No products in the cart.
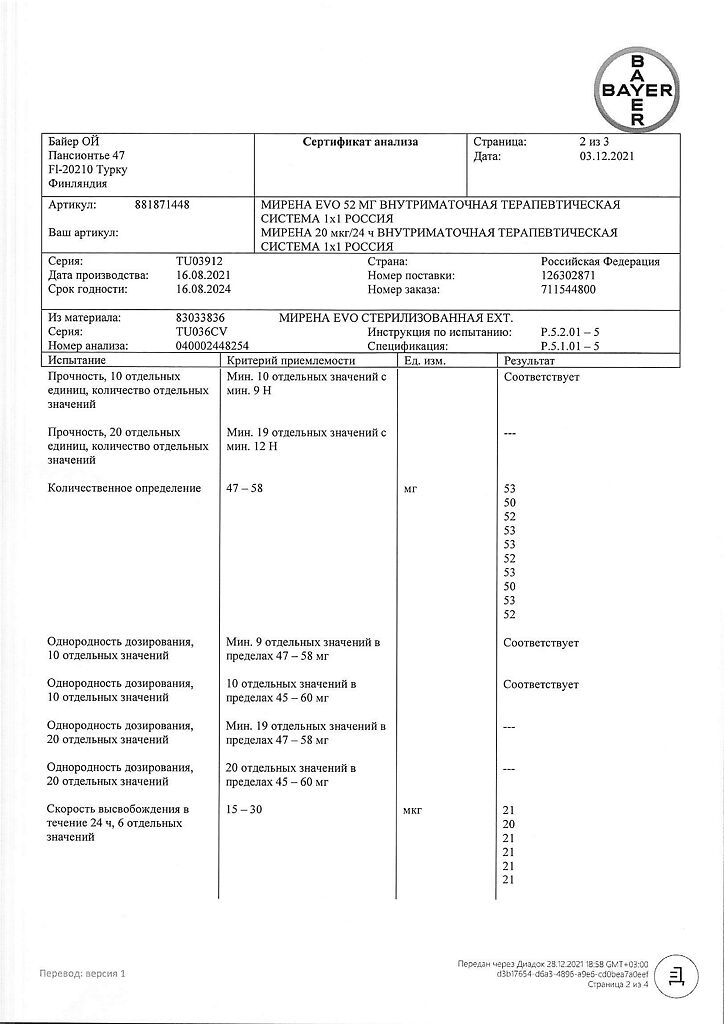
Mirena, intrauterine therapy system (coil) 20 mcg/24 h
€461736.00 €384.78
Description
Mirena®, a levonorgestrel-releasing IUD, has a predominantly local gestagenic effect. The gestagen (levonorgestrel) is released directly into the uterine cavity, allowing it to be used at an extremely low daily dose.
High concentrations of levonorgestrel in the endometrium help reduce the sensitivity of its estrogen and progesterone receptors, making the endometrium immune to estradiol and having a strong antiproliferative effect.
When using Mirena® the morphological changes of endometrium and weak local reaction to the presence of a foreign body in the uterus are observed. An increase in the viscosity of the cervical secretion prevents the penetration of sperm into the uterus. Mirena prevents fertilization by inhibiting sperm motility and function in the uterus and fallopian tubes. In some women there is also a suppression of ovulation.
Pre-use of Mirena® has no effect on fertility. Approximately 80% of women who wish to have a baby become pregnant within 12 months after removal of the IUD.
In the first months of Mirena use, there may be an initial increase in a vaginal discharge of mucus due to inhibition of endometrial proliferation. Subsequent marked suppression of endometrial proliferation leads to a reduction in the duration and volume of menstrual bleeding in women using Mirena®. Scanty bleeding often transforms into oligo- or amenorrhea.
In this case, ovarian function and plasma estradiol concentrations remain normal.
The Mirena drug® may be used to treat idiopathic menorrhagia, i.e. menorrhagia in the absence of endometrial hyperplastic processes (endometrial cancer, metastatic uterine lesions, submucosal or large interstitial myomatous nodules leading to a uterine cavity deformity, adenomyosis), endometritis, extragenital diseases and conditions accompanied by marked hypocoagulation (e.g. Willebrand disease, severe thrombocytopenia), which are symptoms of menorrhagia. After 3 months of Mirena use the menstrual blood loss in women with menorrhagia decreased by 62-94% and by 71-95% after 6 months of use.
When using Mirena® for 2 years its effectiveness (decrease of menstrual bleeding) is comparable to surgical methods of treatment (endometrial ablation or resection).
A less favorable response to treatment is possible in menorrhagia due to submycotic uterine myoma. Reducing menstrual blood loss reduces the risk of iron deficiency anemia. Mirena ® reduces symptoms of dysmenorrhea.
The efficacy of Mirena® in preventing endometrial hyperplasia during ongoing estrogen therapy was equally high with both oral and percutaneous estrogen administration.
Pharmacokinetics
Absorption. After administration of Mirena® levonorgestrel is immediately released into the uterine cavity, as evidenced by measurements of its plasma concentrations. The high local uterine exposure required for the local effects of Mirena® on the endometrium provides a high concentration gradient from endometrium to myometrium (the concentration of levonorgestrel in the endometrium exceeds its concentration in myometrium by more than 100 times) and low concentrations of levonorgestrel in plasma (the concentration of levonorgestrel in the endometrium exceeds its concentration in plasma by more than 1000 times).
The rate of levonorgestrel release into the uterine cavity in vivo is initially approximately 20 mcg/d and decreases to 10 mcg/d after 5 years.
Distribution. Levonorgestrel binds nonspecifically to plasma albumin and specifically to hSPH. About 1-2% of circulating levonorgestrel is present as a free steroid, whereas 42-62% is specifically bound to hGDP. During the use of Mirena®, the concentration of hGH is decreased. Accordingly, the hGH-bound fraction decreases during Mirena® and the free fraction increases. The average apparent Vd of levonorgestrel is about 106L.
After administration of Mirena® levonorgestrel is detectable in plasma one hour later.Cmax is reached 2 weeks after administration of Mirena®. Consistent with the decreasing rate of release, the median plasma concentration of levonorgestrel in women of reproductive age with a body weight greater than 55 kg decreases from 206 pg/ml (25-75th percentile: 151-264 pg/ml) determined after 6 months to 194 pg/ml (146-266 pg/ml) after 12 months and to 131 pg/ml (113-161 pg/ml) after 60 months.
It has been shown that body weight and plasma HSPG concentration influence systemic levonorgestrel concentrations, i.e., low body weight and/or high HSPG concentrations have higher levonorgestrel concentrations. In women of reproductive age with low body weight (37-55 kg), the median plasma concentration of levonorgestrel is about 1.5 times higher.
In postmenopausal women using Mirena® simultaneously with intravaginal or transdermal estrogen administration, the median plasma levonorgestrel concentration decreases from 257 pg/mL (25-75th percentile: 186-326 pg/mL) determined after 12 months to 149 pg/mL (122-180 pg/mL) after 60 months.
When Mirena ® is used concomitantly with oral estrogen administration, plasma levonorgestrel concentrations determined after 12 months increase to approximately 478 pg/mL (25th-75th percentile: 341-655 pg/mL) due to induction of hCG synthesis.
Biotransformation. Levonorgestrel is largely metabolized. The main metabolites in the blood plasma are unconjugated and conjugated forms of 3α, 5β-tetrahydrolevonorgestrel.
Based on in vitro and in vivo studies, the main isoenzyme involved in the metabolism of levonorgestrel is CYP3A4. The isoenzymes CYP2E1, CYP2C19 and CYP2C9 may also be involved in levonorgestrel metabolism, but to a lesser extent.
Elimination. Total blood plasma clearance of levonorgestrel is approximately 1 ml/min/kg. Unchanged levonorgestrel is excreted only in trace amounts. Metabolites are excreted through the intestine and the kidneys with an excretion rate of approximately 1.77.T1/2 in the terminal phase, represented mainly by metabolites, is about one day.
Linearity/nonlinearity. The pharmacokinetics of levonorgestrel depend on the concentration of hGH, which, in turn, is influenced by estrogens and androgens. With Mirena®, a decrease of approximately 30% in mean hGHG concentrations was observed, which was accompanied by a decrease in plasma levonorgestrel concentrations.
This indicates that levonorgestrel pharmacokinetics is non-linear over time. Given the predominantly local action of Mirena®, the effect of changes in systemic levonorgestrel concentrations on the efficacy of Mirena® is unlikely.
Indications
Indications
Contraception;
idiopathic menorrhagia;
prevention of endometrial hyperplasia during estrogen replacement therapy.
Pharmacological effect
Pharmacological effect
The drug Mirena®, an IUD that releases levonorgestrel, has a mainly local gestagenic effect. Progestin (levonorgestrel) is released directly into the uterine cavity, which allows it to be used in an extremely low daily dose.
High concentrations of levonorgestrel in the endometrium help to reduce the sensitivity of its estrogen and progesterone receptors, making the endometrium resistant to estradiol and having a strong antiproliferative effect.
When using Mirena®, morphological changes in the endometrium and a weak local reaction to the presence of a foreign body in the uterus are observed. An increase in the viscosity of the cervical secretion prevents the penetration of sperm into the uterus. Mirena® prevents fertilization due to inhibition of sperm motility and function in the uterus and fallopian tubes. In some women, ovulation is also suppressed.
Previous use of Mirena® does not affect reproductive function. Approximately 80% of women who want to have a child become pregnant within 12 months after removal of the IUD.
In the first months of using Mirena®, due to inhibition of the process of endometrial proliferation, an initial increase in spotting and spotting from the vagina may be observed. Following this, a pronounced suppression of endometrial proliferation leads to a decrease in the duration and volume of menstrual bleeding in women using Mirena®. Scanty bleeding often transforms into oligo- or amenorrhea.
At the same time, ovarian function and the concentration of estradiol in the blood plasma remain normal.
Mirena® can be used to treat idiopathic menorrhagia, i.e. menorrhagia in the absence of hyperplastic processes in the endometrium (endometrial cancer, metastatic lesions of the uterus, submucous or large interstitial myomatous node leading to deformation of the uterine cavity, adenomyosis), endometritis, extragenital diseases and conditions accompanied by severe hypocoagulation (for example, von Willebrand disease, severe thrombocytopenia), the symptoms of which are menorrhagia. After 3 months of using Mirena®, menstrual blood loss in women with menorrhagia is reduced by 62–94% and by 71–95% after 6 months of use.
When using Mirena® for 2 years, the effectiveness of the drug (reduction of menstrual blood loss) is comparable to surgical treatment methods (ablation or resection of the endometrium).
A less favorable response to treatment is possible with menorrhagia caused by submycotic uterine fibroids. Reducing menstrual blood loss reduces the risk of iron deficiency anemia. Mirena® reduces the severity of dysmenorrhea symptoms.
The effectiveness of Mirena® in preventing endometrial hyperplasia during chronic estrogen therapy was equally high with both oral and transdermal estrogen administration.
Pharmacokinetics
Absorption. After administration of the drug Mirena®, levonorgestrel begins to be immediately released into the uterine cavity, as evidenced by measurements of its concentration in the blood plasma. The high local exposure of the drug in the uterine cavity, necessary for the local effect of Mirena® on the endometrium, provides a high concentration gradient in the direction from the endometrium to the myometrium (the concentration of levonorgestrel in the endometrium exceeds its concentration in the myometrium by more than 100 times) and low concentrations of levonorgestrel in the blood plasma (the concentration of levonorgestrel in the endometrium exceeds its concentration in the blood plasma by more than 1000 times).
The rate of release of levonorgestrel into the uterine cavity in vivo is initially approximately 20 mcg/day, and after 5 years it decreases to 10 mcg/day.
Distribution. Levonorgestrel binds nonspecifically to plasma albumin and specifically to SHBG. About 1–2% of circulating levonorgestrel is present as the free steroid, whereas 42–62% is specifically bound to SHBG. During use of the drug Mirena®, the concentration of SHBG decreases. Accordingly, the fraction associated with SHBG during the period of use of Mirena® decreases, and the free fraction increases. The average apparent Vd of levonorgestrel is about 106 L.
After administration of the drug Mirena®, levonorgestrel is detected in the blood plasma after an hour. Cmax is achieved 2 weeks after administration of the drug Mirena®. In accordance with the decreasing release rate, the median plasma concentration of levonorgestrel in women of reproductive age with a body weight above 55 kg decreases from 206 pg / ml (25-75th percentile: 151-264 pg / ml), determined after 6 months, to 194 pg / ml (146-266 pg / ml) – after 12 months and before 131 pg/ml (113–161 pg/ml) – after 60 months.
Body weight and plasma SHBG concentrations have been shown to influence systemic levonorgestrel concentrations, i.e. with low body weight and/or high SHBG concentrations, levonorgestrel concentrations are higher. In women of reproductive age with low body weight (37–55 kg), the median plasma concentration of levonorgestrel is approximately 1.5 times higher.
In postmenopausal women using Mirena® simultaneously with the use of estrogens intravaginally or transdermally, the median concentration of levonorgestrel in the blood plasma decreases from 257 pg/ml (25–75th percentile: 186–326 pg/ml), determined after 12 months, to 149 pg/ml (122–180 pg/ml) – after 60 months.
When using Mirena® simultaneously with oral administration of estrogens, the concentration of levonorgestrel in the blood plasma, determined after 12 months, increases to approximately 478 pg/ml (25–75th percentile: 341–655 pg/ml), which is due to the induction of SHBG synthesis.
Biotransformation. Levonorgestrel is extensively metabolized. The main metabolites in blood plasma are unconjugated and conjugated forms of 3α, 5β-tetrahydrolevonorgestrel.
Based on the results of in vitro and in vivo studies, the main isoenzyme involved in the metabolism of levonorgestrel is CYP3A4. The isoenzymes CYP2E1, CYP2C19 and CYP2C9 may also be involved in the metabolism of levonorgestrel, but to a lesser extent.
Elimination. The total plasma clearance of levonorgestrel is approximately 1 ml/min/kg. Unchanged levonorgestrel is excreted only in trace amounts. Metabolites are excreted through the intestines and kidneys with an excretion coefficient of approximately 1.77. T1/2 in the terminal phase, represented mainly by metabolites, is about a day.
Linearity/nonlinearity. The pharmacokinetics of levonorgestrel depends on the concentration of SHBG, which, in turn, is influenced by estrogens and androgens. When using the drug Mirena®, a decrease in the average concentration of SHBG by approximately 30% was observed, which was accompanied by a decrease in the concentration of levonorgestrel in the blood plasma.
This indicates the nonlinearity of levonorgestrel pharmacokinetics over time. Given the predominantly local effect of Mirena®, the effect of changes in systemic concentrations of levonorgestrel on the effectiveness of Mirena® is unlikely.
Special instructions
Special instructions
Before installing Mirena®, pathological processes in the endometrium should be excluded, since irregular bleeding/spotting is often observed in the first months of its use. Pathological processes in the endometrium should also be excluded if bleeding occurs after starting estrogen replacement therapy in a woman who continues to use Mirena®, previously prescribed for contraception. Appropriate diagnostic measures must also be taken when irregular bleeding develops during long-term treatment.
Mirena® is not used for postcoital contraception.
Mirena® should be used with caution in women with congenital or acquired valvular heart disease, keeping in mind the risk of septic endocarditis. When inserting or removing an IUD, these patients should be given prophylactic antibiotics.
Levonorgestrel in low doses can affect glucose tolerance, and therefore its blood levels should be regularly checked in women with diabetes mellitus using Mirena®. As a rule, no dose adjustment of hypoglycemic drugs is required.
Some manifestations of polyposis or endometrial cancer may be masked by irregular bleeding. In such cases, additional examination is necessary to clarify the diagnosis.
Mirena® is not a first choice drug for young women who have never been pregnant, or for postmenopausal women with severe uterine atrophy.
Available data indicate that the use of Mirena® does not increase the risk of developing breast cancer in postmenopausal women under the age of 50 years. Due to the limited data obtained during the study of Mirena® for the indication “prevention of endometrial hyperplasia during estrogen replacement therapy,” the risk of breast cancer when using Mirena® for this indication cannot be confirmed or refuted.
Oligo- and amenorrhea. Oligo- and amenorrhea in women of fertile age develops gradually, in approximately 57 and 16% of cases by the end of the first year of using Mirena®. If menstruation is absent within 6 weeks after the start of the last menstrual period, pregnancy should be excluded. Repeated pregnancy tests for amenorrhea are not necessary if there are no other signs of pregnancy.
When Mirena® is used in combination with continuous estrogen replacement therapy, most women gradually develop amenorrhea over the first year.
Inflammatory diseases of the pelvic organs (PID). The guide tube helps protect Mirena® from infection during insertion, and the Mirena® delivery device is specially designed to minimize the risk of infection.
PID in patients using IUDs is often classified as a sexually transmitted disease. Having multiple sexual partners has been found to be a risk factor for PID. PID can have serious consequences: it can impair fertility and increase the risk of ectopic pregnancy.
As with other gynecologic or surgical procedures, severe infection or sepsis (including group A streptococcal sepsis) may develop after insertion of an IUD, although this is extremely rare. For recurrent endometritis or PID, as well as for severe or acute infections that are resistant to treatment for several days, Mirena should be removed. If a woman has persistent pain in the lower abdomen, chills, fever, pain associated with sexual intercourse (dyspareunia), prolonged or heavy spotting/bleeding from the vagina, or a change in the nature of vaginal discharge, you should consult your doctor immediately.
Severe pain or fever that occurs soon after insertion of an IUD may indicate a serious infection that must be treated immediately. Even in cases where only individual symptoms indicate the possibility of infection, bacteriological examination and monitoring are indicated.
Expulsion. Possible signs of partial or complete expulsion of any IUD are bleeding and pain. Contractions of the uterine muscles during menstruation sometimes lead to the displacement of the IUD or even its expulsion from the uterus, which leads to the cessation of contraceptive action. Partial expulsion may reduce the effectiveness of Mirena®. Since Mirena® reduces menstrual blood loss, an increase in blood loss may indicate expulsion of the IUD. A woman is advised to check the threads with her fingers, for example while taking a shower. If a woman shows signs of the IUD being dislodged or falling out, or cannot feel the threads, she should avoid sexual intercourse or use other methods of contraception, and consult a doctor as soon as possible.
If the position in the uterine cavity is incorrect, the IUD must be removed. A new system may be installed at this time.
It is necessary to explain to the woman how to check the threads of Mirena®.
Perforation and penetration. Perforation or penetration of the body or cervix of the IUD occurs rarely, mainly during insertion, and may reduce the effectiveness of Mirena®. In these cases, the system should be removed. If there is a delay in diagnosing perforation and IUD migration, complications such as adhesions, peritonitis, intestinal obstruction, intestinal perforation, abscesses or erosions of adjacent internal organs may occur. The risk of uterine perforation is increased in women who are breastfeeding. There may be an increased risk of perforation when installing an IUD after childbirth and in women with a fixed uterine flexion.
Ectopic pregnancy. Women with a history of ectopic (ectopic) pregnancy, tubal surgery or pelvic infection are at higher risk of ectopic pregnancy. The possibility of ectopic pregnancy should be considered in the case of lower abdominal pain, especially if it is combined with cessation of menstruation, or when a woman with amenorrhea begins to bleed. The incidence of ectopic pregnancy when using Mirena® is approximately 0.1% per year. The absolute risk of ectopic pregnancy in women using Mirena® is low. However, if a woman with installed Mirena® becomes pregnant, the relative likelihood of ectopic pregnancy is higher.
Loss of threads. If, during a gynecological examination, the threads for removing the IUD cannot be detected in the cervical area, it is necessary to exclude pregnancy. The threads can be drawn into the uterine cavity or cervical canal and become visible again after the next menstruation. If pregnancy is ruled out, the location of the threads can usually be determined by careful probing with an appropriate instrument. If the threads cannot be detected, the IUD may have expelled from the uterine cavity. Ultrasound can be used to determine the correct placement of the system. If it is unavailable or unsuccessful, X-ray examination is used to determine the localization of Mirena®.
Ovarian cysts. Since the contraceptive effect of Mirena® is mainly due to its local action, women of fertile age usually experience ovulatory cycles with follicle rupture. Sometimes follicular atresia is delayed and follicular development may continue. Such enlarged follicles cannot be clinically distinguished from ovarian cysts. Ovarian cysts were reported as an adverse reaction in approximately 7% of women using Mirena®. In most cases, these follicles do not cause any symptoms, although sometimes they are accompanied by pain in the lower abdomen or pain during intercourse. As a rule, ovarian cysts disappear on their own within 2–3 months of observation. If this does not happen, it is recommended to continue monitoring with ultrasound, as well as therapeutic and diagnostic measures. In rare cases, it is necessary to resort to surgical intervention.
The use of Mirena® in combination with estrogen replacement therapy. When using Mirena® in combination with estrogens, it is necessary to additionally take into account the information specified in the instructions for use of the corresponding estrogen.
Excipients contained in Mirena®. The T-shaped base of Mirena® contains barium sulfate, which becomes visible during X-ray examination.
It must be borne in mind that Mirena® does not protect against HIV infection and other sexually transmitted diseases.
Additional information for patients
Regular inspections. The doctor should examine the patient 4–12 weeks after insertion of the IUD; thereafter, regular medical examinations are required at least once a year.
You should consult your doctor as soon as possible in the following cases:
– the patient stopped feeling the threads in the vagina;
– the patient can feel the lower end of the system;
– the patient assumes that she is pregnant;
– the patient feels constant abdominal pain, fever, or notes a change in the usual nature of vaginal discharge;
– the patient or her partner feels pain during sexual intercourse;
– the patient noted sudden changes in the menstrual cycle (for example, periods were scanty or absent, and then constant bleeding or pain appeared, or periods became excessively heavy);
– the patient has other medical problems, such as migraine-type headache or severe recurring headache, sudden visual disturbances, jaundice, increased blood pressure, or any other diseases and conditions listed in the sections “Contraindications” and “Precautions”.
What to do if the patient wants to become pregnant or remove Mirena® for other reasons. The doctor can easily remove the IUD at any time, after which pregnancy becomes possible. Removal is usually painless. After removing Mirena®, reproductive function is restored.
When pregnancy is undesirable, Mirena® should be removed no later than the 7th day of the menstrual cycle. If Mirena® is removed later than the 7th day of the cycle, you should use barrier methods of contraception (for example, a condom) for at least 7 days before its removal. If there is no menstruation while using Mirena®, 7 days before removal of the IUD, you should start using barrier contraceptive methods and continue their use until menstruation resumes. You can also insert a new IUD immediately after removing the previous one; in this case, no additional measures to prevent pregnancy are required.
How long can you use Mirena®? Mirena® provides protection against pregnancy for 5 years, after which it should be removed. You can install a new IUD after removing the old one.
Restoring the ability to conceive (is it possible to get pregnant after stopping the use of Mirena®). Once Mirena® is removed, it no longer affects normal fertility. Pregnancy can occur during the 1st menstrual cycle after removing Mirena®.
Effect on the menstrual cycle (can Mirena® affect the menstrual cycle). Mirena® affects the menstrual cycle. Under its influence, menstruation may change and acquire the character of spotting, become longer or shorter, occur with more or less bleeding than usual, or stop altogether. In the first 3–6 months after installation of Mirena®, many women experience, in addition to their normal menstruation, frequent spotting or scanty bleeding. In some cases, very heavy or prolonged bleeding is observed during this period. If the patient experiences these symptoms, especially if they do not disappear, the doctor should be informed. It is most likely that when using Mirena®, the number of days of bleeding and the amount of blood lost will gradually decrease with each month. Some women eventually find that their periods have stopped completely. Since the amount of blood lost during menstruation usually decreases when using Mirena®, most women experience an increase in hemoglobin levels in the blood.
After the system is removed, the menstrual cycle returns to normal.
Lack of menstruation. Is it normal not to have menstruation when using Mirena®? If, after installing Mirena®, the patient noted the disappearance of menstruation, this is due to the effect of the hormone on the uterine mucosa. There is no monthly thickening of the mucous membrane, therefore, it is not rejected during menstruation. This does not necessarily mean that the patient has reached menopause or is pregnant. The concentration of own hormones in the blood plasma remains normal. In fact, not having periods can be a big benefit to a woman’s comfort.
How can you find out about pregnancy. Pregnancy in women using Mirena®, even if they do not menstruate, is unlikely. If the patient has not had a menstrual period for 6 weeks and is concerned, a pregnancy test should be performed. If the result is negative, there is no need to do additional tests unless there are other signs of pregnancy, such as nausea, fatigue or breast tenderness.
Can Mirena® cause pain or discomfort. Some women experience pain (similar to menstrual cramps) in the first 2-3 weeks after having an IUD inserted. If the patient experiences severe pain or if the pain continues for more than 3 weeks after installation of the system, you should contact your doctor or the medical institution where Mirena® was installed.
Does Mirena® affect sexual intercourse? Neither the patient nor her partner should feel the IUD during sexual intercourse. Otherwise, sexual intercourse should be avoided until the doctor is sure that the system is in the correct position.
How much time should pass between the installation of Mirena® and sexual intercourse. It is best to abstain from sexual intercourse for 24 hours after inserting Mirena® into the uterus to give the body a rest. However, Mirena® has a contraceptive effect from the moment of installation.
Can I use tampons? It is recommended to use sanitary pads. If the patient uses tampons, they should be changed very carefully so as not to pull out the Mirena® threads.
What happens if Mirena® spontaneously leaves the uterine cavity. Very rarely, expulsion of the IUD from the uterine cavity may occur during menstruation. An unusual increase in blood loss during menstrual bleeding may mean that Mirena® has passed through the vagina. Partial expulsion of the IUD from the uterine cavity into the vagina is also possible (the patient and her partner may notice this during sexual intercourse). If Mirena® is completely or partially removed from the uterus, its contraceptive effect ceases immediately.
What signs can be used to judge that Mirena® is in place. The patient can independently check whether the Mirena® threads are in place after menstruation has ended. After your period ends, carefully insert your finger into the vagina and feel the threads at the end, near the entrance to the uterus (cervix). You should not pull the threads, because… You may accidentally pull Mirena® out of the uterus. If the patient cannot feel the threads, she should consult a doctor.
Impact on the ability to drive vehicles and machinery. Not observed.
Active ingredient
Active ingredient
Levonorgestrel
Composition
Composition
Active ingredient:
levonorgestrel 52 mg;
Excipients:
polydimethylsiloxane elastomer – 52 mg
Pregnancy
Pregnancy
Pregnancy. The use of Mirena® is contraindicated during pregnancy or suspected pregnancy.
Pregnancy in women who have Mirena® installed is extremely rare. But if the IUD falls out of the uterine cavity, the woman is no longer protected from pregnancy and should use other methods of contraception before consulting a doctor.
While using Mirena®, some women experience no menstrual bleeding. The absence of menstruation does not necessarily indicate pregnancy. If a woman does not have menstruation, and at the same time there are other signs of pregnancy (nausea, fatigue, breast tenderness), then it is necessary to consult a doctor for examination and a pregnancy test.
If pregnancy occurs in a woman while using Mirena®, it is recommended to remove the IUD, because Any intrauterine contraceptive device left in situ increases the risk of spontaneous abortion and premature birth. Removing Mirena® or probing the uterus may lead to spontaneous abortion.
If careful removal of the intrauterine contraceptive device is not possible, the feasibility of medical abortion should be discussed.
If a woman wants to continue the pregnancy and the IUD cannot be removed, the patient should be informed about the risks, in particular about the possible risk of septic abortion in the second trimester of pregnancy, postpartum purulent-septic diseases that can be complicated by sepsis, septic shock and death, as well as the possible consequences of premature birth for the child. In such cases, the course of pregnancy should be carefully monitored.
It is necessary to exclude ectopic pregnancy. The woman should be explained that she must report all symptoms that suggest a pregnancy complication, in particular the appearance of cramping pain in the lower abdomen, bleeding or spotting from the vagina, and increased body temperature.
The hormone contained in Mirena® is released into the uterine cavity. This means that the fetus is exposed to a relatively high local concentration of the hormone, although the hormone enters it in small quantities through the blood and placenta.
Due to the intrauterine use and local action of the hormone, it is necessary to take into account the possibility of a virilizing effect on the fetus. Due to the high contraceptive effectiveness of Mirena®, clinical experience regarding pregnancy outcomes with its use is limited.
However, the woman should be informed that at the moment there is no evidence of birth defects caused by the use of Mirena® in cases of pregnancy continuing until childbirth without removal of the IUD.
Breastfeeding period. Breastfeeding a child while using Mirena® is not contraindicated. About 0.1% of the dose of levonorgestrel can enter the child’s body during breastfeeding. However, it is unlikely to pose a risk to the baby at doses released into the uterine cavity after Mirena® insertion.
It is believed that the use of Mirena® 6 weeks after birth does not have a harmful effect on the growth and development of the child. Monotherapy with gestagens does not affect the quantity and quality of breast milk. Rare cases of uterine bleeding have been reported in women using Mirena® during lactation.
After Mirena® is removed, fertility is restored in women.
Contraindications
Contraindications
Pregnancy or suspicion of it;
inflammatory diseases of the pelvic organs (including recurrent);
infections of the lower genitourinary tract;
postpartum endometritis;
septic abortion within the last 3 months;
cervicitis;
diseases accompanied by increased susceptibility to infections;
cervical dysplasia;
malignant neoplasms of the uterus or cervix;
progestogen-dependent tumors, incl. breast cancer;
pathological uterine bleeding of unknown etiology;
congenital and acquired anomalies of the uterus, incl. fibroids leading to deformation of the uterine cavity;
acute liver diseases, liver tumors;
age over 65 years (studies have not been conducted in this category of patients);
hypersensitivity to the components of the drug.
The drug should be used with caution and only after consultation with a specialist for the conditions listed below. The advisability of removing the system should be discussed if any of the following conditions are present or first occur:
migraine, focal migraine with asymmetric vision loss or other symptoms indicating transient cerebral ischemia;
unusually severe headache;
jaundice;
severe arterial hypertension;
severe circulatory disorders, incl. stroke and myocardial infarction;
congenital heart defects or heart valve disease (due to the risk of developing septic endocarditis);
diabetes mellitus
Side Effects
Side Effects
From the immune system: frequency unknown – hypersensitivity to the drug or components of the drug, including rash, urticaria and angioedema.
From the mental side: often – depressed mood, depression.
From the nervous system: very often – headache; often – migraine.
From the digestive system: very often – abdominal pain/pain in the pelvic area; often – nausea.
From the skin and subcutaneous tissues: often – acne, hirsutism; uncommon – alopecia, itching, eczema.
From the musculoskeletal system: often – back pain.
From the reproductive system and mammary glands: very often – changes in the volume of blood loss, including an increase and decrease in the intensity of bleeding, spotting, oligomenorrhea, amenorrhea, vulvovaginitis, discharge from the genital tract; often – infections of the pelvic organs, ovarian cysts, dysmenorrhea, pain in the mammary glands, engorgement of the mammary glands, expulsion of the IUD (full or partial); rarely – uterine perforation (including penetration).
From the cardiovascular system: frequency unknown – increased blood pressure.
Interaction
Interaction
The metabolism of gestagens may be enhanced by the simultaneous use of substances that are enzyme inducers, especially cytochrome P450 isoenzymes involved in the metabolism of drugs, such as anticonvulsants (for example, phenobarbital, phenytoin, carbamazepine) and drugs for the treatment of infections (for example, rifampicin, rifabutin, nevirapine, efavirenz).
The effect of these drugs on the effectiveness of Mirena® is unknown, but it is believed that it is insignificant since Mirena® has mainly a local effect.
Storage conditions
Storage conditions
In a place protected from light, at a temperature not exceeding 30 °C
Shelf life
Shelf life
3 years
Manufacturer
Manufacturer
Bayer Oy, Finland
Additional information
| Shelf life | 3 years |
|---|---|
| Conditions of storage | In a light-protected place, at a temperature not exceeding 30 °C |
| Manufacturer | Bayer Oy, Finland |
| Medication form | intrauterine therapy system |
| Brand | Bayer Oy |
Related products
Buy Mirena, intrauterine therapy system (coil) 20 mcg/24 h with delivery to USA, UK, Europe and over 120 other countries.