No products in the cart.
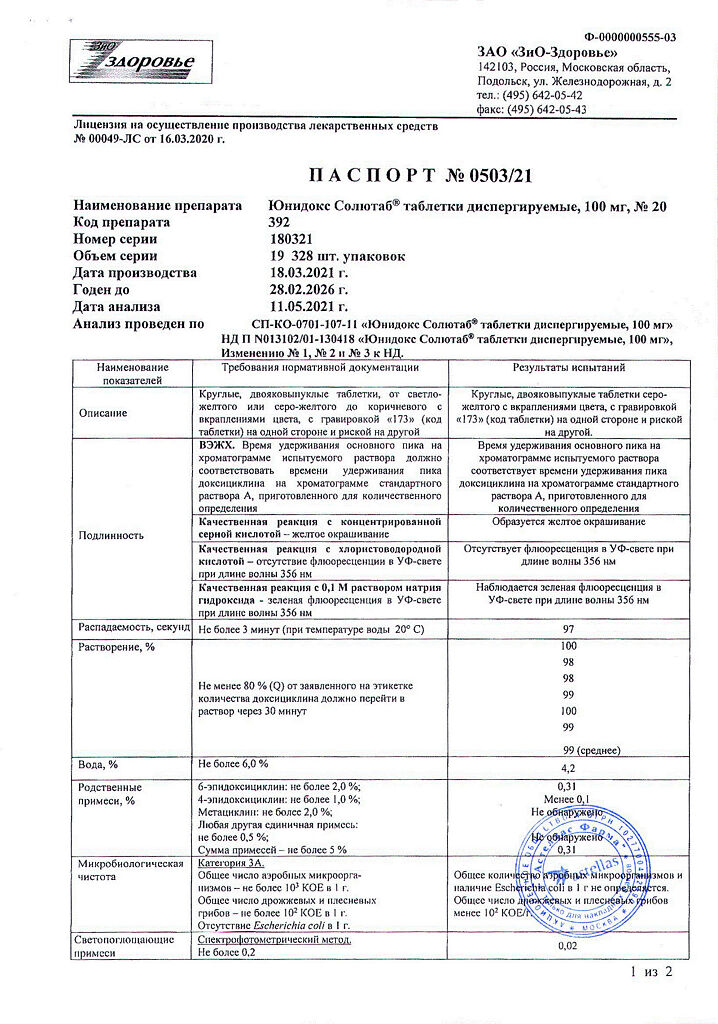
Unidox Solutab, 100 mg 20 pcs
€18.46 €15.39
Description
Pharmacological action – antibacterial, bacteriostatic.
Inhibits protein synthesis in the microbial cell by disrupting the communication of transport RNA of ribosomal membrane.
Pharmacodynamics
A broad-spectrum antibiotic of tetracycline group. It is bacteriostatic, inhibits protein synthesis in microbial cell by interaction with 30S ribosome subunit. It is active against many Gram-positive and Gram-negative microorganisms: Streptococcus spp., Treponema spp., Staphylococcus spp., Klebsiella spp, Enterobacter spp. (including E. aerugenes), Neisseria gonorrhoeae, Neisseria meningitidis, Haemophilus influenzae, Chlamydia spp., Mycoplasma spp., Ureaplasma urealyticum, Listeria monocytogenes, Rickettsia spp., Typhus exanthematicus, Escherichia coli, Shigella spp, Campylobacter fetus, Vibrio cholerae, Yersinia spp. (including Yersinia pestis), Brucella spp., Francisella tularensis, Bacillus anthracis, Bartonella bacilliformis, Pasteurella multocida, Borrelia recurrentis, Clostridium spp. (except Clostridium difficile), Actinomyces spp, Fusobacterium fusiforme, Calymmatobacterium granulomatis, Propionibacterium acnes, some protozoa (Entamoeba spp., Plasmodium falciparum).
As a rule, it has no effect on Acinetobacter spp., Proteus spp., Pseudomonas spp., Serratia spp., Providencia spp., Enterococcus spp.
The possibility of acquired resistance to doxycycline in a number of pathogens should be taken into account, which is often cross-over within the group (i.e., strains resistant to doxycycline will simultaneously be resistant to the entire group of tetracyclines).
Pharmacokinetics
Absorption
Absorption is rapid and high (about 100%). Food intake has little effect on absorption of the drug.
The Cmax of doxycycline in blood plasma (2.6-3 mcg/ml) is reached 2 hours after 200 mg administration, after 24 hours the concentration of active substance in blood plasma decreases to 1.5 mcg/ml.
After administration of 200 mg on the first day of treatment and 100 mg/day on subsequent days, the plasma concentration of doxycycline is 1.5-3 mcg/ml.
Distribution
Doxycycline reversibly binds to plasma proteins (80-90%), penetrates well into organs and tissues, poorly – into the cerebrospinal fluid (10-20% of plasma levels), but doxycycline concentration in the cerebrospinal fluid increases with inflammation of the spinal cord.
The volume of distribution is 1.58 l/kg. Within 30-45 minutes after oral administration doxycycline is found in therapeutic concentrations in the liver, kidneys, lungs, spleen, bones, teeth, prostate, eye tissues, pleural and ascitic fluid, bile, synovial exudate, exudate of maxillary and frontal sinuses and gingival sinus fluid.
In normal hepatic function, the drug level in bile is 5-10 times higher than in plasma.
In saliva, 5-27% of the value of doxycycline concentration in blood plasma is determined.
Doxycycline penetrates the placental barrier, in small amounts is secreted into breast milk.
Accumulates in dentin and bone tissue.
Metabolism
A small portion of doxycycline is metabolized.
Elevation
The T1/2 after a single oral dose is 16-18 hours, after repeated doses is 22-23 hours.
About 40% of the drug taken is excreted by the kidneys and 20-40% is excreted through the intestines as inactive forms (chelates).
Pharmacokinetics in special clinical cases
The half-life of the drug in patients with impaired renal function does not change, because its excretion through the intestine increases.
Hemodialysis and peritoneal dialysis have no effect on plasma concentration of doxycycline.
Indications
Indications
Infectious and inflammatory diseases caused by microorganisms sensitive to the drug:
– respiratory tract infections, incl. pharyngitis, acute bronchitis, exacerbation of COPD, tracheitis, bronchopneumonia, lobar pneumonia, community-acquired pneumonia, lung abscess, pleural empyema;
– infections of ENT organs, incl. otitis media, sinusitis, tonsillitis;
– infections of the genitourinary system (cystitis, pyelonephritis, bacterial prostatitis, urethritis, urethrocystitis, urogenital mycoplasmosis, acute orchiepididymitis; endometritis, endocervicitis and salpingoophoritis as part of combination therapy), incl. sexually transmitted infections (urogenital chlamydia, syphilis in patients with penicillin intolerance, uncomplicated gonorrhea (as an alternative therapy), granuloma inguinale, lymphogranuloma venereum);
– infections of the gastrointestinal tract and biliary tract (cholera, yersiniosis, cholecystitis, cholangitis, gastroenterocolitis, bacillary and amoebic dysentery, travelers’ diarrhea);
– skin and soft tissue infections (including wound infections after animal bites), severe acne (as part of combination therapy);
– other diseases (yaws, legionellosis, chlamydia of various localizations (including prostatitis and proctitis), rickettsiosis, Q fever, Rocky Mountain spotted fever, typhus (including typhus, tick-borne relapsing), Lyme disease (stage I – erythema migrans), tularemia, plague, actinomycosis, malaria; infectious eye diseases (in combination therapy – trachoma); leptospirosis, psittacosis, anthrax (including pulmonary form), bartonellosis, granulocytic ehrlichiosis, brucellosis, sepsis, subacute septic endocarditis, peritonitis);
– prevention of postoperative purulent complications;
– prevention of malaria caused by Plasmodium falciparum during short-term travel (less than 4 months) to areas where strains resistant to chloroquine and/or pyrimethamine-sulfadoxine are common.
Pharmacological effect
Pharmacological effect
Pharmacological action – antibacterial, bacteriostatic.
Suppresses protein synthesis in the microbial cell, disrupting the connection of transport RNAs of the ribosomal membrane.
Pharmacodynamics
Broad-spectrum antibiotic from the tetracycline group. It acts bacteriostatically, inhibits protein synthesis in the microbial cell by interacting with the 30S ribosomal subunit. Active against many gram-positive and gram-negative microorganisms: Streptococcus spp., Treponema spp., Staphylococcus spp., Klebsiella spp., Enterobacter spp. (including E. aerugenes), Neisseria gonorrhoeae, Neisseria meningitidis, Haemophilus influenzae, Chlamydia spp., Mycoplasma spp., Ureaplasma urealyticum, Listeria monocytogenes, Rickettsia spp., Typhus exanthematicus, Escherichia coli, Shigella spp., Campylobacter fetus, Vibrio cholerae, Yersinia spp. (including Yersinia pestis), Brucella spp., Francisella tularensis, Bacillus anthracis, Bartonella bacilliformis, Pasteurella multocida, Borrelia recurrentis, Clostridium spp. (except Clostridium difficile), Actinomyces spp., Fusobacterium fusiforme, Calymmatobacterium granulomatis, Propionibacterium acnes, some protozoa (Entamoeba spp., Plasmodium falciparum).
As a rule, it has no effect on Acinetobacter spp., Proteus spp., Pseudomonas spp., Serratia spp., Providencia spp., Enterococcus spp.
The possibility of acquired resistance to doxycycline in a number of pathogens, which is often cross-resistance within a group, should be taken into account (i.e., strains resistant to doxycycline will simultaneously be resistant to the entire group of tetracyclines).
Pharmacokinetics
Suction
Absorption is fast and high (about 100%). Food intake has little effect on the absorption of the drug.
Cmax of doxycycline in blood plasma (2.6–3 μg/ml) is achieved 2 hours after taking 200 mg; after 24 hours, the concentration of the active substance in blood plasma decreases to 1.5 μg/ml.
After taking 200 mg on the first day of treatment and 100 mg/day on subsequent days, the plasma concentration of doxycycline is 1.5–3 mcg/ml.
Distribution
Doxycycline binds reversibly to plasma proteins (80–90%), penetrates well into organs and tissues, poorly into the cerebrospinal fluid (10–20% of the plasma level), however, the concentration of doxycycline in the cerebrospinal fluid increases with inflammation of the spinal lining.
Volume of distribution – 1.58 l/kg. 30–45 minutes after oral administration, doxycycline is found in therapeutic concentrations in the liver, kidneys, lungs, spleen, bones, teeth, prostate gland, eye tissues, pleural and ascitic fluids, bile, synovial exudate, exudate of the maxillary and frontal sinuses, and in the fluid of the gingival grooves.
With normal liver function, the level of the drug in bile is 5–10 times higher than in plasma.
In saliva, 5–27% of the concentration of doxycycline in blood plasma is determined.
Doxycycline crosses the placental barrier and is secreted into breast milk in small quantities.
Accumulates in dentin and bone tissue.
Metabolism
A small portion of doxycycline is metabolized.
Removal
T1/2 after a single oral dose is 16-18 hours, after repeated doses – 22-23 hours.
Approximately 40% of the drug taken is excreted by the kidneys and 20–40% is excreted through the intestines in the form of inactive forms (chelates).
Pharmacokinetics in special clinical situations
The half-life of the drug in patients with impaired renal function does not change, because its excretion through the intestines increases.
Hemodialysis and peritoneal dialysis do not affect the concentration of doxycycline in blood plasma.
Special instructions
Special instructions
There is a possibility of cross-resistance and hypersensitivity to other tetracycline drugs.
Tetracyclines may increase PT; use of tetracyclines in patients with coagulopathies should be carefully monitored.
The anti-anabolic effect of tetracyclines can lead to an increase in the level of residual urea nitrogen in the blood. As a rule, this is not significant for patients with normal renal function. However, in patients with renal failure, an increase in azotemia may occur. The use of tetracyclines in patients with impaired renal function requires medical supervision.
With long-term use of the drug, periodic monitoring of laboratory blood parameters, liver and kidney function is required.
Due to the possible development of photodermatitis, it is necessary to limit insolation during treatment and for 4–5 days after it.
Long-term use of the drug can cause dysbacteriosis and, as a result, the development of hypovitaminosis (especially B vitamins).
To prevent dyspeptic symptoms, it is recommended to take the drug with meals.
The specific effects on the ability to drive a car and operate machinery have not been studied.
Active ingredient
Active ingredient
Doxycycline
Composition
Composition
1 dispersible tablet contains:
active substances:
doxycycline monohydrate – 100 mg (in terms of doxycycline);
excipients: excipients: MCC; saccharin; hyprolose (low-substituted); hypromellose; colloidal silicon dioxide (anhydrous); magnesium stearate; lactose monohydrate.
Pregnancy
Pregnancy
Unidox Solutab is contraindicated during pregnancy. During treatment with Unidox, breastfeeding should be stopped.
Contraindications
Contraindications
hypersensitivity to tetracyclines;
age up to 8 years;
breast-feeding;
pregnancy;
porphyria;
severe impairment of liver and/or kidney function.
Side Effects
Side Effects
From the gastrointestinal tract: anorexia, nausea, vomiting, dysphagia, diarrhea; enterocolitis, pseudomembranous colitis.
Dermatological and allergic reactions: urticaria, photosensitivity, angioedema, anaphylactic reactions, exacerbation of systemic lupus erythematosus, maculopapular and erythematous rash, pericarditis, exfoliative dermatitis.
From the liver: liver damage during long-term use or in patients with renal or hepatic insufficiency.
From the kidneys: an increase in residual urea nitrogen (due to the anti-anabolic effect).
From the hematopoietic system: hemolytic anemia, thrombocytopenia, neutropenia, eosinophilia, decreased prothrombin activity.
From the nervous system: benign increase in intracranial pressure (anorexia, vomiting, headache, papilledema), vestibular disorders (dizziness or instability).
On the thyroid gland: in patients treated with doxycycline for a long time, reversible dark brown discoloration of the thyroid tissue is possible.
From the side of teeth and bones: doxycycline slows down osteogenesis, disrupts the normal development of teeth in children (the color of teeth irreversibly changes, enamel hypoplasia develops).
Other: candidiasis (stomatitis, glossitis, proctitis, vaginitis) as a manifestation of superinfection.
Interaction
Interaction
Antacids containing aluminum, magnesium, calcium, iron preparations, sodium bicarbonate, magnesium-containing laxatives reduce the absorption of doxycycline, so their use should be separated by an interval of 3 hours.
Due to the suppression of intestinal microflora by doxycycline, the prothrombin index decreases, which requires dose adjustment of indirect anticoagulants.
When doxycycline is combined with bactericidal antibiotics that interfere with cell wall synthesis (penicillins, cephalosporins), the effectiveness of the latter is reduced.
Doxycycline reduces the reliability of contraception and increases the frequency of acyclic bleeding when taking estrogen-containing hormonal contraceptives.
Ethanol, barbiturates, rifampicin, carbamazepine, phenytoin and other stimulants of microsomal oxidation, accelerating the metabolism of doxycycline, reduce its concentration in the blood plasma.
The simultaneous use of doxycycline and retinol increases intracranial pressure.
Overdose
Overdose
Symptoms: increased adverse reactions caused by liver damage (vomiting, fever, jaundice, azotemia, increased transaminase levels, increased PT).
Treatment: immediately after taking large doses, gastric lavage, drinking plenty of fluids, and, if necessary, inducing vomiting are recommended. Activated carbon and osmotic laxatives are prescribed. Hemodialysis and peritoneal dialysis are not recommended due to low efficiency.
Storage conditions
Storage conditions
At a temperature of 15–25 °C. Keep out of the reach of children.
Shelf life
Shelf life
5 years. Do not use after the expiration date stated on the package.
Manufacturer
Manufacturer
ZiO-Zdorovye CJSC, Russia
Additional information
| Shelf life | 5 years. Do not use after the expiration date stated on the package. |
|---|---|
| Conditions of storage | At a temperature of 15-25 °C. Keep out of reach of children. |
| Manufacturer | ZiO-Zdorovye CJSC, Russia |
| Medication form | dispersible tablets |
| Brand | ZiO-Zdorovye CJSC |
Other forms…
Related products
Buy Unidox Solutab, 100 mg 20 pcs with delivery to USA, UK, Europe and over 120 other countries.