No products in the cart.
Coraxan, 7.5 mg 56 pcs
€35.68 €29.73
Description
Coraxan is a heart rhythm slowing drug whose mechanism of action consists in selective and specific inhibition of If channels of the sinus node, which control spontaneous diastolic depolarization in the sinus node and regulate the heart rate (HR). Ivabradine has a selective effect on the sinus node without affecting the timing of impulse conduction along the intraatrial, atrial-ventricular and intraventricular conduction pathways, as well as on myocardial contractility and ventricular repolarization.
Ivabradine may also interact with Ih channels of the retina, similar to If channels of the heart, which are involved in causing temporary changes in the visual perception system by altering the retinal response to bright light stimuli.
In provocative circumstances (e.g., a rapid change in brightness in the visual field), partial inhibition of Ih channels by ivabradine causes the phenomenon of changes in light perception (photopsia). Photopsia is characterized by a transient change in brightness in a limited area of the visual field (see section “Side effects”).
The main pharmacological characteristic of ivabradine is the ability to dose-dependently decrease heart rate (HR).
Indications
Indications
Stable angina
Treatment of stable angina in patients with normal sinus rhythm:
– if you are intolerant or have contraindications to the use of beta-blockers
– in combination with beta-blockers with inadequate control of stable angina against the background of the optimal dose of beta-blocker
Chronic heart failure
To reduce the incidence of cardiovascular complications (mortality from cardiovascular diseases and hospitalization due to increased symptoms of heart failure) in patients with chronic heart failure, with sinus rhythm and a heart rate of at least 70 beats/min.
Pharmacological effect
Pharmacological effect
Coraxan is a drug that slows the heart rate, the mechanism of action of which is the selective and specific inhibition of If channels of the sinus node, which control spontaneous diastolic depolarization in the sinus node and regulate heart rate (HR). Ivabradine has a selective effect on the sinus node, without affecting the timing of impulses along the intra-atrial, atrioventricular and intraventricular pathways, as well as on myocardial contractility and ventricular repolarization.
Ivabradine may also interact with the Ih channels of the retina, similar to the If channels of the heart, which are involved in causing temporary changes in the visual perception system by changing the retinal response to bright light stimuli.
Under provoking circumstances (for example, a rapid change in brightness in the visual field), partial inhibition of Ih channels by ivabradine causes the phenomenon of changes in light perception (photopsia). Photopsia is characterized by a transient change in brightness in a limited area of the visual field (see section “Side effects”).
The main pharmacological feature of ivabradine is the ability to dose-dependently reduce heart rate (HR).
Special instructions
Special instructions
Heart rhythm disturbances
Coraxan is not effective for the treatment or prevention of arrhythmias. Its effectiveness decreases against the background of the development of tachyarrhythmia (for example, ventricular or supraventricular tachycardia). The drug is not recommended for patients with atrial fibrillation (atrial fibrillation) or other types of arrhythmias associated with sinus node function.
During therapy, patients should be clinically monitored for atrial fibrillation (paroxysmal or permanent). For clinical indications (for example, worsening of angina, the appearance of palpitations, irregular heart rhythm), an ECG should be included in routine monitoring.
Use in patients with bradycardia
Coraxan is contraindicated if, before starting therapy, the resting heart rate is less than 60 beats/min. If during therapy the resting heart rate decreases to values less than 50 beats/min, or the patient experiences symptoms associated with bradycardia (such as dizziness, fatigue or hypotension), it is necessary to reduce the dose of the drug. If, when the dose of the drug is reduced, the heart rate remains less than 50 beats/min, or symptoms associated with bradycardia persist, then taking the drug Coraxan should be stopped.
Combined use as part of antianginal therapy
The use of Coraxan in combination with BMCCs that reduce heart rate, such as verapamil or diltiazem, is not recommended. With the combined use of ivabradine with nitrates and BMCA derivatives of the dihydropyridine series, such as amlodipine, no changes in the safety profile of the therapy were noted. It has not been established that combined use with BMCC increases the effectiveness of ivabradine.
Stroke
It is not recommended to prescribe the drug immediately after a stroke, because There are no data on the use of the drug during this period.
Functions of visual perception
Coraxan affects the function of the retina. Currently, no toxic effects of ivabradine on the retina have been identified, but the effect of the drug on the retina with long-term use (over 1 year) is currently unknown. If visual impairments that are not described in these instructions occur, you should consider stopping taking the drug Coraxan?. Patients with retinitis pigmentosa should take Coraxan with caution.
Excipients
The drug contains lactose, so Coraxan is not recommended for patients with lactase deficiency, lactose intolerance, or glucose-galactose malabsorption syndrome.
Arterial hypotension
Due to insufficient clinical data, the drug should be prescribed with caution to patients with arterial hypotension.
Coraxan is contraindicated in severe arterial hypotension (systolic blood pressure less than 90 mm Hg and diastolic blood pressure less than 50 mm Hg).
Atrial fibrillation (atrial fibrillation) – cardiac arrhythmias
There is no proven increase in the risk of developing severe bradycardia while taking the drug Coraxan when restoring sinus rhythm during pharmacological cardioversion. However, due to the lack of sufficient data, if it is possible to delay electrical cardioversion, Coraxan should be discontinued 24 hours before it is performed.
Use in patients with congenital long QT syndrome or patients taking drugs that prolong the QT interval
Coraxan should not be prescribed for congenital long QT syndrome, or in combination with drugs that prolong the QT interval. If such therapy is necessary, strict ECG monitoring is necessary.
Moderate liver failure
In case of moderately severe liver failure (less than 9 points on the Child-Pugh scale), therapy with Coraxan should be carried out with caution.
Severe renal failure
In case of severe renal failure (creatinine clearance less than 15 ml/min), therapy with Coraxan should be carried out with caution.
Active ingredient
Active ingredient
Ivabradin
Composition
Composition
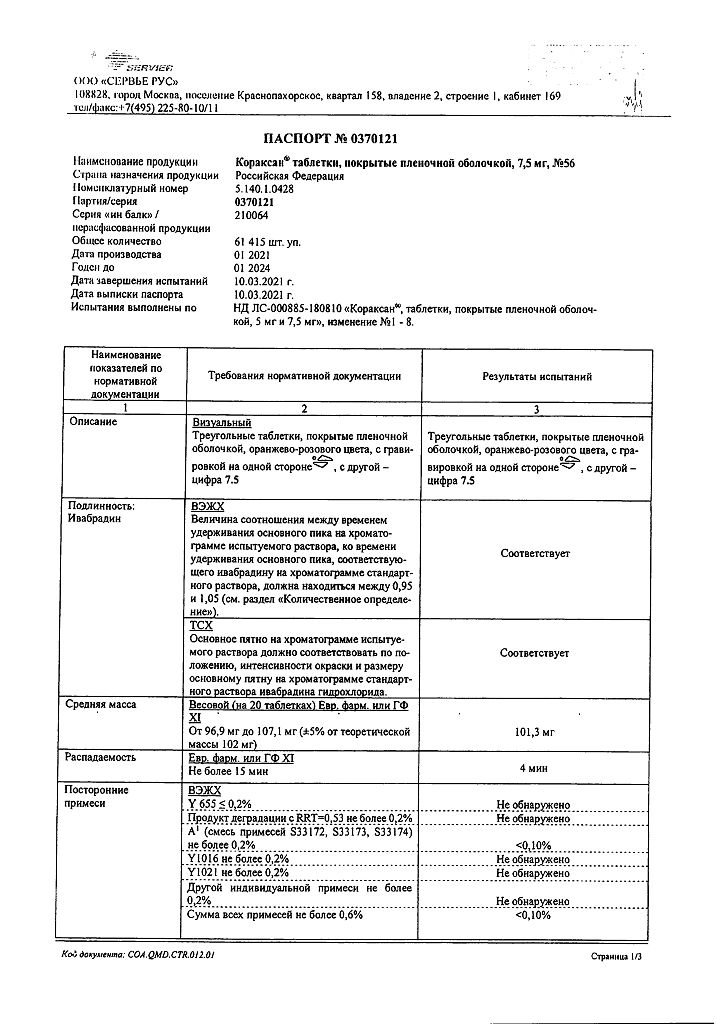
1 tablet of 7.5 mg contains:
active substance:
ivabradine hydrochloride 8.085 mg, corresponding to 7.5 mg base.
excipients:
lactose monohydrate 61.215 mg,
magnesium stearate 0.5 mg,
corn starch 20 mg,
maltodextrin 10 mg,
colloidal silicon dioxide anhydrous 0.20 mg.
Shell:
glycerol 0.08740 mg, hypromellose 1.45276 mg, iron oxide yellow (E172) 0.01457 mg, iron oxide red (E172) 0.00485 mg, macrogol 6000 0.09276 mg, magnesium stearate 0.08740 mg, titanium dioxide (E171) 0.26026 mg.
Pregnancy
Pregnancy
The drug Coraxan is contraindicated for use during pregnancy. Currently, there is insufficient data on the use of the drug during pregnancy. Preclinical studies of ivabradine revealed embryotoxic and teratogenic effects.
The use of Coraxan during breastfeeding is contraindicated. There is no information on the penetration of ivabradine into breast milk.
Contraindications
Contraindications
Hypersensitivity to ivabradine or any of the excipients of Coraxan;
Bradycardia (heart rate at rest less than 60 beats/min (before treatment));
cardiogenic shock;
acute myocardial infarction;
severe arterial hypotension (systolic blood pressure less than 90 mm Hg and diastolic blood pressure less than 50 mm Hg);
severe liver failure (more than 9 points on the Child-Pugh scale);
sick sinus syndrome;
sinoatrial block;
presence of an artificial pacemaker;
unstable angina;
atrioventricular (AV) block of the third degree;
simultaneous use with powerful inhibitors of isoenzymes of the cytochrome P450 3A4 system, such as antifungals of the azole group (ketoconazole, itraconazole), antibiotics of the macrolide group (clarithromycin, erythromycin for oral administration, josamycin, telithromycin), HIV protease inhibitors (nelfinavir, ritonavir) and nefazodone;
pregnancy and lactation;
age under 18 years (the effectiveness and safety of the drug in this age group has not been studied);
lactase deficiency, lactose intolerance, glucose-galactose malabsorption syndrome.
With caution:
Moderate liver failure (less than 9 points on the Child-Pugh scale); severe renal failure (creatinine clearance less than 15 ml/min); congenital prolongation of the QT interval (see section “Interaction with other drugs); simultaneous use of drugs that prolong the QT interval; simultaneous use of moderate inhibitors of cytochrome CYP3A4 isoenzymes and grapefruit juice; asymptomatic left ventricular dysfunction; atrioventricular block of the second degree; recent stroke; pigmentary degeneration of the retina (retinitis pigmentosa); arterial hypotension; simultaneous use with slow calcium channel blockers (SCBCs) that reduce heart rate, such as verapamil or diltiazem.
Side Effects
Side Effects
From the side of the organ of vision:
Very common: changes in light perception (photopsia): observed in 14.5% of patients and described as a transient change in brightness in a limited area of the visual field. As a rule, such phenomena were provoked by a sharp change in the intensity of illumination in the visual field. Basically, photopsia appeared in the first two months of treatment with subsequent recurrence. The severity of photopsia was usually mild or moderate. The appearance of photopsia stopped while therapy was continued (in 77.5% of cases) or after its completion. In less than 1% of patients, the appearance of photopsia was the reason for refusal of treatment.
Common: blurred vision.
From the cardiovascular system:
Often: bradycardia: in 3.3% of patients, especially in the first 2-3 months of therapy, in 0.5% of patients severe bradycardia developed with a heart rate of no more than 40 beats/min; AV block of the first degree; ventricular extrasystole.
Uncommon: palpitations, supraventricular extrasystole.
Unspecified frequency: hypotension, possibly associated with bradycardia
The following adverse events identified in clinical studies occurred with equal frequency in both the group of patients receiving ivabradine and in the control group, which suggests that they are related to the disease itself and not to the use of ivabradine: sinus arrhythmia; angina pectoris, including unstable; atrial fibrillation; myocardial ischemia; myocardial infarction and ventricular tachycardia.
From the digestive system: uncommon: nausea, constipation, diarrhea.
From the central nervous system:
Often: headache, especially in the first month of therapy; dizziness, possibly associated with bradycardia.
Uncommon: shortness of breath, vertigo, muscle spasms.
Unspecified frequency: syncope, possibly associated with bradycardia.
Laboratory indicators: uncommon: hyperuricemia, eosinophilia, increased plasma creatinine concentration.
From the skin and subcutaneous fat: skin rash, itching, erythema, angioedema, urticaria.
General disorders and symptoms: asthenia, fatigue, malaise, possibly associated with bradycardia.
Interaction
Interaction
Pharmacodynamic interaction
Not recommended combinations
Drugs that prolong the QT interval (for example, quinidine, disopyramide, bepridil, sotalol, ibutilide, amiodarone).
Drugs that prolong the QT interval, not related to cardiovascular drugs (for example, pimozide, ziprasidone, sertindole, mefloquine, halofantrine, pentamidine, cisapride, erythromycin IV).
Concomitant use with drugs that prolong the QT interval may increase the decrease in heart rate, so careful cardiac monitoring should be performed if coadministration is necessary.
Pharmacokinetic interaction
Cytochrome P4503A4 (CYP3A4). Ivabradine is metabolized in the liver by enzymes of the cytochrome P450 (CYP3A4) system and is a very weak inhibitor of this cytochrome. Ivabradine does not have a significant effect on the metabolism and plasma concentrations of other cytochrome CYP3A4 substrates. At the same time, CYP3A4 inhibitors and inducers interact with ivabradine and affect its metabolism and pharmacokinetic properties. It was found that inhibitors of cytochrome CYP3A4 increase, and inducers of cytochrome CYP3A4 decrease, plasma concentrations of ivabradine. An increase in the concentration of ivabradine in the blood plasma increases the risk of developing severe bradycardia.
Contraindicated combinations
Concomitant use with strong cytochrome P450 inhibitors, such as azole antifungals (ketoconazole, itraconazole), macrolide antibiotics (clarithromycin, oral erythromycin, josamycin, telithromycin), HIV protease inhibitors (nelfinavir, ritonavir) and nefazodone, ketoconazole (at a dose of 200 mg 1 time per day) or josamycin (at a dose of 1 g 2 times a day), increase the average concentration of ivabradine in the blood plasma by 7–8 times.
Not recommended combinations
Moderate CYP3A4 inhibitors. The combined use of ivabradine and drugs that reduce heart rate – diltiazem or verapamil, was well tolerated by patients and was accompanied by an increase in ivabradine concentration by 2-3 times, with an additional decrease in heart rate of approximately 5 beats/min.
This combination is not recommended.
Combinations requiring caution when using
Moderate CYP3A4 inhibitors. Concomitant use of ivabradine with other CYP3A4 inhibitors (eg fluconazole) should begin with an initial dose of 2.5 mg 2 times a day. When heart rate is less than 60 beats/min, careful monitoring of heart rate is necessary.
Grapefruit juice. When taking grapefruit juice, there was a 2-fold increase in the concentration of ivabradine in the blood. During ivabradine therapy, grapefruit juice intake should be kept to a minimum.
Inducers of CYP3A4, such as rifampicin, barbiturates, phenytoin and herbal preparations containing St. John’s wort (Hypericum perforatum), when used together, may lead to a decrease in the blood concentration and activity of ivabradine and require the selection of a higher dose of ivabradine. During therapy with ivabradine, the use of drugs and products containing St. John’s wort should be kept to a minimum.
Combined use with other drugs
The absence of a clinically significant effect on the pharmacodynamics and pharmacokinetics of ivabradine for the following drugs was demonstrated: proton pump inhibitors (omeprazole, lansoprazole), phosphodiesterase-5 inhibitors (sildenafil), HMG-CoA reductase inhibitors (simvastatin), CCB derivatives of the dihydropyridine series (amlodipine, lacidipine), digoxin and warfarin.
It has been shown that ivabradine does not have a clinically significant effect on the pharmacokinetics of simvastatin, amlodipine, lacidipine, the pharmacokinetics and pharmacodynamics of digoxin, warfarin and the pharmacodynamics of acetylsalicylic acid.
In a phase III pilot study, the use of the following drugs did not have special restrictions, and therefore they can be prescribed in combination with ivabradine without special precautions: ACE inhibitors, angiotensin II receptor antagonists, diuretics, short- and long-acting nitrates, HMG-CoA reductase inhibitors, fibrates, proton pump inhibitors, oral hypoglycemic agents, acetylsalicylic acid acid and other antithrombotic agents.
Overdose
Overdose
Symptoms: severe and prolonged bradycardia.
Treatment of severe bradycardia should be symptomatic and carried out in specialized departments. In case of development of bradycardia in combination with hemodynamic disturbances, symptomatic treatment with intravenous administration of beta-adrenergic agonists, such as isoprenaline, is indicated.
If necessary, an artificial pacemaker can be installed.
Storage conditions
Storage conditions
No special storage conditions required
Shelf life
Shelf life
3 years
Manufacturer
Manufacturer
Servier Rus LLC, Russia
Additional information
| Shelf life | 3 years |
|---|---|
| Conditions of storage | No special storage conditions required |
| Manufacturer | Servier Rus LLC, Russia |
| Medication form | pills |
| Brand | Servier Rus LLC |
Other forms…
Related products
Buy Coraxan, 7.5 mg 56 pcs with delivery to USA, UK, Europe and over 120 other countries.