No products in the cart.
Femostone conti, 28 pcs.
€38.51 €33.37
Description
Pharmacodynamics
Estradiol, as part of the drug and identical to endogenous estradiol, replenishes estrogen deficiency in the female body after menopause.
Estradiol provides effective treatment of psycho-emotional and vegetative menopausal symptoms: “hot flashes”, increased sweating, sleep disorders, increased nervous excitability, dizziness, headache, involution of skin and mucous membranes, especially of urogenital system (dryness and irritation of vaginal mucosa, painfulness during sexual intercourse). Hormone replacement therapy (HRT) with Femostone 1/5 prevents loss of bone mass in the postmenopausal period. Risk factors for osteoporosis in postmenopause include early menopause, long-term use of GCS in the recent past, and smoking.
Pemostone 1/5 changes the lipid profile: It lowers total cholesterol, LDL, and increases HDL.
Didrogesterone is a progestagen that is effective when taken orally and brings about the secreting phase in the endometrium. Didrogesterone reduces the risk of endometrial hyperplasia and/or carcinogenesis increased by estrogen. Didrogesterone has no estrogenic, androgenic, anabolic or glucocorticoid activity.
In order to achieve the maximum prophylactic effect ZGT should be started immediately after the onset of menopause. The effect is evident for the duration of treatment (information on the use of estrogen for more than 10 years is limited).
Pharmacokinetics
Micronized estradiol is easily absorbed after oral administration. It is metabolized in the liver to estrone and estrone sulfate, which also undergoes hepatic biotransformation. Glucuronides of estrone and estradiol are excreted mainly with the urine.
Dydrogesterone is rapidly absorbed from the gastrointestinal tract after oral administration. It is completely metabolized, the main metabolite being 20-dihydrodihydrogesterone (DDH), present mainly as a glucuronic acid conjugate in the urine. T1/2 is 5-7 hours, DHD is 14-17 hours. Complete excretion occurs after 72 hours.
Indications
Indications
hormone replacement therapy for disorders caused by estrogen deficiency in postmenopausal women (no earlier than 12 months after the last menstruation);
prevention of osteoporosis in postmenopausal women at high risk of fractures with intolerance or contraindications to the use of other drugs
Pharmacological effect
Pharmacological effect
Pharmacodynamics
Estradiol, which is part of the drug and is identical to endogenous estradiol, compensates for the deficiency of estrogen in the female body after menopause.
Estradiol provides effective treatment of psycho-emotional and vegetative menopausal symptoms: hot flashes, increased sweating, sleep disturbances, increased nervous excitability, dizziness, headache, involution of the skin and mucous membranes, especially the genitourinary system (dryness and irritation of the vaginal mucosa, pain during sexual intercourse). Hormone replacement therapy (HRT) with Femoston 1/5 prevents bone loss in the postmenopausal period. Risk factors for osteoporosis in postmenopause are early onset of menopause, long-term use of corticosteroids in the recent past, smoking.
Taking Femoston 1/5 changes the lipid profile: it reduces the level of total cholesterol, LDL and increases the level of HDL.
Dydrogesterone is a progestogen, effective when taken orally, which ensures the onset of the secretion phase in the endometrium. Dydrogesterone reduces the risk of endometrial hyperplasia and/or carcinogenesis, increased under the influence of estrogen. Dydrogesterone does not have estrogenic, androgenic, anabolic or glucocorticoid activity.
To achieve the maximum preventive effect, HRT should be started immediately after menopause. The effect is manifested throughout the entire period of treatment (information on the use of estrogens for more than 10 years is limited).
Pharmacokinetics
After oral administration, micronized estradiol is easily absorbed. Metabolized in the liver to estrone and estrone sulfate, which also undergoes hepatic biotransformation. Glucuronides of estrone and estradiol are excreted primarily in the urine.
Dydrogesterone after oral administration is quickly absorbed from the gastrointestinal tract. Metabolized completely, the main metabolite is 20-dihydrodidrogesterone (DHD), present in the urine, mainly in the form of a glucuronic acid conjugate. T1/2 – 5-7 hours, DGD – 14-17 hours. Complete elimination occurs after 72 hours.
Special instructions
Special instructions
Before prescribing or restarting HRT, it is necessary to collect a complete medical and family history, as well as conduct a general and gynecological examination in order to identify possible contraindications and conditions requiring the necessary precautions. During treatment with Femoston®, 1/5 of women are recommended to be examined periodically (the frequency and nature of the examinations are determined individually).
Breast examination and/or mammography are carried out in accordance with accepted standards, taking into account clinical indications.
Femoston 1/5 is prescribed to women who have been postmenopausal for at least 1 year.
When switching from another estrogen-progestogen drug for HRT, Femoston 1/5 should be taken at the end of the estrogen-progestogen phase without a break in taking the tablets.
The use of estrogens may affect the results of the following laboratory tests: glucose tolerance testing, thyroid and liver function tests.
Patients receiving HRT and having the following conditions (currently or in the past) should be under close medical supervision: uterine leiomyoma, endometriosis, history of thrombosis or their risk factors, arterial hypertension, renal dysfunction, diabetes mellitus with vascular complications, bronchial asthma, porphyria, hemoglobinopathy, cholelithiasis, epilepsy, otosclerosis, multiple sclerosis, migraine or intense headache.
Generally recognized risk factors for thrombosis and thromboembolism while taking HRT are a history of thromboembolic complications, severe forms of obesity (body mass index more than 30 kg/m2) and systemic lupus erythematosus. There is no generally accepted opinion regarding the role of varicose veins in the development of thromboembolism.
The risk of developing deep vein thrombosis of the lower extremities may temporarily increase with prolonged immobilization, major trauma, or surgery. In cases where prolonged immobilization is necessary after surgery, temporary cessation of HRT should be considered 4–6 weeks before surgery.
When deciding on HRT in patients with recurrent deep vein thrombosis or thromboembolism receiving anticoagulant treatment, its benefits and risks must be carefully assessed.
If thrombosis develops after starting HRT, Femoston 1/5 should be discontinued. The patient should be informed of the need to consult a doctor if the following symptoms occur: painful swelling of the lower extremities, sudden loss of consciousness, dyspnea, blurred vision.
There is data demonstrating a slight increase in the incidence of breast cancer in women receiving long-term (more than 10 years) hormone replacement therapy. Detection of breast cancer may be due to early diagnosis, the biological effects of HRT, or a combination of both. The likelihood of being diagnosed with breast cancer increases with the duration of treatment and returns to normal 5 years after stopping HRT.
Patients who have previously received HRT using only estrogen drugs should be especially carefully examined before starting treatment with Femoston® 1/5 in order to identify possible endometrial hyperstimulation.
Breakthrough uterine bleeding and mild menstrual-like bleeding may occur in the first months of treatment with the drug. If, despite dose adjustment, such bleeding does not stop, the drug should be discontinued until the cause of the bleeding is determined. If bleeding recurs after a period of amenorrhea or continues after discontinuation of treatment, its etiology should be determined. This may require an endometrial biopsy.
Femoston 1/5 is not a contraceptive. Perimenopausal patients are advised to use non-hormonal contraceptives.
The effect on the ability to drive a car and use other machinery is unknown.
Active ingredient
Active ingredient
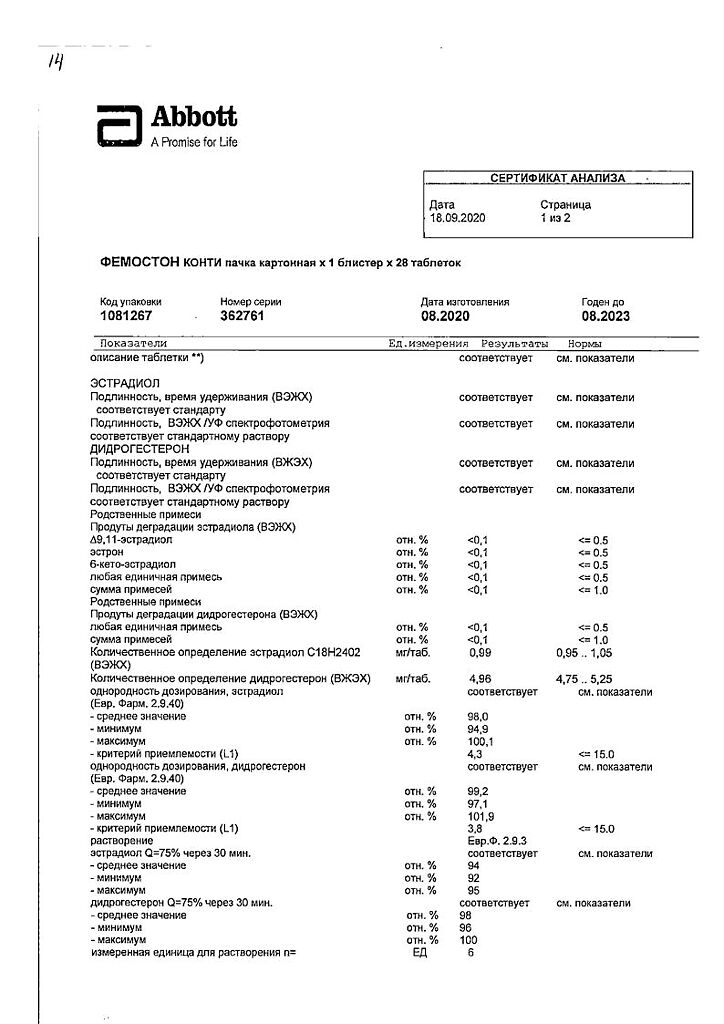
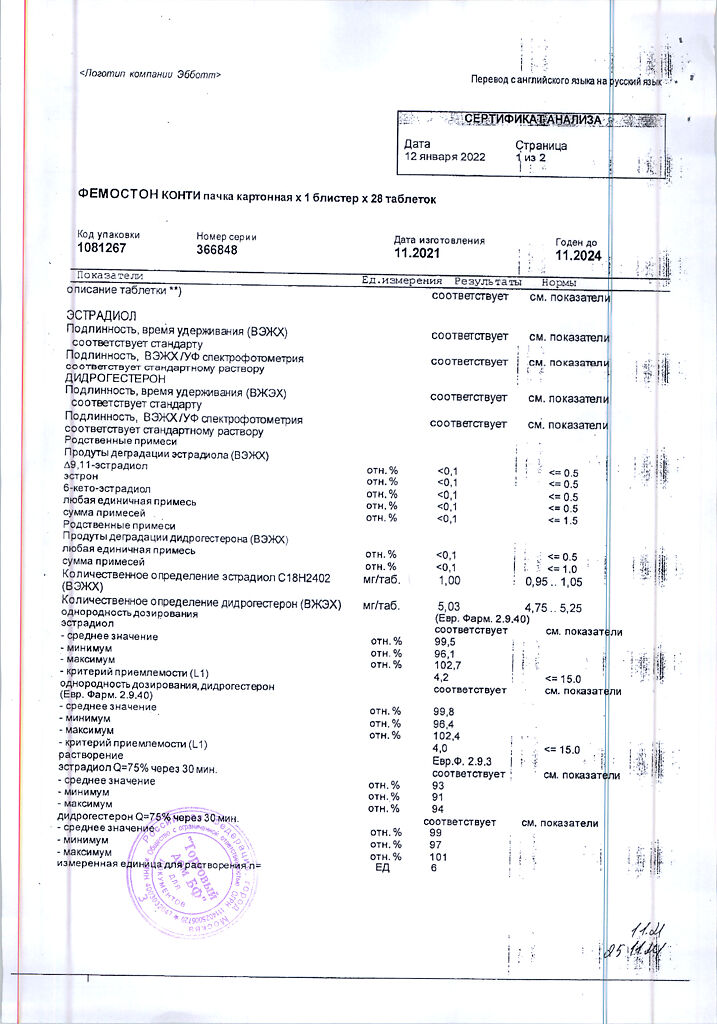
Dydrogesterone, Estradiol
Composition
Composition
1 tablet contains:
active ingredients:
estradiol 1 mg,
dydrogesterone 5 mg;
excipients:
lactose monohydrate,
methylhydroxypropylcellulose,
corn starch,
anhydrous colloidal silicon dioxide,
magnesium stearate,
macrogol 400,
titanium dioxide (E171),
iron oxide yellow and red (E172),
Opadry orange (Y-8734).
Pregnancy
Pregnancy
The drug is contraindicated during pregnancy and breastfeeding. If pregnancy occurs during treatment with Femoston® conti, therapy should be discontinued immediately.
Contraindications
Contraindications
pregnancy and breastfeeding;
diagnosed or suspected breast cancer;
diagnosed or suspected estrogen-dependent malignancies (for example, endometrial cancer);
diagnosed or suspected progestogen-dependent neoplasms (for example, meningioma);
bleeding from the vagina of unknown etiology;
untreated endometrial hyperplasia;
thrombosis (arterial and venous) and thromboembolism currently or in history (including thrombosis, deep vein thrombosis; pulmonary embolism, myocardial infarction, ischemic or hemorrhagic cerebrovascular disorders);
multiple or pronounced factors of arterial or venous thrombosis associated with congenital or acquired predisposition, for example, protein C deficiency, protein S deficiency, antithrombin III deficiency, the presence of antibodies to phospholipids (anticardiolipin antibodies, lupus anticoagulant), angina pectoris, prolonged immobilization, severe forms of obesity (body mass index more than 30 kg/m2), cerebrovascular diseases brain or coronary arteries, transient ischemic attacks, complicated lesions of the heart valve apparatus, atrial fibrillation;
acute or chronic liver diseases currently or in history (before normalization of liver function tests), including malignant liver tumors;
porphyria;
hypersensitivity to the components of the drug;
galactose intolerance, lactase deficiency, glucose-galactose malabsorption syndrome.
Taking Femoston® conti should be discontinued if contraindications are identified and/or if the following conditions occur: jaundice and/or liver dysfunction; uncontrolled arterial hypertension; migraine-like headache that first appeared during the use of drugs for HRT.
Side Effects
Side Effects
Headache, migraine, dizziness;
depression, nervousness;
abdominal pain, nausea, vomiting, flatulence;
tension/soreness of the mammary glands, “spotting” spotting in postmenopause, metrorrhagia, heavy menstrual-like bleeding, scanty or absent menstrual-like bleeding, acyclic bleeding, painful menstrual-like bleeding, pain in the lower abdomen, changes in vaginal secretion, vaginal candidiasis;
back pain (lower back);
allergic reactions such as hives, skin rash and itching;
asthenic conditions (weakness, malaise, fatigue), peripheral edema;
increase in body weight.
Interaction
Interaction
The estrogenic effect of Femoston is reduced when taken simultaneously with drugs that are inducers of microsomal liver enzymes: anticonvulsants (barbiturates, phenytoin, carbamazepine, oxcarbazepine, topiramate, felbamate), antimicrobial drugs (rifampicin, rifabutin, nevirapine, efavirenz); with herbal preparations containing St. John’s wort (Hypericum perforatum).
The estrogenic effect of Femoston may be enhanced when taken simultaneously with drugs that inhibit microsomal liver enzymes (ritonavir, nelfinavir). There are no known interactions between dydrogesterone and other drugs.
The patient should inform the doctor about the medications she is taking during HRT or was taking before prescribing Femoston.
Overdose
Overdose
There have been no reports of overdose symptoms to date.
Symptoms: possible increased side effects of the drug.
Treatment: symptomatic, there is no specific antidote.
Storage conditions
Storage conditions
At a temperature not exceeding 30 °C (do not freeze)
Shelf life
Shelf life
1 year
Manufacturer
Manufacturer
Abbott Biologicals B.V., The Netherlands
Additional information
| Shelf life | 1 year |
|---|---|
| Conditions of storage | At a temperature not exceeding 30 °C (do not freeze) |
| Manufacturer | Abbott Biologicals B.V., The Netherlands |
| Medication form | pills |
| Brand | Abbott Biologicals B.V. |
Related products
Buy Femostone conti, 28 pcs. with delivery to USA, UK, Europe and over 120 other countries.