No products in the cart.
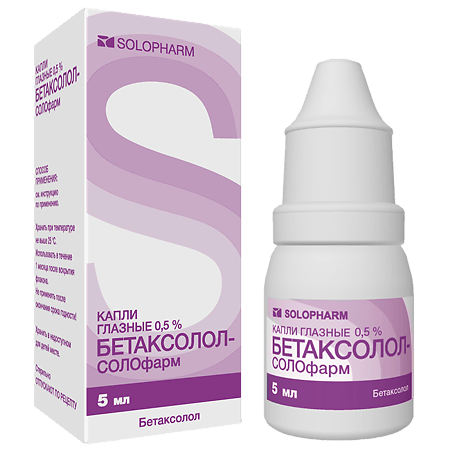
Betaxolol-Solofarm, eye drops 0.5% 5 ml
€10.76 €9.42
Description
Glaucoma, Elevated Intraocular Pressure
Reduction of elevated intraocular pressure in patients with ophthalmohypertension and primary open-angle glaucoma as monotherapy or in combination with other drugs.
Indications
Indications
Reduction of elevated intraocular pressure in patients with ocular hypertension and primary open-angle glaucoma as monotherapy or in combination with other drugs.
Pharmacological effect
Pharmacological effect
Antiglaucoma agent – beta blocker
Special instructions
Special instructions
Diabetes mellitus
Beta-blockers should be prescribed with caution to patients with a tendency to spontaneous hypoglycemia and patients with labile diabetes mellitus, since these drugs may mask the signs and symptoms of acute hypoglycemia.
Thyrotoxicosis
Beta blockers may mask some symptoms of hyperthyroidism (eg, tachycardia). In patients with suspected thyrotoxicosis, beta-blockers should not be abruptly discontinued, as this may cause an increase in symptoms.
Myasthenia gravis
Beta blockers may cause symptoms and signs similar to those of myasthenia gravis (eg, diplopia, ptosis, general weakness).
Surgery
The anesthesiologist should be informed that the patient is taking betaxolol. Before a planned operation, beta-blockers should be gradually (not all at once!) withdrawn 48 hours before general anesthesia, because during general anesthesia, they can reduce the sensitivity of the myocardium to sympathetic stimulation necessary for heart function (for example, they can block the action of adrenaline).
Pulmonology
There have been reports of respiratory reactions, including death, due to bronchospasm in patients with bronchial asthma when using some beta-blockers in ophthalmology.
Betaxolol should be prescribed with caution to patients with moderate to moderate bronchial asthma (including a history of asthma), and to patients with mild to moderate chronic obstructive pulmonary disease.
Anaphylactic reactions
When betaxolol is used by patients with atopy or a history of severe anaphylactic reactions to various allergens, there may be a more pronounced reaction to repeated administration of these allergens and unresponsiveness to standard doses of epinephrine in relieving anaphylactic reactions.
Betaxolol should be used with caution in patients with severe peripheral circulatory disorders (i.e., patients with severe Raynaud’s disease or Raynaud’s syndrome, as well as pheochromocytoma).
When administered locally, beta-blockers can enter the systemic circulation and cause undesirable reactions from the cardiovascular, pulmonary and other systems.
Cases of severe respiratory and cardiovascular disorders have been described, including death from bronchospasm in patients with bronchial asthma and death from heart failure when using betaxolol.
Heart disorders
In patients with cardiovascular disease (eg, coronary artery disease, Prinzmetal’s angina, heart failure) and arterial hypotension, beta-blocker therapy should be critically evaluated with consideration of the possibility of treatment with drugs from other groups. Careful monitoring of the development of signs of exacerbation of the disease and adverse reactions in patients suffering from cardiovascular diseases is necessary.
Corneal diseases
Beta blockers may cause dry eyes. In patients with corneal diseases, the drug should be used with caution.
The main pathogenetic aspect of the treatment of angle-closure glaucoma is the need to open the anterior chamber angle, which is achieved by narrowing the pupil with the help of miotics. Betaxolol does not affect pupil diameter; therefore, for open-angle glaucoma, the drug should be used only in combination with mystics.
Choroidal detachment
Cases of choroidal detachment have been described when using drugs that reduce the production of intraocular fluid (for example, timolol, acetazolamide) after fistulizing antiglaucomatous operations.
This product contains the preservative benzalkonium chloride, which may cause eye irritation and discoloration of soft contact lenses. Direct contact of the drug with soft contact lenses should be avoided. Patients using contact lenses should remove the lenses before using the drug and put them back no earlier than 15 minutes after instillation.
Impact on the ability to drive vehicles. Wed and fur.:
During the treatment period, care must be taken when driving vehicles and engaging in other potentially hazardous activities that require increased concentration and speed of psychomotor reactions.
Active ingredient
Active ingredient
Betaxolol
Composition
Composition
1 ml betaxolol hydrochloride 5.6 mg, which corresponds to the content of betaxolol 5 mg
Excipients:
benzalkonium chloride – 0.1 mg,
sodium chloride – 5.49 mg,
disodium phosphate dihydrate – 3.579 mg,
disodium edetate (trilon B) – 0.5 mg,
sodium dihydrogen phosphate dihydrate – 3.165 mg,
water for d/i – up to 1 ml.
Contraindications
Contraindications
– Hypersensitivity to betaxolol and other components of the drug;
– bradycardia;
– atrioventricular block of the II and III degrees, uncontrolled by an artificial pacemaker;
– sick sinus syndrome;
– sinoatrial blockade;
– cardiogenic shock;
– decompensated chronic heart failure;
– reactive respiratory diseases, including severe bronchial asthma (including a history);
– severe chronic obstructive pulmonary disease.
With caution:
Treatment of patients using β1-blocker agents and betaxolol (eye drops) should be carried out under regular medical supervision due to the risk of a cumulative potential effect on intraocular pressure and the occurrence of undesirable systemic effects.
Despite the weak general effect of betaxolol (in the form of eye drops) on the body, caution should be exercised when prescribing it to patients with thyrotoxicosis and diabetes mellitus (especially unstable), since the drug can mask the manifestations of hypoglycemia.
Before surgery using general anesthesia, the use of the drug should be gradually discontinued, as the drug may change the patient’s response to the use of general anesthetics.
Side Effects
Side Effects
Like other drugs in the form of eye drops, betaxolol can enter the systemic circulation, which leads to the development of systemic side effects.
Adverse reactions are presented in accordance with the following gradation of the frequency of their development: very often (≥ 10%); often (≥ 1, < 10%); uncommon (≥ 0.1, < 1%); rare (≥ 0.01, < 0.1%); very rare (<0.01%); frequency is unknown (it is not possible to determine the frequency of development based on available data).
Visual disorders
Very common: discomfort in the eyes.
Common: blurred vision, increased lacrimation.
Uncommon: punctate keratitis, keratitis, conjunctivitis, blepharitis, visual impairment, photophobia, eye pain, dry eye syndrome, asthenopia, blepharospasm, itching in the eye, eye discharge, crusting on the edges of the eyelids, eye irritation, conjunctival disorders, conjunctival edema, eye hyperemia.
Rarely: cataracts, decreased sensitivity of the cornea, redness of the eyelids.
Nervous system disorders
Often: headache.
Rarely: fainting.
Frequency unknown: dizziness.
Heart disorders
Uncommon: bradycardia, tachycardia.
Frequency unknown: arrhythmia.
Vascular disorders
Rarely: hypotension.
Disorders of the respiratory system, thoracic organs and mediastinum
Uncommon: bronchospasm, shortness of breath, rhinitis.
Rarely: cough, rhinorrhea.
Gastrointestinal disorders
Uncommon: nausea.
Rarely: dysgeusia.
Skin and subcutaneous tissue disorders
Rarely: dermatitis, rash, alopecia.
Reproductive system and breast disorders
Rarely: decreased libido.
Mental disorders
Rarely: anxiety, insomnia, depression.
General disorders and reactions at the injection site
Frequency unknown: asthenia.
Immune system disorders
With unknown frequency: hypersensitivity.
Other undesirable reactions have been observed that can develop with the use of local beta-blockers (there is no information on the frequency of development):
Immune system disorders: systemic allergic reactions, including angioedema, urticaria, local and generalized rash, itching, anaphylactic reactions.
Metabolic and nutritional disorders: hypoglycemia.
Mental disorders: nightmares, memory loss, hallucinations, psychosis, confusion.
Nervous system disorders: cerebrovascular disorders, cerebral ischemia, worsening signs and symptoms of myasthenia gravis, paresthesia.
Violations of the organ of vision: detachment of the choroid after fistulizing antiglaucoma operations, corneal erosion, ptosis, diplopia.
Cardiac disorders: chest pain, palpitations, edema, chronic heart failure, atrioventricular block, cardiac arrest, heart failure, slow atrioventricular conduction or worsening block.
Vascular disorders: Raynaud’s phenomenon, coldness and cyanosis of the hands and feet, aggravation of existing intermittent claudication.
Disorders of the respiratory system, chest and mediastinal organs: bronchospasm (mainly in patients with a history of bronchospastic diseases).
Gastrointestinal disorders: dyspepsia, diarrhea, dry mouth, abdominal pain, vomiting.
Skin and subcutaneous tissue disorders: psoriasiform rash or exacerbation of psoriasis.
Disorders of the musculoskeletal system: myalgia.
Disorders of the genital organs and breast: sexual dysfunction, impotence.
General disorders and reactions at the injection site: fatigue.
Interaction
Interaction
No specific studies have been conducted on the interaction of betaxolol with other drugs.
In patients receiving betaxolol and concomitantly taking other beta-blockers orally, the risk of adverse reactions (both systemic and local) may be higher due to possible additive effects. Such patients should be under close medical supervision.
When beta-blockers for local ophthalmic use are co-administered with oral forms of calcium channel blockers, beta-blockers, antiarrhythmic drugs (including amiodarone), cardiac glycosides, parasympathomimetics, guanethidine and drugs that deplete catecholamines, such as reserpine, an increase in effects such as decreased blood pressure and severe bradycardia. There have been cases of the development of mydriasis with the simultaneous use of beta-blockers and epinephrine.
With the simultaneous administration of betaxolol with muscle relaxants and hypoglycemic agents, an increase in the effect of the latter may be observed.
Beta blockers may reduce the effect of epinephrine used to relieve anaphylactic reactions. Should be used with extreme caution in patients with a history of atopy or anaphylaxis.
When used together with sympathomimetics, their vasoconstrictor effect is enhanced.
Caution should be exercised when using betaxolol and adrenergic psychotropic drugs together due to the possible enhancement of their effect.
If necessary, it can be used in combination with other local ophthalmic drugs, in which case the interval between their use should be at least 10 minutes.
Overdose
Overdose
If the contents of the vial are accidentally ingested, symptoms of beta blockade may develop, including bradycardia, hypotension, acute heart failure and bronchospasm.
Treatment of overdose symptoms should be symptomatic and supportive.
If an excess amount of the drug is applied topically, it is necessary to rinse the eyes with warm water.
Manufacturer
Manufacturer
Grotex LLC, Russia
Additional information
| Manufacturer | Grotex Ltd, Russia |
|---|---|
| Medication form | eye drops |
| Brand | Grotex Ltd |
Related products
Buy Betaxolol-Solofarm, eye drops 0.5% 5 ml with delivery to USA, UK, Europe and over 120 other countries.