Subtotal: €37.44
Cerebrolysin, 5 ml 5 pcs
€29.04 €24.20
Cerebrolysin has a positive effect on cognitive disorders and memory processes.
Indications
chronic cerebrovascular insufficiency;
ischemic stroke;
Alzheimer’s disease;
dementia syndrome of various origins;
traumatic injuries of the brain and spinal cord;
mental retardation in children;
hyperactivity and attention deficit in children;
in complex therapy for endogenous depression resistant to antidepressants.
Pharmacological effect
Cerebrolysin has a positive effect on cognitive impairment and memory processes.
Special instructions
The compatibility of the drug has been tested and confirmed (within 24 hours at room temperature and light) with the following standard solutions for infusion:
– 0.9% sodium chloride solution (9 mg NaCl/ml);
– Ringer’s solution (Na+ – 153.98 mmol/l; Ca2+ – 2.74 mmol/l; K+ – 4.02 mmol/l; Cl− – 163.48 mmol/l).
– 5% glucose solution.
The simultaneous administration of Cerebrolysin with vitamins and drugs that improve cardiac circulation is allowed, but these drugs should not be mixed in the same syringe with Cerebrolysin. Only use a clear solution and only once.
Impact on the ability to drive vehicles and operate machinery. Clinical trials have shown that Cerebrolysin does not affect the ability to drive vehicles and use machinery.
Active ingredient
Brain peptide complex
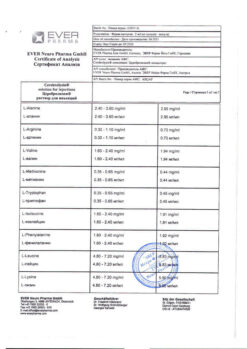
Composition
1 ml of injection solution contains:
active substance:
Cerebrolysin concentrate (a complex of peptides obtained from pig brain) 215.2 mg,
excipients:
sodium hydroxide;
water for injections
Pregnancy
The drug is prescribed with caution in the first trimester of pregnancy and during lactation.
During pregnancy and breastfeeding, Cerebrolysin should be used only after a thorough analysis of the relationship between the positive effect of treatment and the risk associated with it.
The results of experimental studies do not give reason to believe that Cerebrolysin has a teratogenic effect or has a toxic effect on the fetus.
However, similar clinical studies have not been conducted.
Contraindications
acute renal failure;
individual intolerance to the drug;
status epilepticus.
Side Effects
From the gastrointestinal tract: rarely – loss of appetite, dyspepsia, diarrhea, constipation, nausea and vomiting.
From the central nervous system and peripheral nervous system: in rare cases, the expected effect of activation was accompanied by agitation (manifested by aggressive behavior, confusion, insomnia). There are reports of the occurrence in isolated cases (
From the immune system: extremely rarely – hypersensitivity reactions or allergic reactions, manifested by headache, pain in the neck, limbs, lower back, shortness of breath, chills and collapsing state.
Reaction to administration: with excessively rapid administration, in rare cases, a feeling of heat, sweating, and dizziness may occur; in isolated cases – rapid heartbeat or arrhythmias. Therefore, the drug should be administered slowly.
Local reactions: rarely – redness of the skin, itching and burning at the injection site.
Other: extremely rare – studies have reported cases of hyperventilation, arterial hypertension, hypotension, fatigue, tremor, depression, apathy, dizziness and flu-like symptoms (cough, runny nose, respiratory tract infections).
It should be noted that some undesirable effects (excitement, arterial hypertension, arterial hypotension, lethargy, tremor, depression, apathy, dizziness, headache, shortness of breath, diarrhea, nausea) were identified during clinical trials and occurred equally in patients receiving Cerebrolysin and in patients in the placebo group.
Interaction
Taking into account the pharmacological profile of the drug Cerebrolysin, special attention should be paid to possible additive effects when co-administered with antidepressants, incl. MAO inhibitors. In such cases, it is recommended to reduce the dose of the antidepressant.
Cerebrolysin and balanced solutions of amino acids should not be mixed in the same solution for infusion.
Cerebrolysin is incompatible with solutions containing lipids and with solutions that change the pH of the medium (5–8).
Storage conditions
In a place protected from light, at a temperature not exceeding 25 °C
Shelf life
5 years
Manufacturer
Sotex/EVER Pharma Jena GmbH, Russia
| Shelf life | 5 years |
|---|---|
| Conditions of storage | In a light-protected place, at a temperature not exceeding 25 °C |
| Manufacturer | EVER Pharma Jena GmbH, Germany |
| Medication form | solution for injection |
| Brand | EVER Pharma Jena GmbH |
Other forms…
Related products
Buy Cerebrolysin, 5 ml 5 pcs with delivery to USA, UK, Europe and over 120 other countries.