No products in the cart.
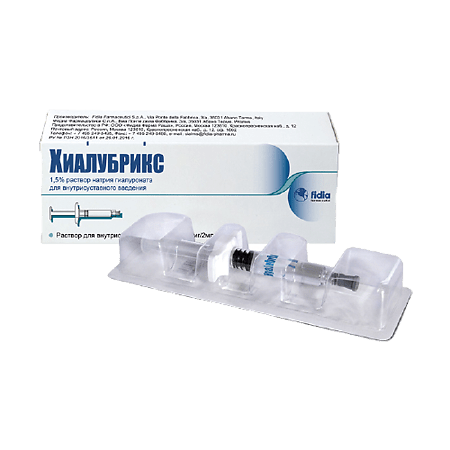
Hialubrix 30mg/2ml syringe
€1.00
Out of stock
(E-mail when Stock is available)
Description
Traumatic and degenerative joint pathology. Auxiliary in orthopedic surgery.
Active ingredient
Active ingredient
Composition
Composition
How to take, the dosage
How to take, the dosage
Interaction
Interaction
Special Instructions
Special Instructions
Synopsis
Synopsis
Contraindications
Contraindications
Side effects
Side effects
Overdose
Overdose
Similarities
Similarities
Additional information
| Shelf life | 30 months. |
|---|---|
| Conditions of storage | Store in the original container out of the reach of children at a temperature not exceeding 25 ° C. |
| Manufacturer | Fidia Farmaceutici S.p.A., Italy |
| Medication form | solution for injection |
| Brand | Fidia Farmaceutici S.p.A. |
Related products
Buy Hialubrix 30mg/2ml syringe with delivery to USA, UK, Europe and over 120 other countries.