No products in the cart.
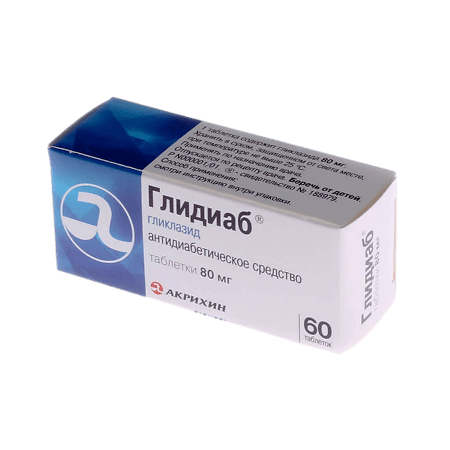
Glidib, tablets 80 mg 60 pcs
€4.35 €3.87
Description
Glediab is hypoglycemic.
Stimulates muscle glycogen synthetase activity, insulin secretion, potentiates insulin-secretory action of glucose, increases peripheral tissue sensitivity to insulin.
Pharmacodynamics
Shortens the time from the time of eating to the beginning of insulin secretion. Restores the early peak of insulin secretion, lowers the peak of hyperglycemia. In addition to action on carbohydrate metabolism, it affects the microcirculation. It reduces platelet adhesion and aggregation, normalizes vascular permeability and prevents the development of microthrombosis and atherosclerosis, restores the process of physiological parietal fibrinolysis; prevents an increased vascular response to adrenaline in microangiopathies. Slows down the development of diabetic retinopathy at the nonproliferative stage; with long-term use, reduces proteinuria in diabetic nephropathy; promotes weight loss in obese patients with an appropriate diet.
Pharmacokinetics
It is well absorbed from gastrointestinal tract, Cmax after oral dosage of 80 mg is reached in 4 hours. Binding to plasma proteins is 94%. T1/2 is about 12 hours. Metabolized in the liver to form several metabolites. Less than 1% is excreted unchanged in the urine, 12% in the feces as metabolites.
Indications
Indications
Diabetes mellitus type II in adults.
Pharmacological effect
Pharmacological effect
Glidiab is hypoglycemic.
Stimulates the activity of muscle glycogen synthetase, insulin secretion, potentiates the insulin secretory effect of glucose, increases the sensitivity of peripheral tissues to insulin.
Pharmacodynamics
Reduces the period of time from the moment of food intake to the onset of insulin secretion. Restores the early peak of insulin secretion, reduces the peak of hyperglycemia. In addition to its effect on carbohydrate metabolism, it has an effect on microcirculation. Reduces platelet adhesion and aggregation, normalizes vascular permeability and prevents the development of microthrombosis and atherosclerosis, restores the process of physiological parietal fibrinolysis; prevents increased vascular response to adrenaline in microangiopathies. Slows down the development of diabetic retinopathy at the non-proliferative stage; with long-term use, it reduces proteinuria in diabetic nephropathy; helps reduce body weight in obese patients when following an appropriate diet.
Pharmacokinetics
Well absorbed from the gastrointestinal tract, Cmax after taking the drug orally at a dose of 80 mg is achieved after 4 hours. Plasma protein binding is 94%. T1/2 – about 12 hours. Metabolized in the liver with the formation of several metabolites. Less than 1% is excreted in the urine unchanged, with feces – 12% in the form of metabolites.
Special instructions
Special instructions
Treatment is carried out in combination with a low-calorie, low-carbohydrate diet. During treatment, fasting and postprandial glucose levels should be regularly monitored.
In case of surgical interventions or in case of decompensation of diabetes mellitus, it is necessary to consider the possibility of using insulin preparations. Patients should be warned about the increased risk of hypoglycemia when taking ethanol, NSAIDs, and fasting.
If you take ethanol, it is also possible to develop disulfiram-like syndrome (abdominal pain, nausea, vomiting, headache). It is necessary to adjust the dose of the drug in case of physical or emotional stress or changes in diet. Elderly people are especially sensitive to the effects of hypoglycemic drugs; patients who do not receive a balanced diet; weakened patients; patients suffering from pituitary-adrenal insufficiency.
Impact on the ability to drive vehicles and operate machinery
At the beginning of treatment, during dose selection, patients prone to developing hypoglycemia are not recommended to engage in activities that require increased attention and speed of psychomotor reactions.
Active ingredient
Active ingredient
Gliclazide
Composition
Composition
Active ingredient:
Gliclazide 80 mg
Pregnancy
Pregnancy
The drug is contraindicated for use during pregnancy.
If use is necessary during lactation, breastfeeding should be discontinued.
Contraindications
Contraindications
low blood sugar
discomfort in the stomach, nausea
headache, weakness
allergic reactions.
Side Effects
Side Effects
From the endocrine system: hypoglycemia (in case of violation of the dosage regimen and inadequate diet) – headache, feeling of fatigue, feeling of hunger, sweating, severe weakness, aggressiveness, anxiety, irritability, inattention, inability to concentrate and slow reaction, depression, visual impairment, aphasia, tremor, feeling of helplessness, sensory disorders, dizziness, loss of self-control, delirium, convulsions, hypersomnia, loss consciousness, shallow breathing, bradycardia.
From the digestive system: dyspepsia (nausea, diarrhea, feeling of heaviness in the epigastrium), anorexia (the severity decreases when taking the drug with meals), liver dysfunction (cholestatic jaundice, increased activity of liver transaminases).
From the hematopoietic system: anemia, thrombocytopenia, leukopenia. Allergic reactions: itching, urticaria, maculopapular rash.
Interaction
Interaction
An increase in the hypoglycemic effect of Glidiab® is observed when used simultaneously with ACE inhibitors (captopril, enalapril), histamine H2 receptor blockers (cimetidine), antifungal drugs (miconazole, fluconazole), NSAIDs (phenylbutazone, indomethacin, diclofenac), fibrates (clofibrate, bezafibrate), anti-tuberculosis drugs (ethionamide), salicylates, indirect coumarin anticoagulants, anabolic steroids, beta-blockers, cyclophosphamide, chloramphenicol, MAO inhibitors, long-acting sulfonamides, fenfluramine, fluoxetine, pentoxifylline, guanethidine, theophylline, drugs, blocking tubular secretion, reserpine, bromocriptine, disopyramide, pyridoxine, allopurinol, ethanol and ethanol-containing drugs, as well as other hypoglycemic drugs (acarbose, biguanides, insulin).
A decrease in the hypoglycemic effect of the drug Glidiab® is observed when used simultaneously with barbiturates, corticosteroids, sympathomimetics (epinephrine, clonidine, ritodrine, salbutamol, terbutaline), with phenytoin, slow calcium channel blockers, carbonic anhydrase inhibitors (acetazolamide), with thiazide diuretics, chlorthalidone, furosemide, triamterene, with asparaginase, baclofen, danazol, diazoxide, isoniazid, morphine, glucagon, rifampicin, with thyroid hormones, lithium salts, in high doses – with nicotinic acid, chlorpromazine, estrogens and oral contraceptives containing them.
When interacting with ethanol, a disulfiram-like reaction may develop. Glidiab® increases the risk of ventricular extrasystole when taking cardiac glycosides. Beta-blockers, clonidine, reserpine, guanethidine can mask the clinical manifestations of hypoglycemia.
Overdose
Overdose
Symptoms: hypoglycemia may develop up to the development of hypoglycemic coma.
Treatment: If the patient is conscious, dextrose (glucose) or sugar solution is prescribed orally. In case of loss of consciousness, a 40% dextrose solution is administered intravenously, and 1-2 mg of glucagon intramuscularly. After regaining consciousness, the patient must be given food rich in easily digestible carbohydrates to avoid re-development of hypoglycemia.
Storage conditions
Storage conditions
In a dry place, protected from light, at a temperature not exceeding 25 °C
Shelf life
Shelf life
4 years
Manufacturer
Manufacturer
Akrikhin JSC, Russia
Additional information
| Shelf life | 4 years |
|---|---|
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25 °C |
| Manufacturer | Akrihin HFC JSC, Russia |
| Medication form | pills |
| Brand | Akrihin HFC JSC |
Related products
Buy Glidib, tablets 80 mg 60 pcs with delivery to USA, UK, Europe and over 120 other countries.