No products in the cart.
Almont, 4 mg 98 pcs.
€1.00
Out of stock
(E-mail when Stock is available)
Description
Bronchial asthma, Bronchospasm, Runny nose (rhinitis)
Prevention and long-term treatment of bronchial asthma in children, including:
- preventing daytime and nighttime symptoms of the disease (for children 2 years and older);
- Treatment of bronchial asthma in patients with hypersensitivity to acetylsalicylic acid (for children 6 years and older);
- preventing exercise-induced bronchospasm (for children 2 years and older).
Pease the symptoms of seasonal and year-round allergic rhinitis in children from 2 years of age.
Indications
Indications
Prevention and long-term treatment of bronchial asthma in children, including:
prevention of daytime and nighttime symptoms of the disease (for children aged 2 years and older);
treatment of bronchial asthma in patients with hypersensitivity to acetylsalicylic acid (for children aged 6 years and older);
prevention of bronchospasm caused by physical activity (for children aged 2 years and older).
Relief of symptoms of seasonal and year-round allergic rhinitis in children from 2 years of age.
Pharmacological effect
Pharmacological effect
Pharmacotherapeutic group:
Special instructions
Special instructions
The drug Almont is not recommended for the treatment of acute attacks of bronchial asthma. Patients with bronchial asthma are advised to always carry emergency medications with them. When an acute attack occurs, short-acting inhaled betag-adrenergic agonists should be used. Patients should consult their physician as soon as possible if they require more inhaled short-acting betag-agonists than usual.
Active ingredient
Active ingredient
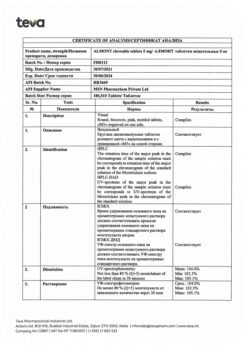
Montelukast
Composition
Composition
1 chewable tablet 4 mg contains:
active substance:
montelukast 4.00 mg (as montelukast sodium 4.16 mg);
excipients:
mannitol 160.96 mg,
microcrystalline cellulose 52.80 mg,
hyprolose 7.20 mg,
croscarmellose sodium 7.20 mg,
dye Pigment Blend PB-24880 (lactose monohydrate 3.60 mg, red iron oxide dye 0.40 mg) 4.00 mg,
magnesium stearate 2.40 mg,
aspartame 1.20 mg,
cherry flavor (Silarom Cherry Flavor 1219813182) 0.08 mg.
Contraindications
Contraindications
Hypersensitivity to the active or any excipient of the drug;
children up to 2 years of age (for a dosage of 4 mg) and up to 6 years of age (for a dosage of 5 mg);
patients with rare hereditary diseases: galactose intolerance, lactase deficiency or glucose-galactose malabsorption;
phenylketonuria (contains aspartame).
Side Effects
Side Effects
Infectious and parasitic diseases: upper respiratory tract infections.
Interaction
Interaction
In patients simultaneously receiving phenobarbital, the area under the pharmacokinetic concentration-time curve of montelukast decreased by approximately 40%, but no dosage adjustment is required in such patients. Since montelukast is metabolized by CYP3A4, caution should be exercised, especially in children, if montelukast is co-administered with CYP3A4 inducers such as phenytoin, phenobarbital and rifampicin.
Overdose
Overdose
Symptoms of drug overdose in patients with chronic bronchial asthma when used at a dose exceeding 200 mg per day for 22 weeks and at a dose of 900 mg per day for 1 week have not been identified.
Shelf life
Shelf life
3 years.
Manufacturer
Manufacturer
Iceland
Additional information
| Shelf life | 3 years. |
|---|---|
| Manufacturer | Iceland |
| Medication form | chewable tablets |
Other forms…
Related products
Buy Almont, 4 mg 98 pcs. with delivery to USA, UK, Europe and over 120 other countries.