No products in the cart.
Actrapid NM Penfill, 100 me/ml 3 ml 5 pcs
€25.67 €21.39
Description
Actrapid® NM is a short-acting insulin preparation produced by recombinant DNA biotechnology using Saccharomyces cerevisiae strain.
Low blood glucose levels are reduced by increasing its intracellular transport after insulin binds to insulin receptors of muscle and adipose tissue and simultaneously reducing the rate of glucose production by the liver.
The normalization of plasma glucose concentration (up to 4.4-6.1 mmol/l) during intravenous administration of Actrapid® NM in intensive care unit patients who underwent serious surgical interventions (204 patients with diabetes and 1344 patients without diabetes) who had hyperglycemia (plasma glucose concentration > 10 mmol/L), reduced mortality by 42% (4.6% instead of 8%).
The action of Actrapid® NM begins within half an hour after administration and the maximum effect is seen within 1.5-3.5 hours, with a total duration of action of about 7-8 hours.
Indications
Indications
Type 1 diabetes
- Diabetes mellitus.
- Incomplicated conditions in diabetic patients with impaired glycemic control.
Composition
Composition
In 1 ml of the drug contains:
Active ingredient:
Soluble insulin (human genetically engineered) 100 ME (3.5 mg);
1 ME corresponds to 0.035 mg of anhydrous human insulin;
Excipients:
Zinc 7 µg (in the form of zinc chloride),
Glycerol (glycerol) 16 mg,
Methacresol 3.0 mg,
sodium hydroxide about 2.6 mg and/or hydrochloric acid about 1.7 mg (for pH adjustment),
water for injection up to 1.0 ml.
How to take, the dosage
How to take, the dosage
The drug is intended for subcutaneous and intravenous administration.
The dose of the drug is adjusted individually, according to the patient’s needs.
In general, insulin requirements are 0.3 to 1 IU/kg/day. Daily insulin requirements may be higher in patients with insulin resistance (e.g., during puberty and in obese patients) and lower in patients with residual endogenous insulin production.
The drug is given 30 minutes before a meal or snack containing carbohydrates.
Actrapid® NM is a short-acting insulin and may be used in combination with long-acting insulins.
Actrapide® NM is usually injected subcutaneously, into the anterior abdominal wall. If it is convenient, injections may also be given in the thigh, gluteal, or deltoid region of the upper arm. When injected in the anterior abdominal wall area, a more rapid absorption is achieved than when injected in other areas.
If an injection is made into a retracted skin fold, the risk of accidental intramuscular injection is minimized. The needle must stay under the skin for at least 6 seconds to ensure that the dose is fully injected. The injection sites within the anatomical area should be changed constantly to reduce the risk of lipodystrophy.
Intramuscular injections are also possible, but only by prescription.
Actrapid® NM is also given intravenously and this can only be done by a healthcare professional.
Intravenous administration of Actrapid® NM Penfill® from a cartridge is only permitted in exceptional circumstances when vials are not available. In this case, the Actrapid® NM must be drawn into an insulin syringe without air collection or infusion using an infusion system. This procedure should only be performed by a physician.
The Actrapid® NM Penfill® is designed for use with Novo Nordisk insulin injection systems and NovoFine® or NovoTwist® needles.
The detailed recommendations for use and administration of the product should be followed (see “Instructions for Use of Actrapid® NM Penfill® to be given to the patient”).
Dose adjustment
Companion diseases, especially those with infection and fever, usually increase the body’s need for insulin.
The dose of the drug may also need to be adjusted if the patient has underlying kidney, liver, adrenal, pituitary, or thyroid disorders.
A dose adjustment may also be necessary if there is a change in physical activity or the patient’s usual diet.
Dose adjustments may be needed when changing from one type of insulin to another.
Interaction
Interaction
There are a number of medications that affect the need for insulin. Hypoglycemic effect of insulin is enhanced by oral hypoglycemic drugs, monoamine oxidase inhibitors, angiotensin-converting enzyme inhibitors, carboenhydrase inhibitors, non-selective beta-adrenoblockers, bromocriptine, sulfonamides, anabolic steroids, tetracyclines, clofibrate, ketoconazole, mebendazole, pyridoxine, theophylline, cyclophosphamide, phenfluramine, lithium preparations, salicylates.
The hypoglycemic effect of insulin is weakened by oral contraceptives, glucocorticosteroids, thyroid hormones, thiazide diuretics, heparin, tricyclic antidepressants, sympathomimetics, growth hormone (somatropin), danazol, clonidine, slow calcium channel blockers, diazoxide, morphine, phenytoin, nicotine.
Beta-adrenoblockers may mask symptoms of hypoglycemia and make recovery from hypoglycemia difficult. Octreotide/lanreotide can both increase and decrease the body’s need for insulin.
Alcohol can increase or decrease the hypoglycemic effect of insulin.
Incompatibilities
Actrapide NM can only be added to compounds with which it is known to be compatible. Some drugs (such as those containing thiols or sulfites) can cause degradation of insulin when added to the insulin solution.
Special Instructions
Special Instructions
Inadequate dosage of the drug or discontinuation of treatment, especially in type 1 diabetes, can lead to the development of hyperglycemia.Typically, the first symptoms of hyperglycemia appear gradually, over a period of hours or days. Symptoms of hyperglycemia include a feeling of thirst, frequent urination, nausea, vomiting, drowsiness, red and dry skin, dry mouth, loss of appetite, and the smell of acetone in the exhaled air. Without appropriate treatment, hyperglycemia in patients with type 1 diabetes can lead to diabetic ketoacidosis, a condition that is potentially fatal.
Hypoglycemia
Hypoglycemia can occur if too high a dose of insulin is given relative to the patient’s need.
Missing a meal or unplanned strenuous exercise can lead to hypoglycemia.
After compensation of carbohydrate metabolism, such as with intensified insulin therapy, patients’ typical hypoglycemic precursor symptoms may change and patients should be informed. Common precursor symptoms may disappear with the long-term course of diabetes mellitus.
The transfer of patients to a different type of insulin or to insulin from a different manufacturer should only be done under medical supervision. Changes in concentration, manufacturer, type, type (human insulin, human insulin analogues) and/or method of manufacture may require a change in insulin dose. Patients switching to Actrapid HM may need to change the dose or increase the frequency of injections compared to previous insulin preparations. If a dose adjustment is necessary when transferring patients to treatment with Actrapid NM, this may be done at the time of the first dose or during the first weeks or months of therapy.
As with other insulin drugs, reactions may develop at the injection site, manifested by pain, redness, urticaria, inflammation, bruising, swelling and itching. Regularly changing the injection site in the same anatomical area can help reduce symptoms or prevent the development of these reactions. Reactions usually disappear within a few days to a few weeks. In rare cases, Actrapid NM may need to be stopped because of reactions at the injection sites. Patients should consult with their physician before traveling due to a time zone change, because a time zone change means that the patient must eat and inject insulin at a different time.
When Actrapid NM is added to infusion solutions, the amount of insulin absorbed by the infusion system is unpredictable, so the use of Actrapid NM in PPIs is not permitted.
The concomitant use of thiazolidinedione and insulin drugs
Cases of chronic heart failure have been reported when treating patients with thiazolidinedione in combination with insulin drugs, especially if these patients have risk factors for chronic heart failure. This fact should be taken into account when prescribing combination therapy with thiazolidinediones and insulin drugs. When prescribing such combination therapy, medical examination of patients should be carried out to detect signs and symptoms of chronic heart failure, weight gain and edema. If patients have worsening symptoms of heart failure, treatment with thiazolidinedione should be discontinued.
Impact on driving and operating machinery
Patients’ ability to concentrate and reaction speed may be impaired during hypoglycemia, which may be dangerous in situations where these abilities are particularly necessary (such as driving or operating machines and mechanisms). Patients should be advised to take measures to prevent hypoglycemia during driving. This is especially important for patients with the absence or reduced severity of symptom precursors of developing hypoglycemia or who suffer from frequent episodes of hypoglycemia. In these cases, consideration should be given to the appropriateness of driving and performing such activities.
Contraindications
Contraindications
- Hypersensitivity to human insulin or to any of the ingredients in this medication.
- Hypoglycemia.
Side effects
Side effects
Hypoglycemia is the most common adverse event when using insulin. During clinical studies, as well as during the use of the drug after its release to the consumer market, it was found that the frequency of hypoglycemia varies depending on the patient population, the drug dosing regime and glycemic control.At the initial stage of insulin therapy, refractive disorders, swelling and reactions in the injection site (including pain, redness, urticaria, inflammation, hematoma, swelling and itching at the injection site) may occur. These symptoms are usually temporary. Rapid improvement in glycemic control can lead to a state of “acute pain neuropathy,” which is usually reversible.
Intensification of insulin therapy with dramatic improvement in carbohydrate metabolism control can lead to a temporary worsening of diabetic retinopathy, while at the same time, long-term improvement in glycemic control reduces the risk of progression of diabetic retinopathy. A list of side effects is shown below.
All side effects presented below, based on data from clinical trials, are categorized according to frequency of development according to MedDRA and organ systems. The frequency of side effects is defined as: very common (≥1/10); common (≥1/100 to <1/10); infrequent (≥1/1000 to <1/100); rare (≥1/10000 to <1/1000), very rare (<1/10000) and unknown (cannot be estimated based on available data).
Immune system disorders: infrequent – urticaria, skin rash; very rare – anaphylactic reactions.
Metabolic and nutrition disorders: very common – hypoglycemia.
Nervous system disorders: infrequent peripheral neuropathy (“acute pain neuropathy”).
VIight organ disorders: infrequent – refractive disorders; very rare – diabetic retinopathy.
Skin and subcutaneous tissue disorders: infrequent – lipodystrophy.
General disorders and disorders at the injection site: infrequent reactions at the injection site; infrequent edema.
Description of individual adverse reactions:
Anaphylactic reactions
Very rare generalized hypersensitivity reactions (including generalized skin rash, itching, increased sweating, gastrointestinal distress, angioedema, difficulty breathing, palpitations, decreased BP, and syncope/loss of consciousness, which are potentially life-threatening.
Hypoglycemia
Hypoglycemia is the most common side effect. It can develop if the insulin dose is too high relative to the insulin requirement. Severe hypoglycemia can lead to loss of consciousness and/or seizures, temporary or permanent brain damage, or even death. Symptoms of hypoglycemia usually develop suddenly. They may include “cold sweats,” pale skin, increased fatigue, nervousness or tremors, anxiety, unusual fatigue, or weakness, disorientation, decreased concentration, sleepiness, marked hunger, visual disturbances, headache, nausea, and palpitations.
Lipodystrophy
Infrequent cases of lipodystrophy have been reported. Lipodystrophy may develop at the site of administration of the drug.
Overdose
Overdose
There is no specific dose required for an insulin overdose, but hypoglycemia can develop gradually if too high doses of insulin are administered relative to the patient’s need.
- Mild hypoglycemia can be managed by the patient by ingesting glucose or sugary foods. Therefore, it is recommended that diabetics carry sugary foods with them at all times.
- In severe hypoglycemia, when the patient is unconscious, 0.5 mg to 1 mg of glucagon intramuscularly or subcutaneously (a trained person can administer), or intravenous dextrose (glucose) solution (only a medical professional can administer). It is also necessary to administer intravenous dextrose if the patient does not regain consciousness 10-15 minutes after glucagon administration. After recovery of consciousness, the patient is advised to take carbohydrate-rich food to prevent recurrence of hypoglycemia.
.
Additional information
| Weight | 0.057 kg |
|---|---|
| Shelf life | 2.5 years |
| Conditions of storage | In the dark place at 2-8 °C (do not freeze) |
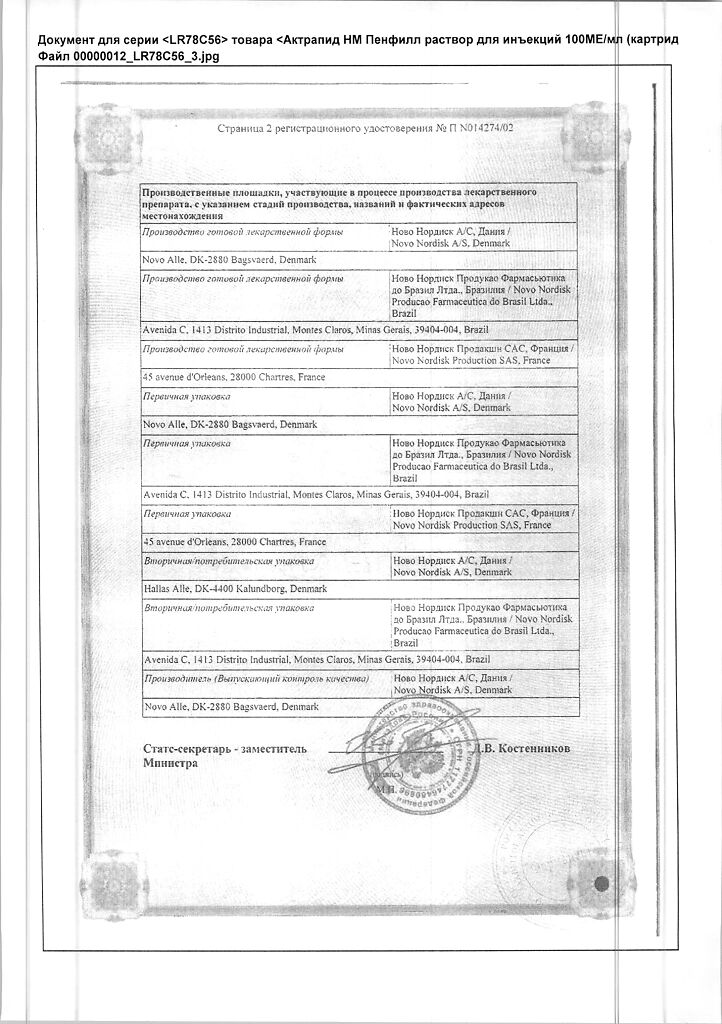
| Manufacturer | Novo Nordisk A/S, Denmark |
| Medication form | solution for injection |
| Brand | Novo Nordisk A/S |
Related products
Buy Actrapid NM Penfill, 100 me/ml 3 ml 5 pcs with delivery to USA, UK, Europe and over 120 other countries.