No products in the cart.
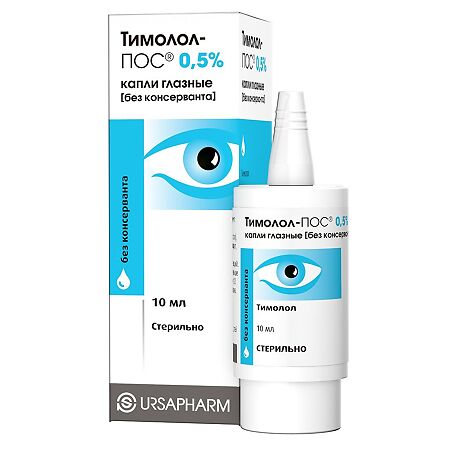
Timolol-pos, eye drops 0.5% 10 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
Pharmacotherapeutic group – Antiglaucoma drug – beta-adrenoblocker.
ATX: S01ED01
Pharmacodynamics
Neselective beta-adrenoceptor blocker without sympathomimetic activity. When used topically, it lowers intraocular pressure by reducing aqueous humor formation and slightly increasing its outflow. Action appeared 20 min after dosing, maximum effect 1-2 h later; time of action – 24 h.
Pharmacokinetics
Timolol maleate rapidly penetrates through the cornea into the eye tissue. A small amount enters the systemic bloodstream by absorption through the conjunctiva, nasal mucosa and lacrimal tract. Metabolites are excreted by the kidneys. In infants and small children the concentration of the active substance is significantly higher than its maximum concentration in adult plasma.
Indications
Indications
– primary open-angle glaucoma;
– secondary glaucoma (uveal, aphakic, post-traumatic);
– congenital glaucoma (if other therapeutic measures are ineffective);
– acute increase in ophthalmotonus;
– primary angle-closure glaucoma (in combination with miotics).
Pharmacological effect
Pharmacological effect
Pharmacotherapeutic group – Antiglaucoma agent – beta-blocker.
ATC: S01ED01
Pharmacodynamics
Non-selective beta-adrenergic receptor blocker without sympathomimetic activity. When applied topically, it lowers intraocular pressure by reducing the formation of aqueous humor and slightly increasing its outflow. The effect appears 20 minutes after instillation, the maximum effect is after 1-2 hours; duration of action – 24 hours
Pharmacokinetics
Timolol maleate quickly penetrates through the cornea into the eye tissue. In small quantities it enters the systemic circulation through absorption through the conjunctiva, nasal mucosa and lacrimal tract. Excretion of metabolites by the kidneys. In newborns and small children, the concentration of the active substance significantly exceeds its maximum concentration in the plasma of adults.
Special instructions
Special instructions
Effectiveness monitoring should be carried out approximately 3-4 weeks after the start of therapy. During the treatment period, tear function, corneal integrity, and visual fields should be monitored at least once every 6 months. During the treatment period, care must be taken when driving vehicles and engaging in other potentially hazardous activities that require increased concentration and speed of psychomotor reactions and good vision (within 0.5 hours after instillation into the eye).
Contact lenses should be removed before instillation and put on no earlier than 15 minutes after.
With long-term use of timolol, the effect may weaken.
Before an upcoming surgical intervention using general anesthesia, discontinue the drug 48 hours in advance.
The original design of the dropper bottle, operating on the principle of an airless pump, does not allow outside air to penetrate into the bottle and thereby ensures the sterility of eye drops in the absence of a preservative in their composition.
The use of Timolol-POS eye drops [without preservative] allows you to avoid unwanted effects of the preservative on the external tissues of the eye, conjunctiva and cornea, and also eliminates the risk of developing allergic reactions to the preservative.
After opening the bottle and the first instillation of Timolol-POS eye drops [without preservative], the drug can be used for 12 weeks. To do this, mark on the bottle and/or on the cardboard box the date when you first instilled the drug. On the label of the bottle and on the cardboard box there is a special column for the patient to fill out: “Date of first use.”
After using the contents of the bottle, you should purchase a new bottle of the drug; the used bottle is not intended for reuse and must be disposed of (throw the used bottle in the trash).
Active ingredient
Active ingredient
Timolol
Composition
Composition
TIMOLOL-POS 0.5%
Active component:
timolol maleate 6.84 mg (in terms of timolol – 5.0 mg)
Excipients: sodium dihydrogen phosphate dihydrate, sodium hydrogen phosphate dodecahydrate, water for injection.
Pregnancy
Pregnancy
During pregnancy, the drug is used if the expected benefit to the mother outweighs the potential risk to the fetus. Breastfeeding should be stopped while using the drug.
Contraindications
Contraindications
severe chronic obstructive pulmonary disease (COPD), including bronchial asthma;
sinus bradycardia (slow heartbeat);
atrioventricular block II and III degrees;
acute and chronic heart failure of II and III degrees;
cardiogenic shock;
dystrophic diseases of the cornea;
severe vasomotor rhinitis;
hypersensitivity to the components of the drug.
Use with caution: sinoatrial blockade, arterial hypotension, childhood, especially the neonatal period, cerebrovascular insufficiency, diabetes mellitus, hypoglycemia, thyrotoxicosis, myasthenia gravis, simultaneous administration of other beta-blockers.
Side Effects
Side Effects
Local reactions:
Eye irritation, manifested in the form of conjunctivitis, blepharitis, keratitis (hyperemia of the skin of the eyelids, burning and itching in the eyes, hyperemia of the conjunctiva, lacrimation or decreased lacrimation, photophobia, swelling of the corneal epithelium, short-term impairment of visual acuity; with long-term use, the development of superficial punctate keratopathy (decreased transparency) is possible cornea) and decreased sensitivity of the cornea), ptosis, diplopia;
System reactions:
From the cardiovascular system: decreased blood pressure, bradycardia, bradyarrhythmia, atrioventricular block, heart failure, cardiac arrest, decreased myocardial contractility, chest pain, transient cerebrovascular accident, collapse;
From the respiratory system: shortness of breath, bronchospasm, pulmonary insufficiency;
From the central nervous system: dizziness, headache, drowsiness, hallucinations, slowing of the speed of psychomotor reactions;
Other side effects: allergic reactions (including urticaria), rhinitis, nasal congestion, nosebleeds, nausea, vomiting, diarrhea, paresthesia, muscle weakness, sexual dysfunction, depression.
Interaction
Interaction
Epinephrine, pilocarpine, systemic beta-blockers enhance the effect. When used with reserpine, severe bradycardia or hypotension may develop (this combination requires careful medical supervision); with calcium channel blockers or cardiac glycosides – there may be impaired atrioventricular conduction, acute left ventricular failure or arterial hypotension.
Enhances the effect of muscle relaxants (timolol should be discontinued 48 hours before the intended general anesthesia with the use of peripheral muscle relaxants). It should not be used simultaneously with antipsychotic drugs (neuroleptics) and anxiolytic drugs (tranquilizers). During treatment, it is not recommended to take ethanol (a sharp decrease in blood pressure is possible).
Prescribe with caution simultaneously with antihypertensive drugs, other beta-blockers, insulin or oral hypoglycemic drugs, glucocorticosteroids, psychoactive drugs.
Overdose
Overdose
When applied topically at the recommended dose, no overdose symptoms were observed. If accidentally ingested, the following symptoms may develop: nausea, vomiting, dizziness, headache, decreased blood pressure, bronchospasm, bradycardia. In case of overdose symptoms, treatment is symptomatic. Isoprenaline can be administered intravenously to eliminate severe bradycardia or bronchospasm, and dobutamine to treat arterial hypotension.
Storage conditions
Storage conditions
At a temperature not higher than 25 0C. Keep out of the reach of children!
Shelf life
Shelf life
In unopened original packaging: 3 years. After opening the bottle 12 weeks.
Do not use after the expiration date stated on the package.
Manufacturer
Manufacturer
Ursafarm Arzneimittel GmbH, Germany
Additional information
| Shelf life | In the closed original package: 3 years. After opening the bottle 12 weeks. Do not use after the expiration date stated on the package. |
|---|---|
| Conditions of storage | At a temperature not exceeding 25 0C. Keep out of reach of children! |
| Manufacturer | Ursapharm Arzneimittel, Germany |
| Medication form | eye drops |
| Brand | Ursapharm Arzneimittel |
Related products
Buy Timolol-pos, eye drops 0.5% 10 ml with delivery to USA, UK, Europe and over 120 other countries.