No products in the cart.
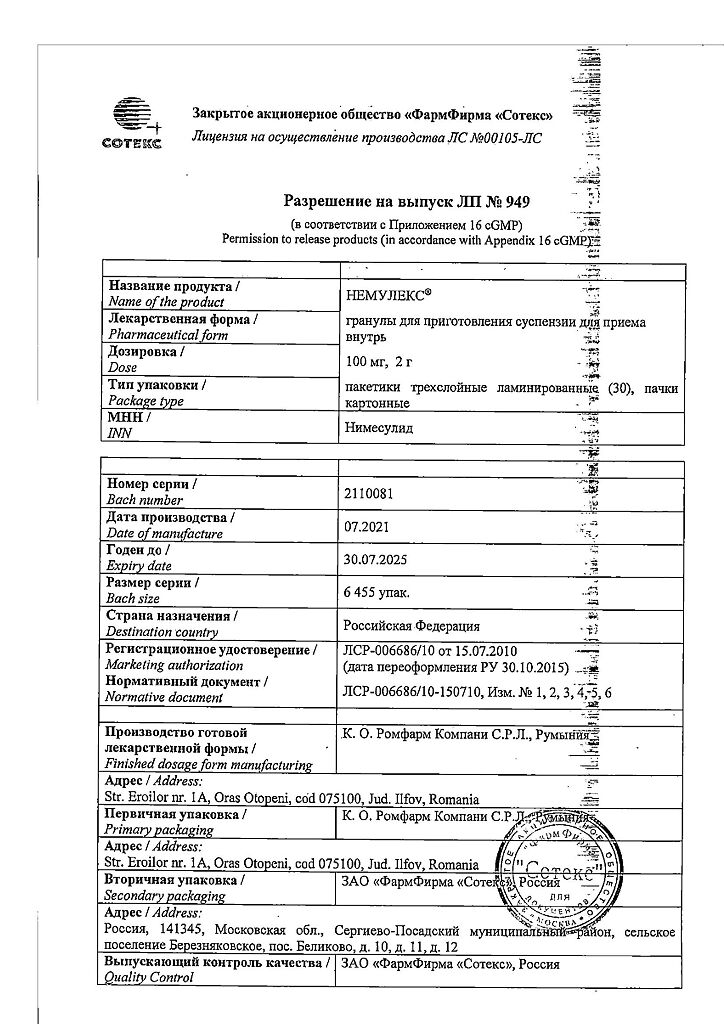
Nemulex, 100 mg/2 g 2 g 30 pcs.
€23.57 €19.64
Description
Pharmgroup:
NSAIDs.
Pharmic action:
Nemulex – NSAID, has anti-inflammatory, analgesic, antipyretic and antiaggregant effect.
Unlike other NSAIDs it selectively inhibits COX-2, inhibits synthesis of Pg in inflamed area; less pronounced inhibitory effect on COX-1 (side effects related to inhibition of Pg synthesis in healthy tissues are less frequent).
Pharmacokinetics:
Absorption when oral administration is high (eating reduces absorption rate, without affecting its degree). Time of reaching maximum concentration in blood plasma (TStah) – 1.5-2.5 h. Binding with plasma proteins is 95%, with erythrocytes – 2%, with lipoproteins -1%, with acidic alpha1-glycoprotein -1%. Changing the dose does not affect the degree of binding. Maximum concentration (Stah) is 3.5-6.5 mg/l. Volume of distribution is 0.19-0.35 l/kg. It penetrates well into the acidic environment of the inflammation focus (40%), synovial fluid (43%).
It easily penetrates through histohematic barriers. It is metabolized in liver by tissue monooxygenases. The main metabolite is 4-hydroxynimesulide (25%), which has similar pharmacological activity, but due to the reduced size of molecules it is able to rapidly diffuse through COX-2 hydrophobic channel to active binding center of methyl group. 4-Hydroxynymesulide is a water-soluble compound that does not require glutathione and phase II metabolic conjugation reactions (sulfation, glucuronidation, etc.) for elimination.
The half-life (T1/2) of nimesulide is 1.56-4.95 h, 4-hydroxynimesulide is 2.89-4.78 h. 4-hydroxynimesulide is excreted by the kidneys (65%) and with the bile (35%), is subjected to enterohepatic recirculation.
Indications
Indications
tendinitis; algodismenorrhea;
Pharmacological effect
Pharmacological effect
Pharmaceutical group:
NSAIDs.
Pharmaceutical action:
Nemulex is an NSAID that has anti-inflammatory, analgesic, antipyretic and antiplatelet effects.
Unlike other NSAIDs, it selectively suppresses COX-2 and inhibits Pg synthesis at the site of inflammation; has a less pronounced inhibitory effect on COX-1 (less likely to cause side effects associated with inhibition of Pg synthesis in healthy tissues).
Pharmacokinetics:
Absorption when taken orally is high (food intake reduces the rate of absorption without affecting its degree). Time to reach maximum concentration in blood plasma (TCmax) – 1.5-2.5 hours. Communication with plasma proteins – 95%, with erythrocytes – 2%, with lipoproteins -1%, with acidic alpha1-glycoprotein -1%. Changing the dose does not affect the degree of binding. The maximum concentration (Cmax) is 3.5-6.5 mg/l. Volume of distribution – 0.19-0.35 l/kg. Penetrates well into the acidic environment of the inflammation site (40%) and synovial fluid (43%).
Easily penetrates histohematic barriers. Metabolized in the liver by tissue monooxygenases. The main metabolite, 4-hydroxynimesulide (25%), has similar pharmacological activity, but due to a decrease in molecular size, it is able to quickly diffuse through the hydrophobic COX-2 channel to the active binding site of the methyl group. 4-hydroxynimesulide is a water-soluble compound, the elimination of which does not require glutathione and conjugation reactions of phase II metabolism (sulfation, glucuronidation, etc.).
The half-life (T1/2) of nimesulide is 1.56-4.95 hours, 4-hydroxynimesulide is 2.89-4.78 hours. 4-hydroxynimesulide is excreted by the kidneys (65%) and bile (35%), and undergoes enterohepatic recirculation.
Special instructions
Special instructions
To reduce the risk of side effects, it is necessary to use Nemulex in the minimum effective dose for the shortest duration. If the patient’s condition does not improve, treatment should be stopped. It is necessary to stop taking the drug if the temperature rises or flu-like symptoms develop while taking it.
If patients taking Nimesulide develop symptoms indicating liver damage (for example, anorexia, nausea, vomiting, abdominal pain, fatigue, dark urine), or increased levels of “liver” transaminases, the drug should be discontinued. It is not recommended to prescribe Nimesulide to such patients in the future. Gastrointestinal bleeding or ulcer/perforation of the stomach or duodenum may develop at any time while using the drug, which may not be accompanied by clinically significant symptoms (including pain). If gastrointestinal bleeding or ulcers occur, the drug should be discontinued.
If renal function deteriorates, Nemulex should be discontinued. If the drug is used for more than 2 weeks, monitoring of liver function indicators is necessary. In patients with liver cirrhosis or renal failure with hypoalbuminemia or hyperbilirubinemia, nimesulide binding is reduced.
Elderly patients most often develop side effects when taking the drug, including gastrointestinal bleeding, perforation, dysfunction of the heart, kidneys and liver. Therefore, regular clinical monitoring of the patient’s condition is recommended. The use of nimesulide may adversely affect female fertility and is not recommended for women planning pregnancy.
If, when using the drug Nemulex, adverse events occur on the part of the central nervous system and sensory organs, it is necessary to refrain from driving vehicles and engaging in activities that require increased concentration and speed of psychomotor reactions.
Active ingredient
Active ingredient
Nimesulide
Composition
Composition
Active substance:
nimesulide 100 mg;
Excipients:
macrogol cetostearate (Cremophor A25) – 100 mg;
anhydrous citric acid – 19 mg;
colloidal silicon dioxide (Aerosil) – 40 mg;
sucrose – 1721 mg;
orange flavor – 20 mg.
Contraindications
Contraindications
hemophilia and other bleeding disorders;
Side Effects
Side Effects
From the central nervous system: dizziness, feeling of fear, nervousness, nightmares, headache, drowsiness, encephalopathy (Reye’s syndrome).
Interaction
Interaction
It is not recommended to take nimesulide simultaneously with diuretics, which have a damaging effect on renal hemodynamics. Physiological concentrations of unsaturated fatty acids do not affect the binding of nimesulide to serum albumin. At therapeutic concentrations, the binding of nimesulide was not affected by warfarin, furosemide, glibenclamide, or digitoxin. In the presence of nimesulide, the free fractions of methotrexate may increase significantly.
Overdose
Overdose
Symptoms: increased blood pressure, acute renal failure, nausea, vomiting, drowsiness, apathy, gastrointestinal bleeding, respiratory depression;
Storage conditions
Storage conditions
List B.: In a dry place, protected from light, at a temperature not exceeding 25 °C.
Shelf life
Shelf life
4 years
Manufacturer
Manufacturer
K.O.Rompharm Company S.R.L./Sotex, Romania
Additional information
| Shelf life | 4 years |
|---|---|
| Conditions of storage | List B.: In a dry, light-protected place at a temperature not exceeding 25 °C. |
| Manufacturer | C.O.Rompharm Company S.R.L./Sotex, Romania |
| Medication form | granules for preparation of oral suspension |
| Brand | C.O.Rompharm Company S.R.L./Sotex |
Other forms…
Related products
Buy Nemulex, 100 mg/2 g 2 g 30 pcs. with delivery to USA, UK, Europe and over 120 other countries.