No products in the cart.
5-Fluorouracil-Ebeve, 50 mg/ml 20 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
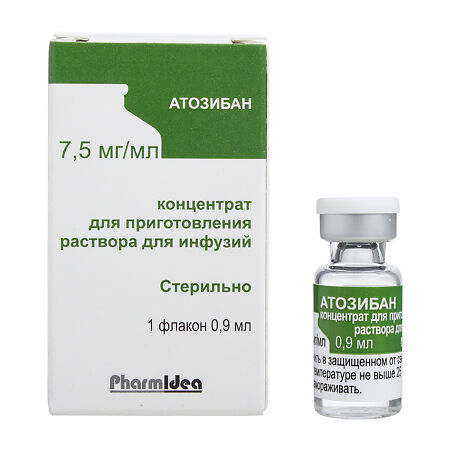
A labor-inhibiting agent, an oxytocin receptor blocker. Atosiban is a synthetic peptide which is a competitive antagonist of human oxytocin at the receptor level. Atosiban binds to oxytocin receptors, reducing the frequency of uterine contractions and myometrial tone, leading to suppression of uterine contractility. Atosiban also binds to vasopressin receptors, inhibiting the effect of vasopressin, but has no effect on the cardiovascular system.
In the case of preterm labor in a woman, atosiban inhibits uterine contractions at recommended doses and provides the uterus with functional rest. Uterine relaxation begins immediately after administering atosiban and myometrial contractile activity is significantly reduced within 10 minutes, maintaining stable functional peace of the uterus (≤4 contractions per hour) for 12 hours.
Pharmacokinetics
Indications
Indications
At risk of preterm birth in pregnant women: regular uterine contractions of at least 30 seconds duration and frequency of more than 4 within 30 minutes; cervical opening of 1 to 3 cm (0-3 cm for non-pregnant women); age over 18 years; gestational age of 24 to 33 full weeks; normal fetal heart rate.
Active ingredient
Active ingredient
Atoziban
Composition
Composition
1 ml contains:
- Active substances: atosiban – 7.5 mg,
- Additives: mannitol – 50 mg, hydrochloric acid solution 1M – to pH 4.5, water d / i – to 1 ml.
How to take, the dosage
How to take, the dosage
Injected by IV infusion immediately after diagnosis of preterm labor in three steps: 1) an initial dose of 6.75 mg is administered for 1 min first; 2) an infusion of 300 mcg/min (infusion rate 24 ml/h, atosiban dose 18 mg/h) is given immediately thereafter for 3 h; 3) an extended (up to 45 h) atosiban infusion of 100 mcg/min (infusion rate 8 ml/h, atosiban dose 6 mg/h) is followed.
The total duration of treatment should not exceed 48 h. The maximum dose of atosiban for the entire course is 330 mg.
If there is a need for repeated administration of atosiban, it should also be started with step 1, followed by infusion (steps 2 and 3). Re-application can be started at any time after the first use of the drug and can be repeated up to 3 cycles. If uterine contractility persists after 3 cycles of atosiban therapy, another drug should be considered.
Interaction
Interaction
Concomitant use of atosiban with ergot alkaloids is not advisable due to the opposite pharmacological action and indications for use.
Special Instructions
Special Instructions
If premature rupture of membranes is suspected, the benefit of using atosiban to prolong labor should be weighed against the potential risk of chorioamnionitis.
Atosiban should be used with caution in multiple pregnancies and at 24-27 weeks’ gestation (due to insufficient clinical experience).
In case of persistent uterine contractions during administration of atosiban, uterine contractions should be monitored and the fetal heart rate should be monitored. Other medications should also be considered.
There is no experience with atosiban in patients with impaired hepatic or renal function.
Atosiban is not used in cases of abnormal placental attachment.
In cases of intrauterine fetal retardation, the decision to continue or to reintroduce atosiban depends on the assessment of fetal maturity.
As an oxytocin antagonist, atosiban can theoretically promote uterine relaxation and induce postpartum uterine bleeding, so the degree of blood loss after delivery should be continually assessed.
Atosiban does not affect the ability to drive and operate machinery
Atosiban does not affect the ability to drive and operate machinery.
Contraindications
Contraindications
Gestational age less than 24 or more than 33 full weeks; premature rupture of membranes in pregnancies over 30 weeks; intrauterine fetal retardation; abnormal fetal heart rate; uterine bleeding requiring immediate delivery; eclampsia and severe pre-eclampsia requiring immediate delivery; Intrauterine fetal death; suspected intrauterine infection; placenta previa; placenta detachment; any maternal or fetal condition in which the maintenance of pregnancy is dangerous; breastfeeding; hypersensitivity to atosiban.
Side effects
Side effects
Digestive system disorders: very often (>1/10) – nausea, rarely – vomiting.
Metabolic system disorders: often (>1/100, <1/100) – hyperglycemia.
Nervous system disorders: frequently (>1/100, <1/100) – headache, dizziness; rarely (>1/1000, <1/100) – insomnia.
Cardiovascular system: often (>1/100, <1/100) – tachycardia, arterial hypotension, hot flashes.
Skin side: rare (>1/1000, <1/100) – itching, skin rash.
Social system: very rare (>1/10,000, <1/1000) – uterine bleeding, uterine atony.
Other: very rare (>1/10,000, <1/1000) – allergic reactions.
Additional information
| Weight | 0.015 kg |
|---|---|
| Manufacturer | Pharmidea Ltd., Latvia |
| Medication form | concentrate for preparation of infusion solution |
| Brand | Pharmidea Ltd. |
Related products
Gynecology and Obstetrics
Gynecology and Obstetrics
Buy 5-Fluorouracil-Ebeve, 50 mg/ml 20 ml with delivery to USA, UK, Europe and over 120 other countries.