No products in the cart.
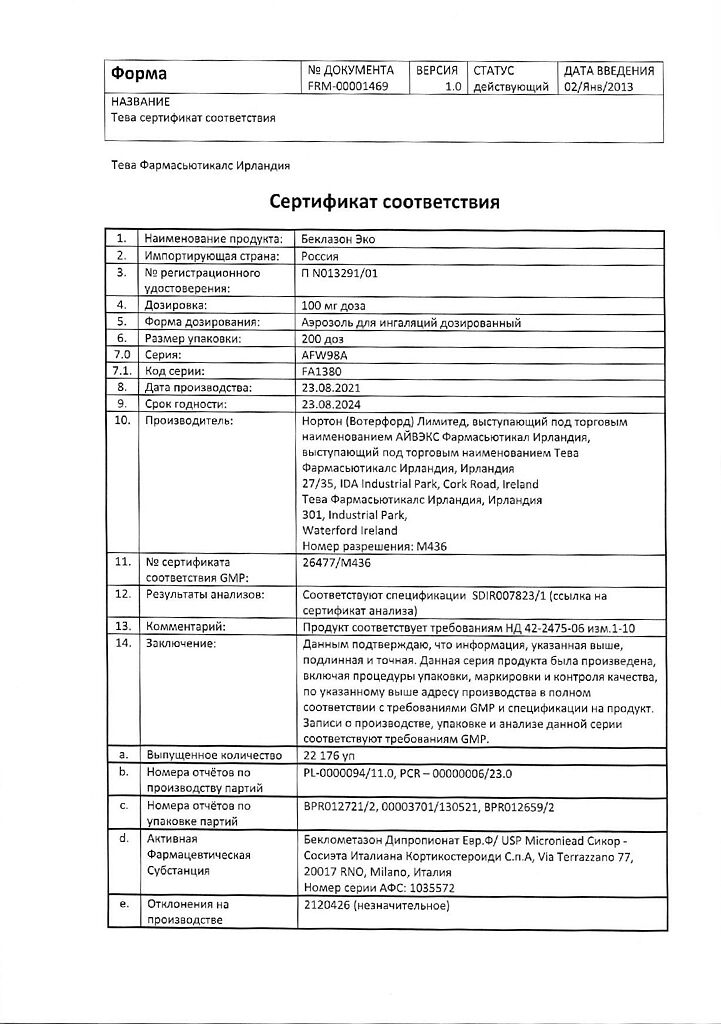
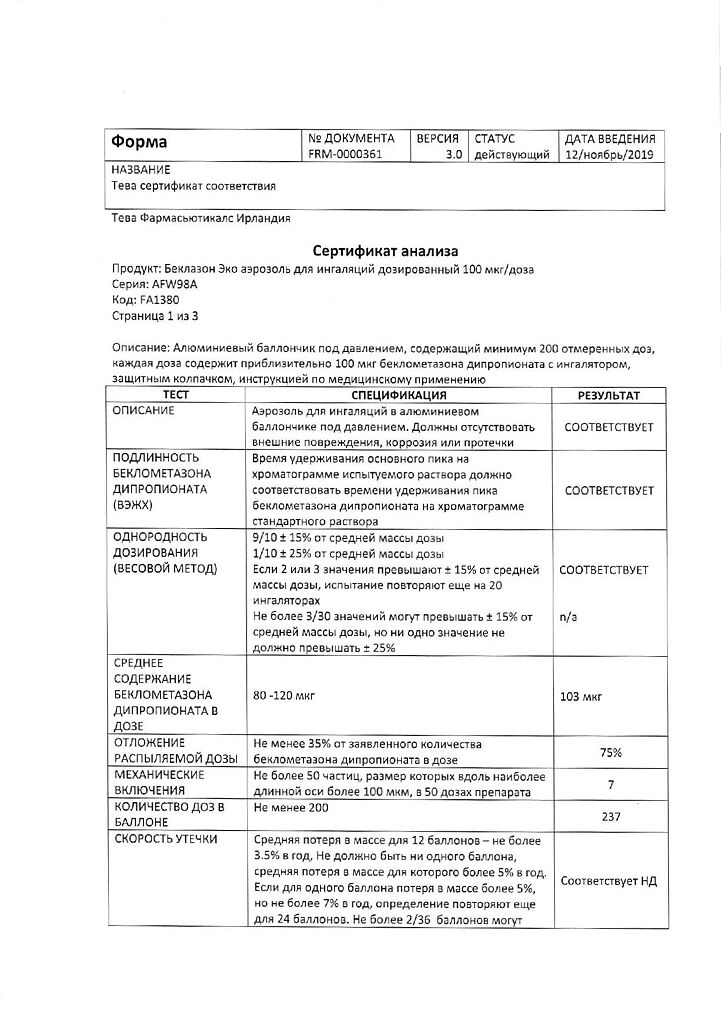
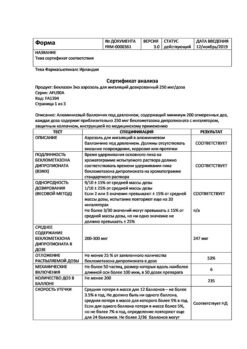
Beclazon Eco, aerosol 100 mcg/dose 200 doses
€9.67 €8.06
Description
Pharmacotherapeutic group: glucocorticosteroid for topical use
ATX code: R03VA01
Pharmacological properties
Pharmacodynamics. Beclomethasone is a glucocorticosteroid (GCS) and has weak tropism to GCS receptors. Under the action of enzymes it is converted into the active metabolite – beclomethasone-17-monopropionate (B-17-MP), which has a pronounced local anti-inflammatory effect. It reduces inflammation by reducing the formation of chemotaxis substance (effect on “late” allergy reactions), inhibits the development of an “immediate” allergic reaction (due to inhibition of production of arachidonic acid metabolites and reducing the release of inflammatory mediators from mast cells) and improves mucociliary transport. Beclomethasone reduces the number of mast cells in the bronchial mucosa, decreases epithelial edema, mucus secretion by bronchial glands, bronchial hyperresponsiveness, marginal accumulation of neutrophils, inflammatory exudate and lymphokine production, inhibits macrophage migration, reduces the intensity of infiltration and granulation. It increases the number of active beta-adrenoreceptors, restores the patient’s response to bronchodilators, allows reducing the frequency of their use. It has practically no resorptive effect after inhalation administration.
It does not relieve bronchospasm; the therapeutic effect develops gradually, usually after 5-7 days of course use of beclomethasone.
Pharmacokinetics. More than 25% of the dose of inhaled beclomethasone is deposited in the respiratory tract; the remaining amount is deposited in the mouth, pharynx, and swallowed. In the lungs, beclomethasone is extensively metabolized to the active metabolite B-17-MP prior to absorption. Systemic absorption of B-17-MP occurs in the lungs (36% of the pulmonary fraction) and in the gastrointestinal tract (26% of the ingested dose). The absolute bioavailability of unchanged beclomethasone and B-17-MP is approximately 2% and 62% of the inhaled dose, respectively. Beclomethasone is rapidly absorbed; time to reach maximum plasma concentration (Tmax) is 0.3 h. B-17-MP is more slowly absorbed, with a Tmax of 1 h. There is an approximately linear relationship between increasing inhaled dose and systemic exposure to beclomethasone.
Tissue distribution is 20 L for beclomethasone and 424 L for B-17-MP. The binding to plasma proteins is relatively high at 87%.
Beclomethasone and B-17-MP have high plasma clearance (150 l/h and 120 l/h, respectively). The elimination half-life is 0.5 h and 2.7 h, respectively.
Indications
Indications
Basic therapy for various forms of bronchial asthma in adults and children.
Pharmacological effect
Pharmacological effect
Pharmacotherapeutic group: glucocorticosteroid for local use
ATX code: R03BA01
Pharmacological properties
Pharmacodynamics
Special instructions
Special instructions
Before using Beclazon Eco, you must ensure that the patient is using the inhalation device correctly to ensure sufficient dosing of the drug.
Patients using the drug Beclazon Eco at home should be warned that if the effect of the usual dose becomes less effective or shorter lasting, they should not independently increase the dose or frequency of use of the drug, but should immediately consult a doctor.
With long-term use of beclomethasone in high doses (more than 400 mcg/day), candidiasis of the oral cavity and pharynx develops, especially in patients who have previously had a fungal infection, which is confirmed by the high level of precipitating antibodies against Candida fungi in the blood. As a rule, the use of antifungal drugs helps to quickly eliminate the fungal infection. The dose of beclomethasone in this case should not be changed.
If treatment with inhaled beclomethasone begins while taking oral corticosteroids, then reducing the dose of corticosteroids can begin only 1-2 weeks after the start of simultaneous use. The dose reduction regimen for oral corticosteroids depends on the dose and duration of corticosteroid use. Regular use of inhaled beclomethasone in most cases allows you to reduce the dose of GCS for oral administration. As a rule, patients taking no more than 15 mg of prednisolone can completely switch to taking inhaled beclomethasone only. The first months after switching to inhaled therapy with beclomethasone, it is necessary to carefully monitor the state of the HPA axis to prevent its suppression.
Patients with reduced adrenal function who are completely transferred to treatment with inhaled beclomethasone should always have a supply of GCS with them and carry a warning card with information that in stressful situations they need to use systemic GCS. After the end of the stressful situation, the dose of GCS can be reduced or GCS can be discontinued.
An increase in the dose of GCS is required in case of sudden and progressive worsening of bronchial asthma. An indirect indicator of the ineffectiveness of therapy is the more frequent use of short-acting beta2-agonists.
When transferring patients from oral corticosteroids to inhaled corticosteroids, including beclomethasone, various allergic reactions may occur, including allergic rhinitis and allergic dermatitis, which did not appear during treatment with systemic corticosteroids.
Beclomethasone for inhalation is intended for regular daily use and not for relieving bronchospasm attacks. To relieve bronchospasm attacks, beta2-adronomimetics, including salbutamol, are used. In case of severe bronchial asthma or insufficient effectiveness of inhaled beclomethasone, it is necessary to increase its dose, and also consider the use of corticosteroids orally or, for example, the use of antibiotics in case of infectious inflammation.
If paradoxical bronchospasm develops, beclomethasone should be discontinued, the patient examined and the possibility of using another drug considered.
Treatment with inhaled beclomethasone should not be abruptly stopped.
With long-term use of inhaled beclomethasone at a dose of more than 1.5 g / day, the development of systemic reactions of various types, including symptoms of suppression of adrenal function, decreased growth rate in children, decreased bone mineral density, cataracts, glaucoma, Cushing’s syndrome, characteristic signs of Cushingoid, psychological or behavioral effects, including psychomotor hyperactivity, sleep disturbances, anxiety, depression or aggression (especially in children). Therefore, when a therapeutic effect is achieved, the dose of inhaled beclomethasone should be reduced to the minimum effective dose that controls the course of the disease. Patients at high risk of developing adrenal insufficiency should be under medical supervision.
With long-term use of beclomethasone in children, it is necessary to monitor the dynamics of their growth.
Caution should be exercised when using inhaled corticosteroids in patients with active and inactive forms of pulmonary tuberculosis.
Visual disturbances have been reported with systemic and local use of glucocorticosteroids. If a patient experiences symptoms such as blurred vision or other visual disturbances, consider referring the patient to an ophthalmologist to evaluate possible causes, which may include cataracts, glaucoma, or rare diseases such as central serous chorioretinopathy (CSC).
It is necessary to protect your eyes from contact with the drug Beclazon Eco.
The aluminum container with the drug Beclazon Eco cannot be pierced, disassembled or heated, even if it is empty.
Impact on the ability to drive vehicles and machinery
Inhalations of the drug Beclazon Eco do not affect the ability to drive vehicles and machinery.
Active ingredient
Active ingredient
Beclomethasone
Composition
Composition
1 dose of the drug contains
active ingredient: beclomethasone dipropionate 50.00 mcg/100.00 mcg/250.00 mcg;
excipients: hydrofluoroalkane (HFA-134a) 75.86 mg/74.79 mg/71.75 mg, ethanol 2.09 mg/3.11 mg/6.00 mg
Pregnancy
Pregnancy
Beclazon Eco should be used with extreme caution during pregnancy and breastfeeding, only if the expected benefit to the mother outweighs the possible risk to the fetus and infant.
Contraindications
Contraindications
Hypersensitivity to beclomethasone and other components of the drug; children up to 4 years old.
With caution
For glaucoma, systemic infections (bacterial, viral, fungal, parasitic), osteoporosis, pulmonary tuberculosis, liver cirrhosis, hypothyroidism, pregnancy, during breastfeeding, in patients with reduced immunity. See also section “Special instructions”.
Side Effects
Side Effects
The incidence of side effects is classified according to the recommendations of the World Health Organization: very often (≥1/10); often (≥1/100, 1/1000, <1/100); rare (≥1/10,000, <1/1000); very rare (< 1/10,000), frequency unknown (cannot be determined from available data).
Infectious and parasitic diseases: very often – laryngitis, pharyngitis; often – candidiasis of the oral cavity and upper respiratory tract and pharynx (with long-term use in high doses of more than 400 mcg per day); pneumonia (in patients with COPD).
Immune system disorders: rarely – allergic reactions: angioedema of the eyes, throat, lips and face.
Endocrine system disorders: very rarely – suppression of the hypothalamic-pituitary-adrenal system (HPA) (with long-term use of high doses of more than 1.5 g / day), including growth retardation in children and adolescents.
Mental disorders: frequency unknown – psychomotor hyperactivity, sleep disorders, anxiety, depression, aggression, behavior changes (mainly in children).
Nervous system disorders: uncommon – headache.
Violations of the organ of vision: infrequently – blurred vision (see section “Special instructions”); very rarely – cataract, glaucoma (systemic effect); frequency unknown – central serous retinopathy.
Disorders of the respiratory system, chest and mediastinal organs: often – hoarseness of voice, irritation of the mucous membrane of the throat (the use of a spacer reduces the likelihood of developing these side effects); rarely – paradoxical bronchospasm (treated with inhaled short-acting beta2-adrenergic agonists); very rarely – eosinophilic pneumonia.
Disorders of the skin and subcutaneous tissues: very rarely – urticaria, rash, itching, redness.
Musculoskeletal and connective tissue disorders: very rarely – decreased bone mineral density (systemic effect).
With inhaled treatment of GCS (including beclomethasone dipropionate), other systemic effects may occur, primarily with long-term treatment with high doses. The likelihood of such effects occurring is significantly less than with the treatment of oral corticosteroids. Headache, nausea, bruising or thinning of the skin.
Interaction
Interaction
Beclomethasone restores the patient’s response to beta-agonists, allowing them to reduce the frequency of their use, and, when used simultaneously, enhances their effect.
When used simultaneously with phenobarbital, phenytoin, rifampicin and other inducers of microsomal liver enzymes, the effectiveness of beclomethasone is reduced.
When used simultaneously with methandienone, estrogens,
beta2-adrenergic agonists, theophylline, and systemic corticosteroids increase the effectiveness of beclomethasone.
Concomitant use with oral corticosteroids or potassium-sparing diuretics such as thiazides or furosemide may cause excessive potassium loss.
Overdose
Overdose
An acute overdose of the drug can occur with inhalation of a high single dose of more than 1 g. The manifestation, in this case, of symptoms of suppression of the function of the adrenal cortex does not require emergency treatment, since the function is restored within several days, which is confirmed by the level of cortisol in the plasma.
In case of chronic overdose (long-term treatment with a dose of more than 1.5 g), persistent suppression of the function of the adrenal cortex may be observed. In such cases, it is recommended to monitor the reserve function of the adrenal cortex. In case of overdose, treatment with beclomethasone can be continued in doses sufficient to maintain the therapeutic effect.
Storage conditions
Storage conditions
Store at a temperature not exceeding 25°C, protecting from direct sunlight. Do not freeze.
Keep out of the reach of children.
Shelf life
Shelf life
3 years.
Do not use after the expiration date stated on the package.
Manufacturer
Manufacturer
Norton (Waterford) Limited/IVEX Pharmaceuticals I, Ireland
Additional information
| Shelf life | 3 years. Do not use after the expiration date stated on the package. |
|---|---|
| Conditions of storage | Store at a temperature not exceeding 25 ° C, protecting it from direct sunlight. Do not freeze. Keep out of reach of children. |
| Manufacturer | Norton (Waterford) Limited/IWAX Pharmaceuticals E, Ireland |
| Medication form | metered aerosol for inhalation |
| Brand | Norton (Waterford) Limited/IWAX Pharmaceuticals E |
Other forms…
Related products
Buy Beclazon Eco, aerosol 100 mcg/dose 200 doses with delivery to USA, UK, Europe and over 120 other countries.