No products in the cart.
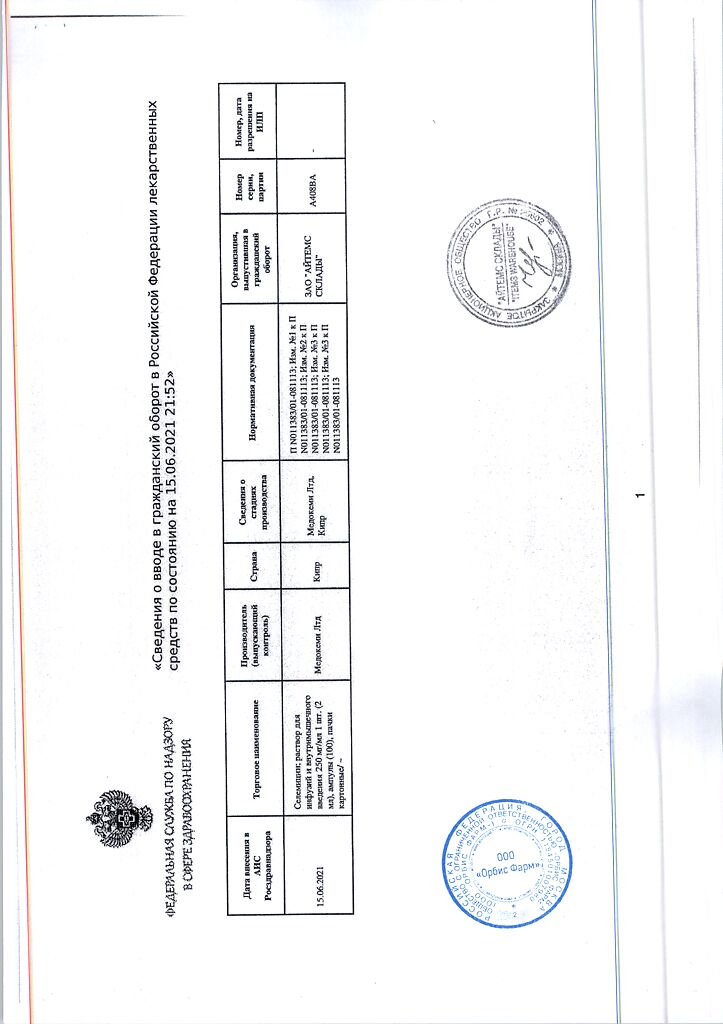
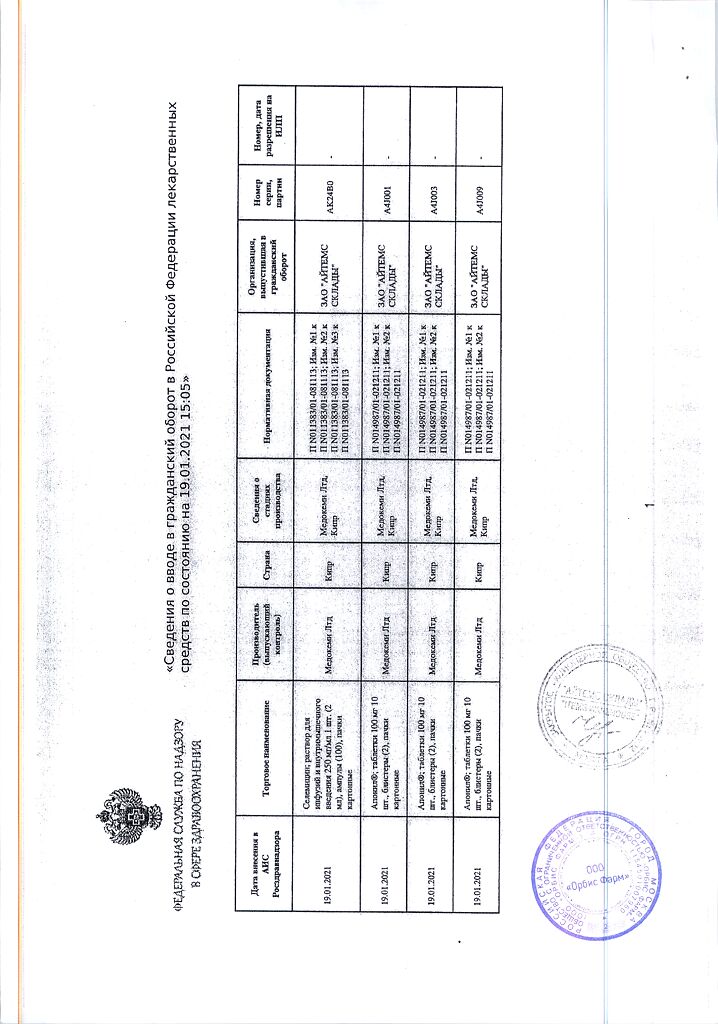
Selemicin, 250 mg/ml 2 ml 100 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
Pharmacological properties
Semi-synthetic broad-spectrum antibiotic, acts bactericidally. It binds with 30S ribosome subunit, prevents formation of transport and matrix ribonucleic acid (RNA) complex, blocks protein synthesis and destroys cytoplasmic membranes of bacteria.
Highly active against aerobic Gram-negative microorganisms – Pseudomonas aeruginosa, Escherichia coli, Klebsiella spp., Serratia spp., Providencia spp., Enterobacter spp., Salmonella spp, Shigella spp.; some Gram-positive microorganisms – Staphylococcus spp. (including those resistant to penicillin, some cephalosporins); moderately active against Streptococcus spp.
In concomitant administration with benzylpenicillin it has a synergistic effect against strains of Enterococcus faecalis.
It has no effect on anaerobic microorganisms.
Amicacin does not lose activity under the action of enzymes that inactivate other aminoglycosides and may remain active against strains of Pseudomonas aeruginosa resistant to tobramycin, gentamicin and netilmicin.
Pharmacokinetics
After intramuscular (i.m.) administration, absorption is rapid and complete. Maximum concentration (Cmax) with 7.5 mg/kg intramuscular administration is 21 µg/ml, after 30 min intravenous (IV) infusion of 7.5 mg/kg is 38 µg/ml. Time of reaching maximum concentration (TCmax) is about 1.5 h after intravenous administration. Binding to plasma proteins is 4-11%.
It is well distributed in extracellular fluid (contents of abscesses, pleural effusion, ascitic fluid, pericardial, synovial, lymphatic and peritoneal fluid); in high concentration it is found in urine; in low concentration – in bile, breast milk, aqueous humor of eyes, bronchial secretion, sputum and cerebrospinal fluid (CSF). It penetrates well into all body tissues, where it accumulates intracellularly; high concentrations are noted in organs with good blood supply: lungs, liver, myocardium, spleen, and especially in kidneys, where it accumulates in cortical substance, lower concentrations – in muscles, adipose tissue and bones.
When administered in medium therapeutic doses (normal) in adults, amikacin does not penetrate the blood-brain barrier (BBB); in inflammation of the brain membranes permeability increases slightly. Neonates reach higher concentrations in the CSF than adults; it passes through the placenta and is detected in fetal blood and amniotic fluid. Distribution volume in adults is 0.026 l/kg, in children – 0.2-0.4 l/kg, in newborns – less than 1 week old and weighing less than 1500 g – up to 0.68 l/kg, in those less than 1 week old and weighing more than 1500 g – up to 0.58 l/kg, in cystic fibrosis patients – 0.3-0.39 l/kg. Average therapeutic concentration when administered by IV or IM is maintained for 10-12 hours.
It is not metabolized. Elimination half-life (T1/2) in adults is 2-4 hours, in infants – 5-8 hours, in older children – 2.5-4 hours. Final value of T1/2 is more than 100 hours (release from intracellular depots).
Extracted by the kidneys by glomerular filtration (65-94%), mainly unchanged. Renal clearance is 79-100 ml/min.
The T1/2 in adults with impaired renal function varies depending on the degree of impairment – up to 100 h, in patients with cystic fibrosis – 1-2 h, in patients with burns and hyperthermia T1/2 may be shorter compared to the average figures due to increased clearance.
It is excreted with hemodialysis (50% in 4-6 hours), peritoneal dialysis is less effective (25% in 48-72 hours).
Indications
Indications
Infectious and inflammatory diseases caused by gram-negative microorganisms (resistant to gentamicin, sizomycin and kanamycin) or associations of gram-positive and gram-negative microorganisms:
respiratory tract infections (bronchitis, pneumonia, pleural empyema, lung abscess);
sepsis;
septic endocarditis;
central nervous system infections (including meningitis);
abdominal infections (including peritonitis);
genitourinary tract infections (pyelonephritis, cystitis, urethritis, prostatitis, gonorrhea);
purulent infections of the skin and soft tissues (including infected burns, infected ulcers and bedsores of various origins);
biliary tract infections;
infections of bones and joints (including osteomyelitis);
wound infection;
postoperative infections;
otitis.
Pharmacological effect
Pharmacological effect
Pharmacological properties
A semi-synthetic broad-spectrum antibiotic with a bactericidal effect. By binding to the 30S subunit of ribosomes, it prevents the formation of a complex of transport and matrix ribonucleic acids (RNA), blocks protein synthesis, and also destroys the cytoplasmic membranes of bacteria.
Highly active against aerobic gram-negative microorganisms – Pseudomonas aeruginosa, Escherichia coli, Klebsiella spp., Serratia spp., Providencia spp., Enterobacter spp., Salmonella spp., Shigella spp.; some gram-positive microorganisms – Staphylococcus spp. (including those resistant to penicillin and some cephalosporins); moderately active against Streptococcus spp.
When administered simultaneously with benzylpenicillin, it has a synergistic effect against strains of Enterococcus faecalis.
Does not affect anaerobic microorganisms.
Amikacin does not lose activity under the influence of enzymes that inactivate other aminoglycosides, and can remain active against strains of Pseudomonas aeruginosa resistant to tobramycin, gentamicin and netilmicin.
Pharmacokinetics
After intramuscular (IM) administration, it is absorbed quickly and completely. The maximum concentration (Cmax) with intramuscular administration of 7.5 mg/kg is 21 mcg/ml, after 30 minutes of intravenous (IV) infusion of 7.5 mg/kg is 38 mcg/ml. The time to reach maximum concentration (TCmax) is about 1.5 hours after IM administration. Bonding with plasma proteins is 4–11%.
Well distributed in extracellular fluid (abscess contents, pleural effusion, ascitic, pericardial, synovial, lymphatic and peritoneal fluid); found in high concentrations in urine; in low levels – in bile, breast milk, aqueous humor of the eye, bronchial secretions, sputum and cerebrospinal fluid (CSF). Penetrates well into all tissues of the body, where it accumulates intracellularly; high concentrations are observed in organs with good blood supply: lungs, liver, myocardium, spleen, and especially in the kidneys, where they accumulate in the cortex; lower concentrations are found in muscles, adipose tissue and bones.
When prescribed in average therapeutic doses (normally) to adults, amikacin does not penetrate the blood-brain barrier (BBB); with inflammation of the meninges, permeability increases slightly. Neonates achieve higher CSF concentrations than adults; passes through the placenta and is found in fetal blood and amniotic fluid. Volume of distribution in adults – 0.026 l/kg, in children – 0.2-0.4 l/kg, in newborns – less than 1 week old and weighing less than 1500 g – up to 0.68 l/kg, less than 1 week old and weighing more than 1500 g – up to 0.58 l/kg, in patients with cystic fibrosis – 0.3-0.39 l/kg. The average therapeutic concentration with intravenous or intramuscular administration lasts for 10–12 hours.
Not metabolized. The half-life (T1/2) in adults is 2-4 hours, in newborns – 5-8 hours, in older children – 2.5-4 hours. The final T1/2 value is more than 100 hours (release from intracellular depots).
It is excreted by the kidneys by glomerular filtration (65–94%), mainly unchanged. Renal clearance is 79–100 ml/min.
T1/2 in adults with impaired renal function varies depending on the degree of impairment – up to 100 hours, in patients with cystic fibrosis – 1-2 hours, in patients with burns and hyperthermia T1/2 may be shorter than the average due to increased clearance.
It is eliminated by hemodialysis (50% in 4–6 hours), peritoneal dialysis is less effective (25% in 48–72 hours).
Special instructions
Special instructions
Before use, the sensitivity of the isolated pathogens is determined using disks containing 30 μg of amikacin. With a diameter of the growth-free zone of 17 mm or more, the microorganism is considered sensitive, from 15 to 16 mm – moderately sensitive, less than 14 mm – resistant.
The concentration of amikacin in plasma should not exceed 25 mcg/ml (a concentration of 15–25 mcg/ml is therapeutic).
During the treatment period, it is necessary to monitor the function of the kidneys, auditory nerve and vestibular apparatus at least once a week.
The likelihood of developing nephrotoxicity is higher in patients with impaired renal function, as well as when prescribing high doses or for a long time (in these patients, daily monitoring of renal function may be required).
If audiometric tests are unsatisfactory, the dose of the drug is reduced or treatment is stopped.
Patients with infectious and inflammatory diseases of the urinary tract are recommended to take increased amounts of fluid.
In the absence of positive clinical dynamics, one should remember the possibility of the development of resistant microorganisms. In such cases, it is necessary to discontinue treatment and begin appropriate therapy.
The disulfite contained in sodium ampoules can cause the development of allergic complications in patients (including anaphylactic reactions), especially in patients with a burdened allergic history.
It is recommended to avoid the simultaneous use of oto- and nephrotoxic drugs.
Impact on the ability to drive vehicles and maintain moving mechanisms
Persons experiencing sensory side effects (dizziness) should refrain from driving vehicles or other potentially dangerous machinery.
Active ingredient
Active ingredient
Amikacin
Composition
Composition
1 ml contains:
Active substance:
Amikacin sulfate, equivalent to amikacin base – 250 mg;
Excipients:
Sodium citrate dihydrate – 25.10 mg,
Sodium disulfite – 6.60 mg,
concentrated sulfuric acid – up to pH 3.5–5.5,
water for injections – up to 1 ml.
Pregnancy
Pregnancy
The drug is contraindicated during pregnancy.
If it is necessary to prescribe the drug during lactation, the issue of stopping breastfeeding should be resolved.
Contraindications
Contraindications
Hypersensitivity to amikacin (including a history of other aminoglycosides) and other components of the drug, auditory neuritis, severe chronic renal failure with azotemia and uremia, pregnancy.
With caution
Myasthenia gravis, parkinsonism, botulism (aminoglycosides can cause disruption of neuromuscular transmission, which leads to further weakening of skeletal muscles), dehydration, renal failure, neonatal period, prematurity, old age.
Side Effects
Side Effects
Allergic reactions: skin rash, itching, skin hyperemia, fever, Quincke’s edema.
From the digestive system: nausea, vomiting, liver dysfunction (increased activity of “liver” transaminases – aspartate aminotransferase (AST), alanine aminotransferase (ALT), hyperbilirubinemia).
From the nervous system: headache, drowsiness, neurotoxic effect (muscle twitching, numbness, tingling, convulsions), disruption of neuromuscular transmission (respiratory arrest).
From the urinary system: nephrotoxicity – impaired renal function (oliguria, proteinuria, microhematuria).
From the hematopoietic organs: anemia, leukopenia, granulocytopenia, thrombocytopenia, hypercreatininemia, azotemia, eosinophilia.
From the senses: ototoxicity (hearing loss, vestibular and labyrinthine disorders, irreversible deafness), toxic effects on the vestibular apparatus (discoordination of movements, dizziness, nausea, vomiting).
From the musculoskeletal system: arthralgia.
Local reactions: pain at the injection site, dermatitis.
Interaction
Interaction
Pharmaceutically incompatible with penicillins, heparin, cephalosporins, capreomycin, amphotericin B, hydrochlorothiazide, erythromycin, nitrofurantoin, vitamins B and C, potassium chloride.
Shows synergism when interacting with carbenicillin, benzylpenicillin, cephalosporins (in patients with severe chronic renal failure, when used together with beta-lactam antibiotics, the effectiveness of aminoglycosides may be reduced).
Nalidixic acid, polymyxin B, cisplatin, and vancomycin increase the risk of ototoxicity and nephrotoxicity.
Diuretics (especially furosemide), cephalosporins, penicillins, sulfonamides and non-steroidal anti-inflammatory drugs, competing for active secretion in the nephron tubules, block the elimination of aminoglycosides, increase their concentration in the blood serum, increasing nephro- and neurotoxicity.
Enhances the muscle relaxant effect of non-depolarizing muscle relaxants.
Methoxyflurane, polymyxins for parenteral administration capreomycin and other drugs that block neuromuscular transmission (halogenated hydrocarbons as drugs for inhalation anesthesia, opioid analgesics), transfusion of large amounts of blood with nitrate preservatives increase the risk of respiratory arrest (especially with intraperitoneal administration of amikacin).
Parenteral administration of indomethacin increases the risk of developing toxic effects of aminoglycosides (increased half-life and decreased clearance).
Reduces the effect of antimyasthenic drugs.
Overdose
Overdose
Symptoms:
Toxic reactions (hearing loss, ataxia, dizziness, urinary disorders, thirst, loss of appetite, nausea, vomiting, ringing or fullness in the ears, breathing problems).
Treatment:
To relieve the blockade of neuromuscular transmission and its consequences – hemodialysis or peritoneal dialysis; anticholinesterase drugs, calcium salts, artificial ventilation, other symptomatic and supportive therapy.
Storage conditions
Storage conditions
The drug should be stored in a dry place, protected from light and out of reach of children, at a temperature not exceeding 25°C.
Shelf life
Shelf life
3 years
Manufacturer
Manufacturer
Medokemi Ltd, Cyprus
Additional information
| Shelf life | 3 years |
|---|---|
| Conditions of storage | The drug should be stored in a dry place, protected from light and out of reach of children, at a temperature not exceeding 25 ° C. |
| Manufacturer | Medokemi Ltd, Cyprus |
| Medication form | Infusion and intravenous solution |
| Brand | Medokemi Ltd |
Related products
Buy Selemicin, 250 mg/ml 2 ml 100 pcs with delivery to USA, UK, Europe and over 120 other countries.