No products in the cart.
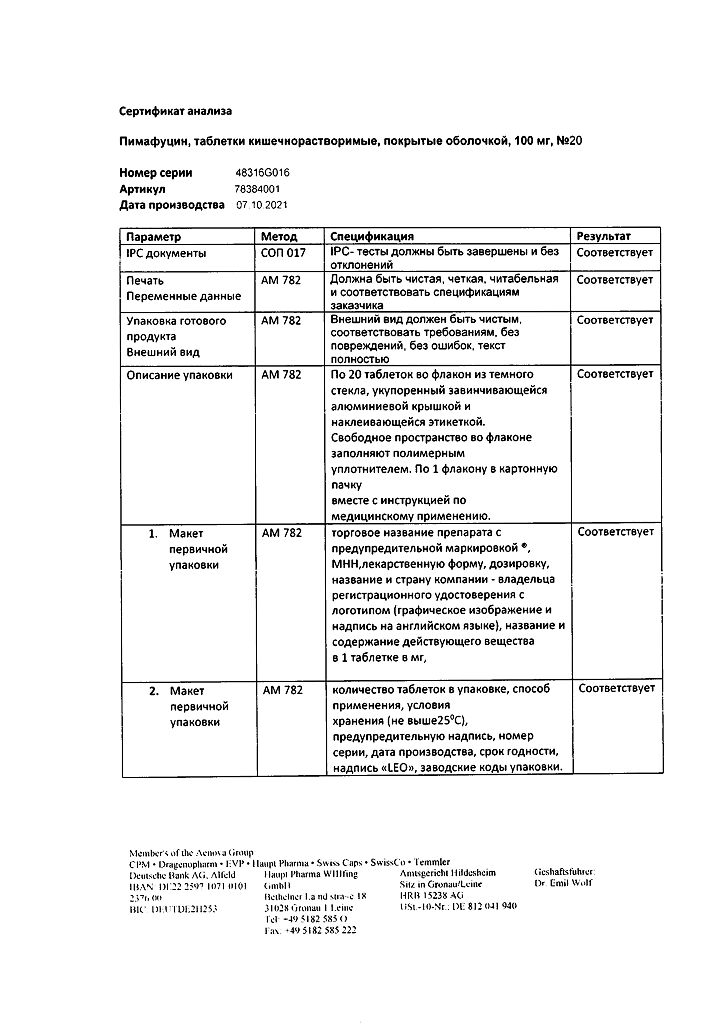
Pimafucin, 100 mg 20 pcs
€19.32 €16.10
Out of stock
(E-mail when Stock is available)
Description
Antifungal polyene (tetraene) antibiotic from the macrolide group. It has a predominantly fungicidal effect. Natamycin irreversibly binds to the ergosterol of the cell membrane of the fungus, which leads to violation of its integrity, loss of cytoplasmic content and cell death.
Pharmacodynamics
The majority of pathogenic yeasts and molds including genera Candida, Aspergillus, Cephalosporium, Fusarium and Penicillium are sensitive to natamycin. Dermatophytes and Pseudoallescheria boydii are less sensitive to natamycin. Cases of resistance to natamycin in clinical practice have not been noted. Natamycin in the dosage form of enteric coated tablets acts only in the intestinal lumen.
Pharmacokinetics
In oral administration natamycin is practically not absorbed from the gastrointestinal tract and has no systemic action.
Indications
Indications
Intestinal candidiasis;
acute pseudomembranous and acute atrophic candidiasis in patients with cachexia, immune deficiency, as well as after therapy with antibiotics, corticosteroids, cytostatics;
sanitation of the intestinal reservoir of fungi of the genus Candida for candidiasis of the skin and mucous membranes, incl. for candidiasis vaginitis, vulvitis and vulvovaginitis.
Pharmacological effect
Pharmacological effect
Antifungal polyene (tetraene) antibiotic from the macrolide group. It has a predominantly fungicidal effect. Natamycin irreversibly binds to ergosterol in the cell membrane of the fungal cell, which leads to disruption of its integrity, loss of cytoplasmic contents and cell death.
Pharmacodynamics
Most pathogenic yeasts and molds are sensitive to natamycin, including the genera Candida, Aspergillus, Cephalosporium, Fusarium and Penicillium. Dermatophytes and Pseudoallescheria boydii are less sensitive to natamycin. There are no cases of resistance to natamycin in clinical practice. Natamycin in the dosage form of enteric-coated tablets acts only in the intestinal lumen.
Pharmacokinetics
When taken orally, natamycin is practically not absorbed from the gastrointestinal tract and has no systemic effect.
Special instructions
Special instructions
Patients with rare congenital intolerance to galactose, fructose, lapp lactase deficiency, glucose-galactose malabsorption or sucrase-isomaltase deficiency should take into account that the drug contains lactose monohydrate (66 mg) and sucrose (122 mg). If the patient suffers from one of the listed diseases, be sure to consult a doctor before taking Pimafucin® tablets.
Pimafucin® in this dosage form is not recommended for use in infants due to possible difficulty in swallowing tablets.
Impact on the ability to drive vehicles and other mechanisms that require increased concentration
Pimafucin® does not affect the ability to drive vehicles and machines.
Interaction with alcohol
There is no data on interaction with alcohol.
Conditions for dispensing from pharmacies
The drug is approved for use as an over-the-counter product.
Active ingredient
Active ingredient
Natamycin
Composition
Composition
Active ingredients:
natamycin 100 mg.
Excipients:
potato starch – 79.2 mg,
povidone K30 – 5 mg,
magnesium stearate – 2.5 mg,
lactose monohydrate – 66 mg.
Shell composition:
cellacephate – 18 mg,
triacetin – 2.7 mg,
sucrose – 122 mg,
methyl parahydroxybenzoate – 0.07 mg,
calcium carbonate – 30 mg,
kaolin – 14.2 mg,
talc – 21.7 mg,
titanium dioxide – 3 mg,
acacia gum – 7.3 mg,
gelatin – 3.3 mg,
white beeswax – 0.13 mg.
Contraindications
Contraindications
Hypersensitivity to the components of the drug.
Side Effects
Side Effects
When using Pimafucin, the following are possible:
From the digestive system: nausea and diarrhea are possible (occurring in the first days of taking the drug and going away on their own during treatment).
The patient should be warned that if any of the side effects indicated in the instructions get worse, or he notices any other side effects not listed in the instructions, he should inform the doctor.
Interaction
Interaction
No drug interactions between Pimafucin® and other drugs have been detected.
Overdose
Overdose
Currently, no cases of overdose of Pimafucin® have been reported.
Storage conditions
Storage conditions
At a temperature not exceeding 25 °C
Shelf life
Shelf life
4 years
Manufacturer
Manufacturer
Haupt Pharma Wülfing GmbH, Germany
Additional information
| Shelf life | 4 years |
|---|---|
| Conditions of storage | At a temperature not exceeding 25 °C |
| Manufacturer | Haupt Pharma Wulfing GmbH, Germany |
| Medication form | enteric soluble tablets |
| Brand | Haupt Pharma Wulfing GmbH |
Other forms…
Related products
Buy Pimafucin, 100 mg 20 pcs with delivery to USA, UK, Europe and over 120 other countries.