No products in the cart.
Description
Pharmacodynamics
An alkylating antitumor agent containing platinum. Mechanism of action is associated with formation of cross-links between neighboring pairs of guanine bases in DNA, which leads to suppression of nucleic acid biosynthesis and cell death.
Pharmacokinetics
Metabolized by hydrolysis to form active compounds that interact with DNA. Binding to proteins is very low. However, platinum formed from carboplatin binds irreversibly to plasma proteins and is slowly excreted with a minimum T1/2 of 5 days.
The T1/2 of carboplatin is 1.1-2 h in the initial phase and 2.6-5.9 h in the final phase. Renal excretion is 71% within 24 hours with a CK of 60 ml/min or more.
Indications
Indications
Ovarian cancer;
germ cell tumors of the testicle and ovaries;
seminoma;
malignant melanoma;
head and neck tumors;
lung cancer;
cancer of the cervix and uterine body;
bladder cancer;
osteogenic sarcoma.
Pharmacological effect
Pharmacological effect
Pharmacodynamics
Antitumor agent with alkylating action, contains platinum. The mechanism of action is associated with the formation of cross-links between adjacent guanine base pairs in DNA, which leads to suppression of nucleic acid biosynthesis and cell death.
Pharmacokinetics
Metabolized by hydrolysis to form active compounds that interact with DNA. Protein binding is very low. However, platinum formed from carboplatin is irreversibly bound to plasma proteins and is slowly eliminated with a minimum T1/2 of 5 days.
T1/2 of carboplatin is 1.1-2 hours in the initial phase, 2.6-5.9 hours in the final phase. 71% is excreted by the kidneys within 24 hours with a CC of 60 ml/min or more.
Special instructions
Special instructions
Caution should be used in cases of suppression of bone marrow hematopoiesis (including against the background of concomitant radiation or chemotherapy), previous therapy with nephrotoxic drugs (for example, cisplatin), hearing impairment, acute infectious diseases of a viral, fungal or bacterial nature, in patients with ascites or exudative pleurisy, post-vaccination period.
Caution should be exercised when using carboplatin in patients following a course of radiation therapy.
Carboplatin is used only under the supervision of a physician experienced in chemotherapy. Before starting treatment and during it, it is necessary to monitor renal function, peripheral blood picture, neurological status, and conduct audiometry. Changes in biochemical parameters are possible: increased levels of urea and creatinine in the blood serum, decreased concentrations of magnesium, potassium, calcium.
While using carboplatin, it is not recommended to vaccinate patients or their family members.
Active ingredient
Active ingredient
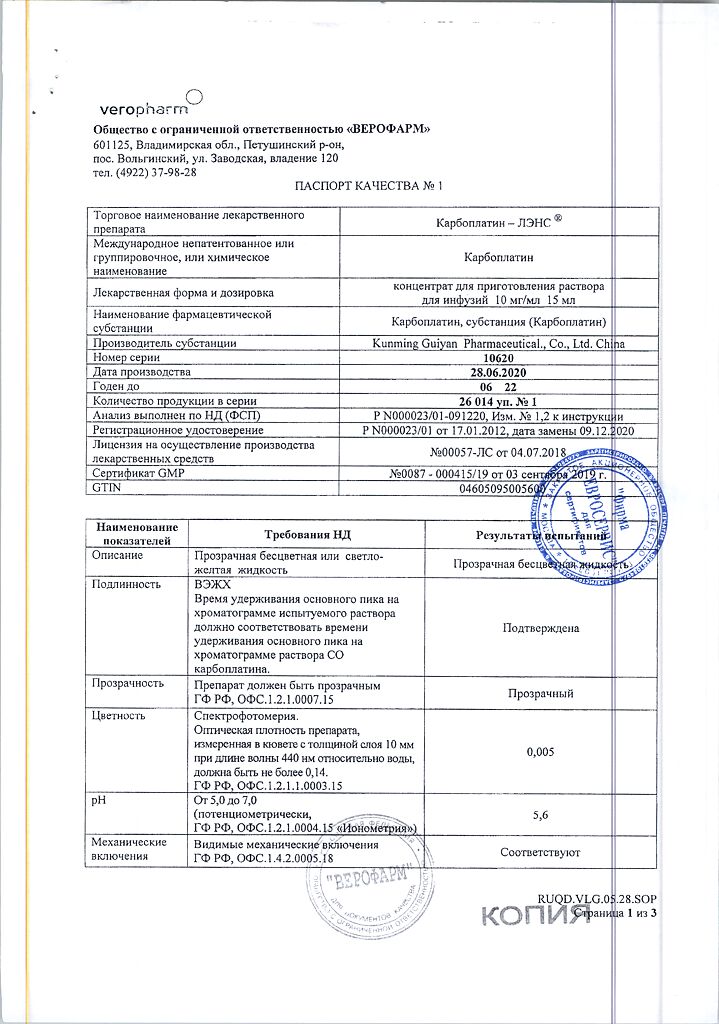
Carboplatin
Composition
Composition
1 ml (1 bottle) contains:
Active substances:
carboplatin 10 mg (150 mg).
Pregnancy
Pregnancy
Contraindicated for use during pregnancy and lactation (breastfeeding).
Women of childbearing potential receiving carboplatin therapy should use reliable contraception.
Experimental studies have shown that carboplatin has a teratogenic and embryotoxic effect.
Contraindications
Contraindications
Severe renal dysfunction;
previous severe myelodepression;
recent significant blood loss;
pregnancy;
lactation period (breastfeeding);
childhood;
hypersensitivity to carboplatin and drugs containing platinum.
Side Effects
Side Effects
From the hematopoietic system: inhibition of bone marrow hematopoiesis.
From the digestive system: nausea, vomiting, stomatitis, diarrhea or constipation, abdominal pain, decreased appetite, impaired liver function (increased AST activity, alkaline phosphatase and bilirubin concentration in the blood serum).
From the nervous system: asthenia, peripheral polyneuropathy (paresthesia, decreased deep tendon reflexes), decreased visual acuity up to complete loss of vision or loss of the ability to distinguish colors (improvement or complete restoration of vision usually occurs within a few weeks after stopping the drug; cortical blindness was observed in patients with impaired renal function treated with high doses of carboplatin), decreased hearing, tinnitus; long-term therapy may result in cumulative neurotoxicity.
From the urinary system: increased concentrations of creatinine and urea in the blood serum (acute kidney damage was observed rarely, the risk of nephrotoxicity while taking carboplatin increases with increasing doses of carboplatin, as well as in patients who have previously been treated with cisplatin).
From the reproductive system: azoospermia, amenorrhea.
From the side of water and electrolyte balance: hypokalemia, hypocalcemia, hyponatremia and hypomagnesemia.
Allergic reactions: erythematous rash, fever, itching, urticaria, bronchospasm, decreased blood pressure, anaphylactoid reactions, allergic reactions at the injection site; rarely – exfoliative dermatitis.
Other: changes in taste, alopecia, flu-like symptoms (fever, fever), hemolytic-uremic syndrome, myalgia/arthralgia, heart failure, cerebrovascular disorders.
Interaction
Interaction
When used simultaneously with drugs that have myelosuppressive, nephrotoxic effects, mutual enhancement of toxic effects is possible.
Overdose
Overdose
Symptoms: increased adverse reactions.
Treatment: Specific antidotes for carboplatin overdose are unknown. Treatment is symptomatic. In the first 3 hours after administration of the drug, hemodialysis may be used.
Storage conditions
Storage conditions
In a place protected from light, at a temperature not exceeding 25 °C (do not freeze)
Shelf life
Shelf life
2 years
Manufacturer
Manufacturer
Veropharm LLC, Russia
Additional information
| Shelf life | 2 years |
|---|---|
| Conditions of storage | In the dark place at a temperature not exceeding 25 °C (do not freeze) |
| Manufacturer | Veropharm AO, Russia |
| Medication form | concentrate for preparation of infusion solution |
| Brand | Veropharm AO |
Related products
Buy Carboplatin-LENS, 10 mg/ml 15 ml with delivery to USA, UK, Europe and over 120 other countries.