No products in the cart.
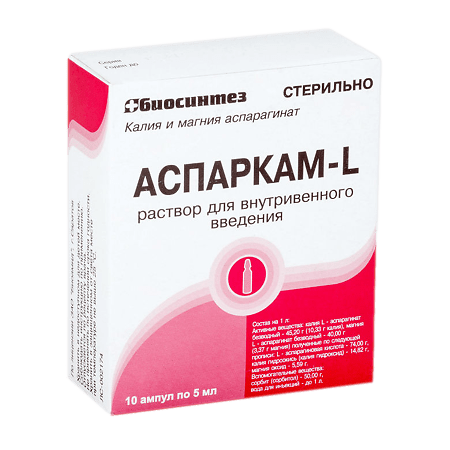
Asparkam-L, 45.2mg/ml+40 mg/ml 5 ml 10 pcs
€3.08 €2.00
Out of stock
(E-mail when Stock is available)
Description
The mechanism of action of Asparkam is related to effective participation of L-asparaginate in transport of magnesium and potassium ions into intracellular space.Magnesium activates sodium-potassium-ATPase, removing sodium ions from cell and returning potassium ions; reduces sodium concentration and prevents its exchange for calcium in vascular smooth muscles, reducing their resistance. Potassium stimulates the synthesis of ATP, glycogen, proteins, acetylcholine, etc.
The two ions support the polarization of cell membranes.
The drug regulates the conduction of impulses along the nerve fibers, synaptic transmission, muscle contraction, and the work of the heart muscle. Due to magnesium ions the product participates in the processes of energy input and output, normalizes membrane permeability, neuromuscular conduction, DNA and RNA synthesis, cell growth, cell division, oxygen assimilation and phosphate synthesis.
Indications
Indications
Heart palpitations, Heart pain (cardialgia), Strengthening the cardiovascular system, Tachycardia
- To eliminate potassium and magnesium deficiency;
- .As an adjuvant in various manifestations of coronary heart disease, including acute myocardial infarction;
- Chronic circulatory failure;
- arrhythmias of the heart (arrhythmias caused by overdose of cardiac glycosides).
.
Active ingredient
Active ingredient
Potassium asparaginate, Magnesium asparaginate
Composition
Composition
1 ml of solution for intravenous administration contains:
acting substances:
Magnesium asparaginate anhydrous 40 mg (magnesium content – 3.37 mg),
Potassium asparaginate anhydrous 45.2 mg (potassium content 10.33 mg);
excipients:
sorbitol (sorbitol),
water for injection.
How to take, the dosage
How to take, the dosage
The drug is administered intravenously (by drip or stream). The dose of the drug is determined individually depending on the indications for use.
Intravenously by drop infusion at a rate of 25 drops per 1 minute 1 to 2 times a day of 10-20 ml (1-2 ampoules of 10 ml or 2-4 ampoules of 5 ml) diluted in 100-200 ml of sodium chloride isotonic solution or 5% glucose solution.
In case of jet injection the contents of 1 ampoule of 10 ml or 2 ampoules of 5 ml are diluted in 20 ml of sterile water for injection or 5% glucose solution and injected slowly (not more than 5 ml per minute) into a vein.
Interaction
Interaction
Potassium-saving diuretics (triamterene, spironolactone), beta-adrenoblockers, cyclosporine, heparin, angiotensin-converting enzyme inhibitors, nonsteroidal anti-inflammatory drugs – risk of hyperkalemia up to the development of arrhythmia and asystole.
Manages hypokalemia caused by glucocorticosteroids.
Reduces unwanted effects of cardiac glycosides.
Due to the content of magnesium ions it reduces the effect of neomycin, polymyxin B, tetracycline and streptomycin.
Anesthetics increase the suppressive effect of magnesium on the central nervous system.
May increase neuromuscular blockade caused by depolarizing myorelaxants (atracurium besylate, decamethonium bromide, suxamethonium (chloride, bromide, iodide).
Calcitriol increases the concentration of magnesium in the blood plasma, calcium preparations reduce the effect of magnesium.
The solution is pharmaceutically compatible with solutions of cardiac glycosides (improves their tolerability).
Special Instructions
Special Instructions
There have been no studies to evaluate the effect of the drug on the ability to drive vehicles and engage in other potentially dangerous activities requiring increased concentration and quick psychomotor reactions.
Contraindications
Contraindications
- High sensitivity to Asparkam,
- hyperkalemia,
- hypermagnesemia,
- acute and chronic renal insufficiency,
- insufficiency of the adrenal cortex,
- trioventricular and cholerentricular block of the II and III degree,
- hypermagnesemia./li>
- adrenal insufficiency,
- atrioventricular block of degree II and III,
- cardiogenic shock,
- oliguria – anuria,
- acute metabolic acidosis,
- severe myasthenia gravis,
- dehydration, Addison’s disease,
- pregnancy,
- lactation period,
- age below 18 years (efficacy and safety not established).
.
Side effects
Side effects
- Possible nausea, dizziness,
- red face,
- thirst,
- decreased blood pressure, hyporeflexia.
- hyperkalemia (nausea, vomiting, diarrhea, paresthesia) and hypermagnesemia (facial hyperemia, thirst, bradycardia, decreased blood pressure, muscle weakness, fatigue, paresis, coma, areflexia, respiratory depression, seizures);
- in parenteral administration – atrioventricular block, paradoxical reaction (increased number of extrasystoles), phlebitis.
- All symptoms of hyperkalemia and hypermagnesemia may appear during rapid intravenous administration.
Overdose
Overdose
Symptoms: hyperkalemia and hypermagnesemia.
Treatment: intravenous calcium chloride; if necessary, hemodialysis and peritoneal dialysis.
Similarities
Similarities
Panangin, Asparkam, Potassium and Magnesium Asparaginate, Asparkam-L
Additional information
| Weight | 0.116 kg |
|---|---|
| Shelf life | 2 years |
| Conditions of storage | In a light-protected place at 0-25 °C |
| Manufacturer | Biosintez, Russia |
| Medication form | solution for infusion |
| Brand | Biosintez |
Related products
Buy Asparkam-L, 45.2mg/ml+40 mg/ml 5 ml 10 pcs with delivery to USA, UK, Europe and over 120 other countries.