No products in the cart.
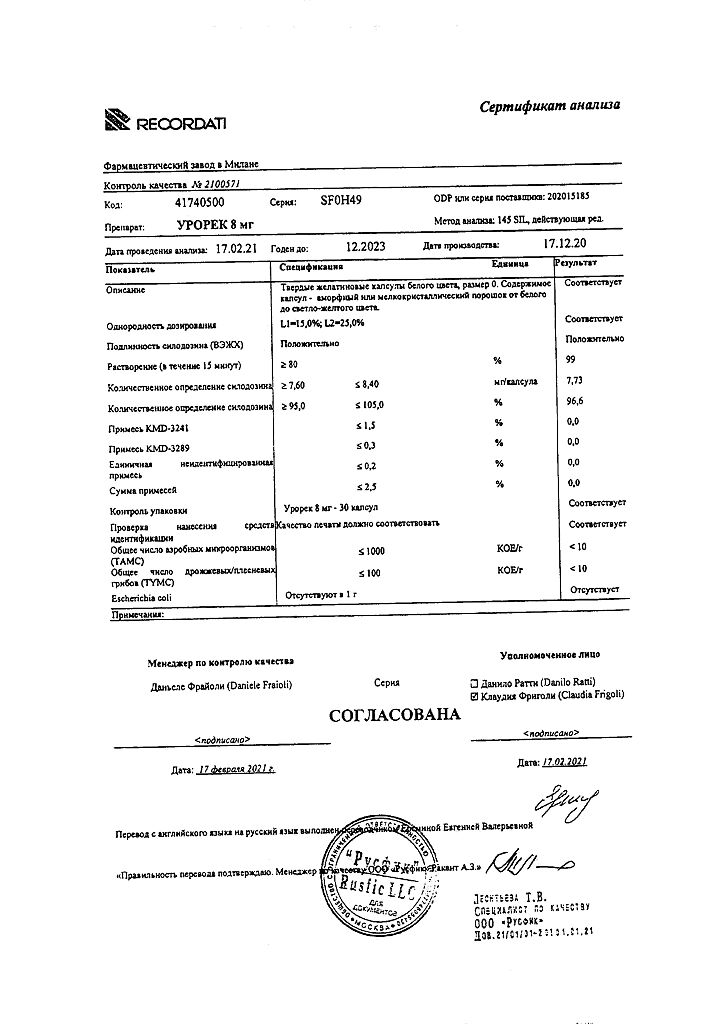
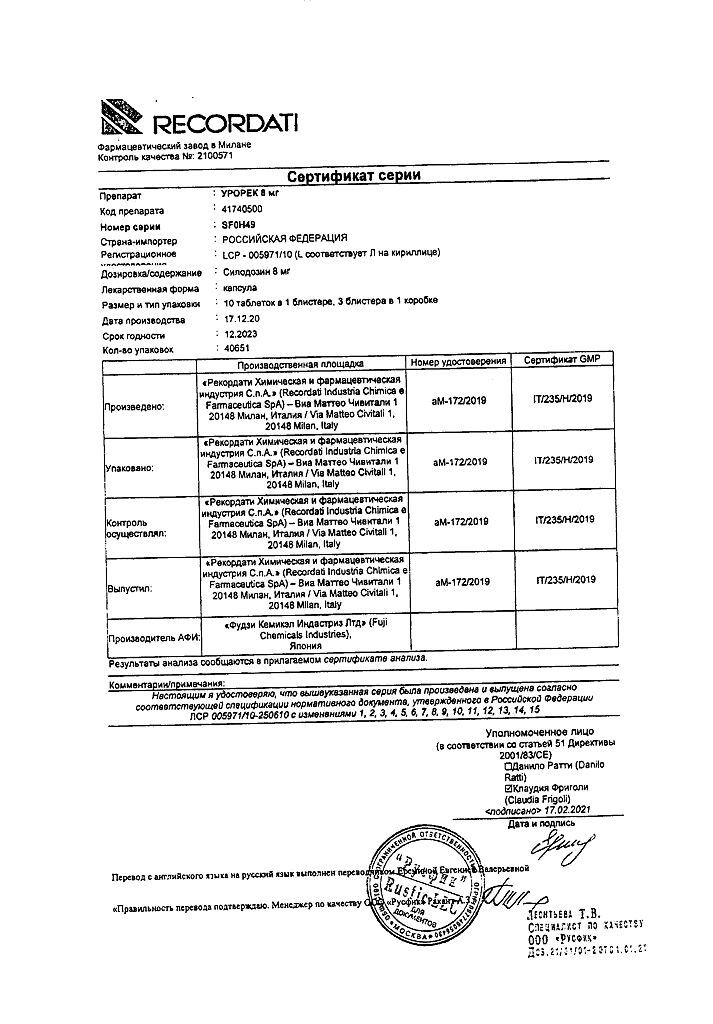
Urorek, 8 mg capsules 30 pcs
€32.50 €27.09
Description
Pharmacodynamics.
The mechanism of action
. Silodosin, a highly selective competitive antagonist of alpha 1a-adrenoreceptors, blocks postsynaptic alpha 1a-adrenoreceptors located in the smooth muscle of the prostate, bladder neck and prostatic part of the urethra.
It reduces tone of the smooth muscles of the prostate, bladder neck and the prostatic part of the urethra, improving the outflow of urine. At the same time the symptoms of obstruction and irritation associated with benign prostatic hyperplasia are reduced.
The affinity to alpha| 1a-adrenoreceptors located in the urinary bladder is 162 times higher than its ability to interact with alpha-1b-adrenoreceptors located in vascular smooth muscle. Due to high selectivity, it does not cause clinically significant reduction of arterial pressure (BP) in patients with baseline normal BP.
Pharmacokinetics
Silodosine is well absorbed when taken orally. Absolute bioavailability is 32%. Food reduces maximum concentration (Cmax) by approximately 30%, extending the time to reach maximum concentration (tmax) to approximately 1 h and has minimal effect on the area under the concentration-time curve (AUC).
Cmax87±51 mg/mL, tmax 2.5 h, urinary mean concentration 433±286 ng*h/mL. The volume of distribution of silodosine is 0.81 l/kg and is bound to plasma proteins by 96.6%. Binding of silodosine carbamoylglucuronide by plasma proteins is 91%. Silodosine is metabolized through glucuronidation (involving UGT2B7), with the participation of alcohol dehydrogenase and aldehyde dehydrogenase, oxidative pathways, mainly involving CYP3A4.
The main active metabolite in plasma is carbamoylglucuronide (K.MD-3213G), which reaches a plasma concentration 4 times higher than silodosine itself. Silodosin has no potential to induce or inhibit cytochrome P450 isoenzymes.
33.5% of silodosine is excreted through the kidneys and 54.9% through the intestine. Clearance of silodosine is about 23.1 L/h. Silodosin is excreted mainly as metabolites and in very small amounts unchanged in the urine. T1/2 of silodosine and carbamoylglucuronide is 11 hours and 18 hours, respectively.
Pharmacokinetics in different groups of patients
Elderly patients: pharmacokinetics of silodosin and metabolites were not significantly dependent on age. Clearance of silodosine did not change in patients older than 75 years. Patients with impaired liver function: In patients with moderate hepatic impairment (7-9 points on the Child-Pugh scale), the pharmacokinetics of silodosine did not change significantly. In patients with severe hepatic impairment, the pharmacokinetics of silodosin have not been studied.
Patients with impaired renal function: No dose reduction was required for patients with moderate renal impairment. For patients with severe renal impairment, administration of silodosin is not recommended.
Indications
Indications
Urorek is intended for the treatment of benign prostatic hyperplasia.
Pharmacological effect
Pharmacological effect
Pharmacodynamics.
Mechanism of action
Silodosin, a highly selective competitive alpha 1a adrenergic receptor antagonist, blocks postsynaptic alpha 1a adrenergic receptors located in the smooth muscle of the prostate, bladder neck and prostatic urethra.
Reduces the tone of the smooth muscles of the prostate gland, bladder neck and prostatic part of the urethra, improving the outflow of urine. At the same time, the symptoms of obstruction and irritation associated with benign prostatic hyperplasia are reduced.
Affinity for alpha| 1a-adrenergic receptors located in the bladder are 162 times more capable of interacting with alpha-1b-adrenergic receptors, which are located in vascular smooth muscle. Due to its high selectivity, it does not cause a clinically significant decrease in blood pressure (BP) in patients with initially normal blood pressure.
Pharmacokinetics
When taken orally, silodosin is well absorbed. Absolute bioavailability is 32%. Food reduces the maximum concentration (Cmax) by approximately 30%, prolonging the time to reach maximum concentration (tmax) to approximately 1 hour and has a minimal effect on the area under the concentration-time curve (AUC).
Cmax87±51 mg/ml, tmax – 2.5 hours, average concentration in urine 433±286 ng*h/ml. The volume of distribution of silodosin is 0.81 l/kg and is 96.6% bound to plasma proteins. The binding of silodosin carbamoyl glucuronide by plasma proteins is 91%. Silodosin is metabolized through glucuronidation (involving UGT2B7), alcohol dehydrogenase and aldehyde dehydrogenase, oxidative pathways, mainly involving CYP3A4.
The main active metabolite in plasma, carbamoyl glucuronide (K.MD-3213G), reaches plasma concentrations 4 times higher than silodosin itself. Silodosin does not have the potential to induce or inhibit cytochrome P450 isoenzymes.
33.5% of silodosin is excreted through the kidneys and 54.9% through the intestines. The clearance of silodosin is approximately 23.1 l/h. Silodosin is excreted mainly in the form of metabolites and in very small quantities unchanged in the urine. T1/2 of silodosin and carbamoyl glucuronide is 11 hours and 18 hours, respectively.
Pharmacokinetics in different patient groups
Elderly patients: the pharmacokinetics of silodosin and metabolites did not significantly depend on age. The clearance of silodosin did not change in patients over 75 years of age. Patients with impaired liver function: In patients with moderate hepatic impairment (7-9 points on the Child-Pugh scale), the pharmacokinetics of silodosin does not change significantly. The pharmacokinetics of silodosin have not been studied in patients with severe hepatic impairment.
Patients with impaired renal function: For patients with moderate renal impairment, no dose reduction was required. Silodosin is not recommended for patients with severe renal impairment.
Special instructions
Special instructions
As with the use of other alpha1-blockers, a decrease in blood pressure and orthostatic hypotension may be observed during treatment with silodosin. At the first signs of orthostatic hypotension (dizziness, weakness), the patient should sit or lie down and remain in this position until the symptoms of orthostatic hypotension disappear.
Because benign prostatic hyperplasia and prostate tumors have similar symptoms and can develop together, patients with suspected benign prostatic hyperplasia should be examined to exclude prostate tumors before prescribing the drug.
Patients who are taking or have taken alpha1-blockers may experience floppy iris syndrome during cataract surgery, which can lead to complications during surgery. It is necessary to discontinue treatment with alpha1-blockers 1-2 weeks before such surgery. Patients scheduled for cataract surgery are not recommended to begin treatment with Urorec.
Effect on the ability to drive vehicles and operate machinery: no studies have been conducted on the effect on the ability to drive vehicles and equipment. However, patients should be informed about the possible manifestations of symptoms associated with orthostatic hypotension (for example, dizziness), and should also be warned that they should refrain from driving vehicles and equipment until the patient’s individual tolerance to Urorec is determined.
Active ingredient
Active ingredient
Silodosin
Composition
Composition
Composition per capsule:
Active substance:
silodosin 4 mg or 8 mg.
Excipients:
mannitol, pregelatinized starch (starch 1500™),
pregelatinized starch (PCS™ PC-10 starch),
sodium lauryl sulfate, magnesium stearate.
Composition of the gelatin capsule:
gelatin,
titanium dioxide,
iron oxide yellow dye (E-172)
Pregnancy
Pregnancy
Urorek is contraindicated for children under 18 years of age, pregnant women and lactation.
Contraindications
Contraindications
Hypersensitivity to the active substance or any of the excipients.
components or intolerance to them.
Children under 18 years of age.
Severe renal failure (creatinine clearance less than 30 ml/min).
Severe liver failure (insufficient clinical data).
Side Effects
Side Effects
Adverse reactions with frequencies are listed below: very common: >10, common: > 1/100 to 1/1000 to 1/10,000 to <1/1000, frequency unknown (available data do not allow frequency to be determined).Nervous system
Common: dizziness.
Frequency unknown: syncope
Cardiovascular system
Common: orthostatic hypotension.
Respiratory system
Common: nasal congestion.
Gastrointestinal tract
Common: diarrhea.
Uncommon: nausea, dry mouth.
Reproductive system
Very common: retrograde ejaculation, anejaculation
Uncommon; decreased libido, erectile dysfunction
Intraoperative syndrome
Frequency unknown:
Intraoperative floppy iris syndrome during cataract surgery.
Interaction
Interaction
Combination with other alpha-blockers is not recommended due to possible potentiation of action.
The combined use of inhibitors of the CYP3A4 isoenzyme (ketoconazole, clarithromycin, itraconazole, ritonavir) is not recommended, as it increases the concentration of silodosin in plasma.
Phosphodiesterase-5 inhibitors (sildenafil, tadalafil) when used together may increase the risk of developing dizziness.
Antihypertensive drugs beta-blockers, calcium antagonists, drugs acting on the renin-angiotensin-aldosterone system, diuretics, when used together, increase orthostatic hypotension.
Overdose
Overdose
Symptoms: marked decrease in blood pressure, compensatory tachycardia.
Treatment: gastric lavage, taking activated charcoal or an osmotic laxative, symptomatic therapy aimed at increasing circulating blood volume (CBV), vasoconstrictor drugs. Monitoring kidney function. Dialysis is ineffective due to the intense binding of the drug to plasma proteins.
Storage conditions
Storage conditions
The drug should be stored in a dry place, protected from light, out of reach of children, at a temperature not exceeding 30°C.
Shelf life
Shelf life
2 years
Manufacturer
Manufacturer
Recordati chemical and pharmaceutical industry, Italy
Additional information
| Shelf life | 2 years |
|---|---|
| Conditions of storage | The drug should be stored in a dry place, protected from light, out of reach of children at a temperature not exceeding 30 ° C. |
| Manufacturer | Recordati chemical and pharmaceutical industry, Italy |
| Medication form | capsules |
| Brand | Recordati chemical and pharmaceutical industry |
Other forms…
Related products
Buy Urorek, 8 mg capsules 30 pcs with delivery to USA, UK, Europe and over 120 other countries.