No products in the cart.
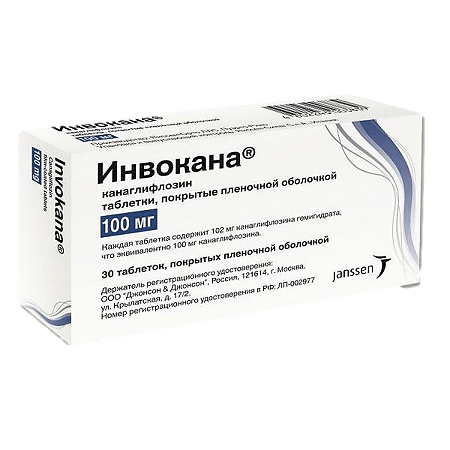
Invokana, 100 mg 30 pcs.
€92.15 €76.79
Description
The glucose transporter 2 (NZTG2) in the proximal renal tubules is responsible for most of the reabsorption of filtered glucose from the tubule lumen.It is proven that diabetic patients have increased renal glucose reabsorption, may be one of the causes of consistently elevated blood glucose concentrations.
Canagliflozin is an active oral NZTG2 inhibitor. By inhibiting NZTG2, canagliflozin reduces filtered glucose reabsorption and lowers the renal threshold for glucose (RTH ), increasing urinary glucose excretion (EG) and reducing plasma glucose concentration through this insulin-independent mechanism in patients with type 2 diabetes. Increased glucose excretion with urine when NZTG2 is inhibited also leads to osmotic diuresis, with diuretic effect leading to lower systolic blood pressure; calorie loss and weight loss are the result of increased glucose excretion with urine, which has been demonstrated in studies in patients with type 2 diabetes.
The ability of canagliflozin to increase EH with urine by directly reducing plasma glucose levels is independent of insulin. In clinical trials, canagliflozin has been observed to improve the homeostatic model of β-cell function assessment (NOMA of β-cells) and improve insulin secretion by beta-cells in response to a mixed food trial.
In phase 3 studies, administration of a 300-mg dose of canagliflozinu before meals reduced postprandial glycemia more than administration of a 100-mg dose. This effect of the 300 mg dose of canagliflozinu may be due in part to local inhibition of intestinal NZTG1 (an important glucose transporter in the gut) due to temporarily high concentrations of canagliflozinu in the intestine before absorption of the drug (canagliflozin is a weak NZTG1 inhibitor). Studies have not demonstrated glucose malabsorption when canagliflozinu is used.
Indications
Indications
Type 2 diabetes mellitus in adults in combination with diet and exercise to improve glycemic control (as monotherapy, as part of combination therapy with other hypoglycemic drugs, including insulin).
Pharmacological effect
Pharmacological effect
Sodium glucose transporter 2 (NSTG2) in the proximal renal tubule is responsible for the majority of filtered glucose reabsorption from the tubular lumen. Diabetic patients have been shown to have increased renal glucose reabsorption, which may be one cause of persistently elevated blood glucose concentrations.
Canagliflozin is an active orally administered NSTG2 inhibitor. By inhibiting NSTG2, canagliflozin reduces the reabsorption of filtered glucose and lowers the renal threshold for glucose (RTG), increasing urinary excretion of glucose (EG) and decreasing plasma glucose concentrations through this non-insulin-independent mechanism in patients with type 2 diabetes. Increased urinary glucose excretion due to NSTH2 inhibition also leads to osmotic diuresis, with the diuretic effect leading to a decrease in systolic blood pressure; Calorie loss and weight loss result from increased urinary glucose excretion, which has been demonstrated in studies in patients with type 2 diabetes.
The ability of canagliflozin to increase urinary EG by directly reducing plasma glucose levels is independent of insulin. In clinical trials with canagliflozin, improvements in homeostatic beta cell function assessment (HOMA beta cell function) and beta cell insulin secretion in response to a mixed meal challenge were observed.
In phase 3 studies, canagliflozin 300 mg before meals reduced postprandial glycemia more than 100 mg. This effect of the 300 mg canagliflozin dose may be due in part to local inhibition of intestinal NSTG1 (an important intestinal glucose transporter) due to transient high concentrations of canagliflozin in the intestinal cavity prior to drug absorption (canagliflozin is a weak inhibitor of NSTG1). Studies have not demonstrated glucose malabsorption with canagliflozin.
Special instructions
Special instructions
General
The use of canagliflozin in patients with type 1 diabetes mellitus has not been studied, and therefore its use in this category of patients is contraindicated.
The use of canagliflozin is contraindicated in diabetic ketoacidosis, in patients with end-stage chronic renal failure (ESRD) or in patients on dialysis, as such treatment will not be effective in these clinical situations.
Carcinogenicity and mutagenicity
Preclinical data do not demonstrate a specific hazard to humans based on pharmacological safety, repeated dose toxicity, genotoxicity, reproductive and developmental toxicity studies.
Fertility
The effect of canagliflozin on fertility in humans has not been studied. No effects on fertility were observed in animal studies.
Hypoglycemia when used simultaneously with other hypoglycemic drugs
It has been shown that the use of canagliflozin as monotherapy or as an adjunct to hypoglycemic agents (the use of which is not accompanied by the development of hypoglycemia) rarely led to the development of hypoglycemia. It is known that insulin and hypoglycemic agents that enhance its secretion (for example, sulfonylurea derivatives) cause the development of hypoglycemia. When canagliflozin was used as an adjunct to insulin or insulin secretagogues (eg, sulfonylureas), the incidence of hypoglycemia was higher than when using placebo.
Therefore, to reduce the risk of hypoglycemia, a reduction in the dose of insulin or insulin secretagogues is recommended.
Decreased intravascular volume
Canagliflozin has a diuretic effect by increasing renal excretion of glucose, causing osmotic diuresis, which can lead to a decrease in intravascular volume. In clinical studies of canagliflozin, an increase in the incidence of intravascular volume depletion-related adverse reactions (eg, postural dizziness, orthostatic hypotension, or hypotension) was observed more frequently during the first three months of canagliflozin 300 mg. Patients who may be more susceptible to intravascular volume depletion-related adverse reactions include patients receiving loop diuretics, patients with moderate renal impairment, and patients ≥75 years of age.
Patients should report clinical symptoms of decreased intravascular volume. These adverse reactions often led to discontinuation of canagliflozin use and were often corrected by changing the regimen of antihypertensive drugs (including diuretics) while taking canagliflozin. In patients with intravascular volume depletion, this condition should be corrected before initiating canagliflozin treatment.
During the first six weeks of canagliflozin treatment, there were cases of a small mean decrease in estimated glomerular filtration rate (eGFR) due to a decrease in intravascular volume. Patients predisposed to greater intravascular volume loss, as noted above, sometimes experienced greater reductions in GFR (>30%), which subsequently resolved and occasionally required interruptions in canagliflozin treatment.
Fungal infections of the genital organs
In clinical studies, the incidence of vulvovaginal candidiasis (including vulvovaginitis and vulvovaginal fungal infections) was higher in women receiving canagliflozin compared with placebo. Patients with a history of vulvovaginal candidiasis who were treated with canagliflozin were more likely to develop this infection. Among patients treated with canagliflozin, 2.3% experienced more than one episode of infection. Most reports of vulvovaginal candidiasis occurred in the first four months after initiation of canagliflozin treatment. 0.7% of all patients discontinued canagliflozin due to vulvovaginal candidiasis. The diagnosis of vulvovaginal candidiasis has typically been made based on symptoms alone. Clinical studies have demonstrated the effectiveness of topical or oral antifungal treatment prescribed by a physician or self-administered during ongoing therapy with canagliflozin.
In clinical trials, candidal balanitis or balanoposthitis was observed more frequently in patients treated with canagliflozin 100 mg and 300 mg compared to placebo. Balanitis or balanoposthitis occurred primarily in men who were not circumcised and occurred more frequently in men with a history of balanitis or balanoposthitis. 0.9% of patients treated with canagliflozin experienced more than one episode of infection. 0.5% of all patients discontinued canagliflozin due to candidal balanitis or balanoposthitis. In clinical studies, most cases of infection were treated with topical antifungals prescribed by a physician or self-administered during ongoing canagliflozin therapy. Rare cases of phimosis have been reported, and circumcision surgery has sometimes been performed.
Bone fractures
In a cardiovascular outcomes study of 4327 patients with known cardiovascular disease or high cardiovascular risk, the incidence of bone fractures was 16.3, 16.4, and 10.8 per 1,000 patient-years of Invocana 100 mg and 300 mg and placebo, respectively. An imbalance in fracture prevalence occurred in the first 26 weeks of therapy.
In a pooled analysis of other studies of Invokana®, which included approximately 5800 patients with diabetes mellitus from the general population, the prevalence of bone fractures was 10.8, 12.0 and 14.1 per 1000 patient-years of use of Invokana® 100 mg and 300 mg and placebo, respectively.
Over 104 weeks of treatment, canagliflozin had no negative effect on bone mineral density.
Impact on the ability to drive vehicles and machinery
Canagliflozin has not been found to affect the ability to drive or operate machines. However, patients should be aware of the risk of hypoglycemia when using canagliflozin as an adjunct to insulin or insulin secretagogue therapy, the increased risk of adverse reactions associated with intravascular volume depletion (postural dizziness), and the deterioration of the ability to drive vehicles and machines when adverse reactions occur.
Active ingredient
Active ingredient
Canagliflozin
Composition
Composition
Pills
Active ingredient:
102.0 mg canagliflozin hemihydrate, equivalent to 100.0 mg canagliflozin.
Excipients (core):
microcrystalline cellulose 39.26 mg,
lactose anhydrous 39.26 mg,
croscarmellose sodium 12.00 mg,
hyprolose 6.00 mg,
magnesium stearate 1.48 mg.
Contraindications
Contraindications
hypersensitivity to canagliflozin or any excipient of the drug;
diabetes mellitus type 1;
diabetic ketoacidosis;
severe renal failure;
severe liver failure;
pregnancy;
breastfeeding period;
children under 18 years of age.
Side Effects
Side Effects
Data on adverse reactions observed during clinical trials1 of canagliflozin with a frequency of ≥2% are systematized for each organ system depending on the frequency of occurrence using the following classification: very common (≥1/10), common (≥1/100,
Gastrointestinal disorders:
Frequent: constipation, thirst2, dry mouth.
Renal and urinary tract disorders:
Frequent: polyuria and pollakiuria3, urinary urgency, urinary tract infection4, urosepsis.
Disorders of the genital organs and breast:
Frequent: balanitis and balanoposthitis5, vulvovaginal candidiasis6, vaginal infections.
1 Includes monotherapy and add-on therapy with metformin, metformin and sulfonylureas, as well as metformin and pioglitazone.
2 The category “thirst” includes the term “thirst”, and the term “polydipsia” also falls into this category.
3 The category “polyuria or pollakiuria” includes the terms “polyuria”; this category also includes the terms “increased volume of urine excreted”, “nocturia”.
4 The category “urinary tract infections” includes the term “urinary tract infections” and also includes the terms “cystitis” and “kidney infections”.
5 The category “balanitis or balanoposthitis” includes the terms “balanitis” and “balanoposthitis”, as well as the terms “candida balanitis” and “genital fungal infections”.
6 The category “vulvovaginal candidiasis” includes the terms “vulvovaginal candidiasis”, “vulvovaginal fungal infections”, “vulvovaginitis”, as well as the terms “vulvitis” and “genital fungal infections”.
Other adverse reactions that occurred in placebo-controlled studies of canagliflozin with a frequency of
Adverse reactions associated with a decrease in intravascular volume
The incidence of all adverse reactions associated with intravascular volume depletion (postural dizziness, orthostatic hypotension, hypotension, dehydration, and syncope) was higher. In a pooled analysis, patients receiving loop diuretics, patients with moderate renal impairment (GFR 30 to 2), and patients aged ≥75 years had a higher incidence of these adverse reactions. In a cardiovascular risk study, the incidence of serious intravascular volume depletion-related adverse reactions was not increased with canagliflozin, and treatment discontinuations due to this type of adverse reaction were infrequent.
Hypoglycemia when used as an adjunct to therapy with insulin or insulin secretagogues
Hypoglycemia was reported more frequently when canagliflozin was used as an adjunct to insulin or sulfonylurea therapy. This is consistent with the expected increase in the incidence of hypoglycemia in cases where a drug that is not associated with the development of this condition is added to insulin or drugs that enhance its secretion (for example, sulfonylureas).
Changes in laboratory parameters
Increased serum potassium concentration
Elevated serum potassium concentrations (>5.4 mEq/L and 15% above baseline concentration) were observed in 4.4% of patients receiving canagliflozin 100 mg, 7.0% of patients receiving canagliflozin 300 mg, and 4.8% of patients receiving placebo. Rarely, greater increases in serum potassium concentrations have been observed in patients with moderate renal impairment who have previously experienced increased potassium concentrations and/or who were receiving multiple drugs that reduce potassium excretion (potassium-sparing diuretics and angiotensin-converting enzyme (ACE) inhibitors). In general, the increase in potassium concentration was transient and did not require special treatment.
Increased serum creatinine and urea concentrations
During the first six weeks of treatment, there was a small mean increase in creatinine concentrations (the proportion of patients with a greater reduction in GFR (>30%) from baseline at any time during treatment was 2.0% with canagliflozin 100 mg, 4.1% with canagliflozin 300 mg, and 2.1% with placebo. These reductions in GFR were often transient, with However, by the end of the study, a similar decrease in GFR was observed in fewer patients. In a pooled analysis of patients with moderate renal failure, the proportion of patients with a greater decrease in GFR (>30%) compared with the baseline level observed at any stage of treatment was 9.3% when using canagliflozin at a dose of 100 mg, 12.2% when using a dose of 300 mg, and 4.9% when using placebo. After discontinuation of canagliflozin, these changes in laboratory parameters underwent positive dynamics or returned to the initial level.
Increased concentrations of low-density lipoproteins (LDL)
A dose-dependent increase in LDL cholesterol concentrations was observed with canagliflozin. The mean percentage changes in LDL cholesterol from baseline compared to placebo were 0.11 mmol/L (4.5%) and 0.21 mmol/L (8.0%) with canagliflozin 100 mg and 300 mg, respectively. Mean baseline LDL concentrations were 2.76 mmol/L, 2.70 mmol/L, and 2.83 mmol/L with canagliflozin 100 and 300 mg and placebo, respectively.
Increased hemoglobin concentration
With canagliflozin 100 mg and 300 mg, there was a small increase in the mean percentage change in hemoglobin concentration from baseline (3.5% and 3.8%, respectively) compared with a slight decrease in the placebo group (−1.1%). There was a comparable small increase in mean percentage change in red blood cell count and hematocrit from baseline. The majority of patients experienced an increase in hemoglobin concentration (>20 g/L), occurring in 6.0% of patients receiving canagliflozin 100 mg, 5.5% of patients receiving canagliflozin 300 mg, and 1.0% of patients receiving placebo. Most values remained within normal limits.
Decreased serum uric acid concentration
Canagliflozin 100 mg and 300 mg doses showed a modest decrease in mean uric acid concentration from baseline (−10.1% and −10.6%, respectively) compared with placebo, which showed a small increase in mean uric acid concentration from baseline (1.9%). The reduction in serum uric acid concentrations in the canagliflozin groups was maximum or near maximum at week 6 and was maintained throughout therapy. There was a transient increase in the concentration of uric acid in the urine. A pooled analysis of canagliflozin 100 mg and 300 mg showed that the incidence of nephrolithiasis was not increased.
Cardiovascular safety
There was no increase in cardiovascular risk with canagliflozin compared with the placebo group.
Interaction
Interaction
Drug interactions (in vitro data)
Canagliflozin did not induce the expression of CYP450 system isoenzymes (3A4, 2C9, 2C19, 2B6 and 1A2) in cultured human hepatocytes. It also did not inhibit cytochrome P450 isoenzymes (1A2, 2A6, 2C19, 2D6 or 2E1) and weakly inhibited CYP2B6, CYP2C8, CYP2C9, CYP3A4 in laboratory studies using human liver microsomes. In in vitro studies, canagliflozin has been shown to be a substrate of the drug metabolizing enzymes UGT1A9 and UGT2B4 and the drug transporters P-glycoprotein (P-gp) and MRP2. Canagliflozin is a weak P-gp inhibitor.
Canagliflozin undergoes minimal oxidative metabolism. Therefore, a clinically significant effect of other drugs on the pharmacokinetics of canagliflozin via the cytochrome P450 system is unlikely.
Effect of other drugs on canagliflozin
Clinical data indicate that the risk of significant interactions with concomitant medications is low.
Drugs that induce enzymes of the UDP-glucuronyl transferase (UGT) family and drug transporters
Concomitant use with rifampicin, a non-selective inducer of a number of enzymes of the UGT family and drug transporters, incl. UGT1A9, UGT2B4, P-gp, and MRP2 decreased canagliflozin exposure. Reduced exposure to canagliflozin may lead to a decrease in its effectiveness. If it is necessary to prescribe an inducer of UGT enzymes and drug transporters (for example, rifampicin, phenytoin, phenobarbital, ritonavir) simultaneously with canagliflozin, it is necessary to monitor the concentration of glycated hemoglobin HbA1c in patients receiving canagliflozin at a dose of 100 mg 1 time / day, and consider increasing the dose of canagliflozin to 300 mg 1 once a day, if additional glycemic control is necessary.
Drugs that inhibit UDP-glucuronyl transferase (UGT) family enzymes and drug transporters
Probenecid: Coadministration of canagliflozin with probenecid, a non-selective inhibitor of several UGT family enzymes and drug transporters, including UGT1A9 and MRP2, did not have a clinically significant effect on the pharmacokinetics of canagliflozin. Because canagliflozin is glucuronidated by two different UGT family enzymes and the glucuronidation is high potency/low affinity, other drugs are unlikely to have a clinically significant effect on the pharmacokinetics of canagliflozin via glucuronidation.
Cyclosporine: Clinically significant pharmacokinetic interactions with simultaneous use of canagliflozin with cyclosporine, an inhibitor of P-glycoprotein (P-gp), CYP3A and several drug transporters, including. MRP2 was not observed. The development of mild, transient “hot flashes” was noted with the simultaneous use of canagliflozin and cyclosporine. Dose adjustments of canagliflozin are not recommended. Significant drug interactions are not expected with other P-gp inhibitors.
Overdose
Overdose
Symptoms: There are no known cases of canagliflozin overdose. Single doses of canagliflozin, up to 1600 mg in healthy subjects and 300 mg twice daily for 12 weeks in patients with type 2 diabetes, were generally well tolerated.
Treatment: In case of overdose, usual supportive measures should be carried out, for example, removal of unabsorbed substance from the gastrointestinal tract, clinical monitoring and supportive treatment based on the clinical condition of the patient. Canagliflozin was virtually not eliminated during 4-hour dialysis. Canagliflozin is not expected to be eliminated by peritoneal dialysis.
Storage conditions
Storage conditions
In a dry place, protected from light, at a temperature not exceeding 30 ºС.
Shelf life
Shelf life
2 years
Manufacturer
Manufacturer
Johnson and Johnson, France
Additional information
| Shelf life | 2 years |
|---|---|
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 30 ºC. |
| Manufacturer | Johnson & Johnson, France |
| Medication form | pills |
| Brand | Johnson & Johnson |
Related products
Buy Invokana, 100 mg 30 pcs. with delivery to USA, UK, Europe and over 120 other countries.