the lower abdomen.
From the immune system: development of allergic reactions.
Clindamycin
The safety of clindamycin vaginal cream was evaluated in both non-pregnant patients and patients in the second and third trimesters of pregnancy. Adverse drug reactions reported when using clindamycin vaginal cream. Definition of the frequency of adverse reactions: very common (≥1/10), common (≥1/100 to < 1/10), infrequent (≥1/1000 to < 1/100), rare (≥1/10 000 to < 1/1000), very rare (< 1/10 000), frequency unknown (cannot be estimated based on available data).
Infectious and parasitic diseases: often – fungal infections caused by fungi of the genus Candida; infrequent – bacterial infections; frequency unknown – candidiasis of the skin.
Infections of the immune system: infrequent – hypersensitivity.
From the endocrine system: frequency unknown – hyperthyroidism.
From the nervous system: often – headache, dizziness, dysgeusia.
Hearing and vestibular system side: infrequently – vertigo.
Breathing system side: frequently – upper respiratory tract infections; infrequently – nasal bleeding.
In the digestive system:often – abdominal pain, constipation, diarrhea, nausea, vomiting; infrequent – bloating, flatulence, bad breath; frequency unknown – pseudomembranous colitis *, gastrointestinal disorders, dyspepsia.
Skin and subcutaneous tissue: frequently – skin itching, rash; infrequently – urticaria, erythema; frequency unknown – maculopapular rash.
Muscular system disorders:often – back pain.
Urinary system disorders:often – urinary tract infections, glucosuria, proteinuria; infrequent – dysuria.
Pregnancy, postpartum and perinatal conditions: often – abnormal delivery.
From the genital organs and mammary gland: very common – vulvovaginal candidiasis; common – vulvovaginitis, vulvovaginal disorders, menstrual cycle disorders, vulvovaginal pain, metrorrhagia, vaginal discharge; infrequent – trichomonad vulvovaginitis, vaginal infections, pelvic pain; frequency unknown – endometriosis.
General disorders and reactions at the point of administration: frequency unknown – inflammation, pain.
Data of laboratory and instrumental studies: infrequent – abnormal microbiological test results.
* Adverse drug reactions identified in the post-registration period.
Overdose
Overdose
No overdose has been established during intravaginal administration of the drug. Absorption of clindamycin in amounts sufficient for the development of systemic reactions is possible. Accidental ingestion of the drug in the gastrointestinal tract may also cause systemic effects similar to those occurring after oral administration of clindamycin in therapeutic doses.
Possible systemic side effects include diarrhea, hemorrhagic diarrhea including pseudomembranous colitis (see sections “Side effects” and “Cautions”).
Treatment: symptomatic and supportive.
Additional information
| Weight | 0.060 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | The drug should be kept out of reach of children at 15°C to 25°C. |
| Manufacturer | Akrihin HFC JSC, Russia |
| Medication form | vaginal cream |
| Brand | Akrihin HFC JSC |
Related products
Gynecology and Obstetrics
Gynecology and Obstetrics
Buy Clindacin B prolong, vaginal cream 2%+2% 20 g with delivery to USA, UK, Europe and over 120 other countries.