No products in the cart.
Description
Cancer
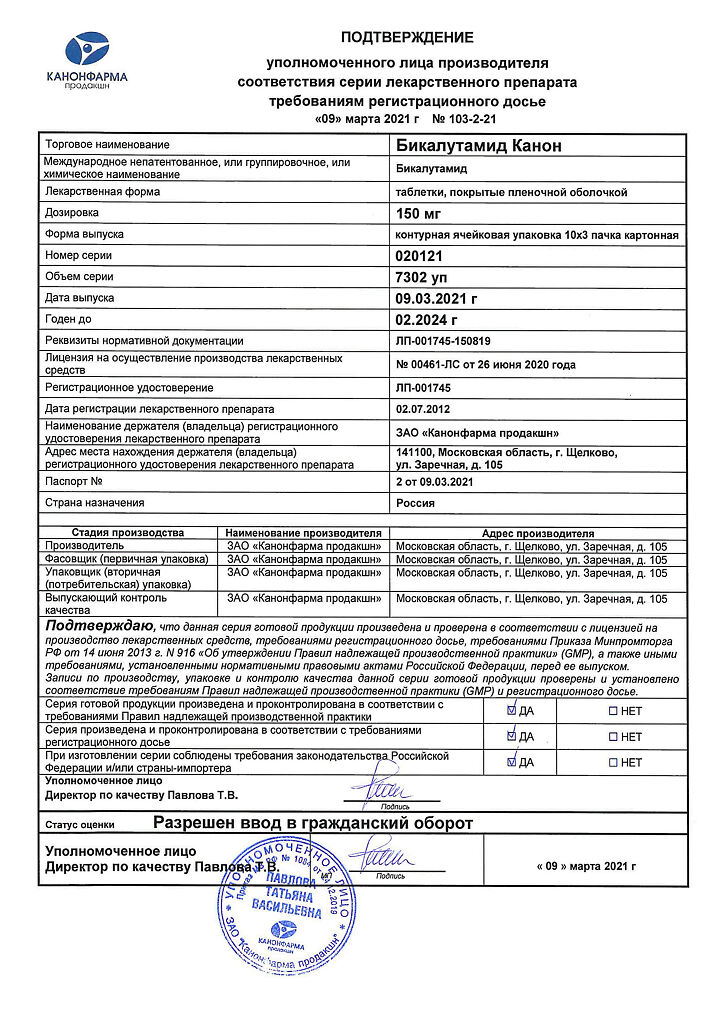
Prostate cancer – as part of combination therapy with a GnRH analogue or with surgical castration.
Indications
Indications
Prostate cancer – as part of combination therapy with a GnRH analogue or with surgical castration.
Pharmacological effect
Pharmacological effect
Antitumor hormonal agents and hormone antagonists
Special instructions
Special instructions
Use with caution in patients with moderate to severe liver dysfunction, because accumulation of bicalutamide in the body is possible.
During treatment, regular monitoring of liver function is required. If severe changes develop, you should stop taking bicalutamide.
Active ingredient
Active ingredient
Bicalutamide
Composition
Composition
Active ingredients:
bicalutamide 150 mg.
Excipients:
corn starch 107 mg,
croscarmellose sodium 19.5 mg,
lactose monohydrate 132.9 mg,
povidone 7.6 mg,
sodium stearyl fumarate 3 mg.
Film shell composition:
Opadry II white 12 mg.
Pregnancy
Pregnancy
Not prescribed for female patients.
Contraindications
Contraindications
Simultaneous use with terfenadine, astemizole, cisapride;
childhood;
hypersensitivity to bicalutamide;
not prescribed to female patients.
Side Effects
Side Effects
From the endocrine system: hot flashes, breast tenderness and gynecomastia, decreased libido.
From the digestive system: abdominal pain, diarrhea, nausea, vomiting, increased transaminase levels, jaundice, cholestasis.
From the side of the central nervous system: asthenia, depression are possible.
Dermatological reactions: alopecia, hair regrowth, itching, dry skin.
Other: hematuria.
Interaction
Interaction
When bicalutamide was used for 28 days while taking midazolam, the AUC of midazolam increased by 80%.
With simultaneous use of bicalutamide with cyclosporine or calcium channel blockers, potentiation or development of adverse reactions is possible.
It is theoretically possible to increase the concentration of bicalutamide in the blood plasma when used simultaneously with inhibitors of microsomal liver enzymes, which may be accompanied by an increase in the incidence of adverse reactions.
In vitro studies have shown that bicalutamide can displace the coumarin anticoagulant warfarin from plasma protein binding sites.
Overdose
Overdose
Cases of overdose in humans have not been described.
There is no specific antidote. Treatment is symptomatic.
Dialysis is not effective, since bicalutamide is tightly bound to proteins and is not excreted unchanged by the kidneys. General supportive therapy and monitoring of vital body functions are indicated.
Storage conditions
Storage conditions
In a dry place, protected from light, at a temperature not exceeding 25 °C
Shelf life
Shelf life
2 years
Manufacturer
Manufacturer
Kanonpharma production CJSC, Russia
Additional information
| Shelf life | 2 years |
|---|---|
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25 °C |
| Manufacturer | Kanonfarma Production ZAO, Russia |
| Medication form | pills |
| Brand | Kanonfarma Production ZAO |
Related products
Buy Bicalutamide Canon, 150 mg 30 pcs with delivery to USA, UK, Europe and over 120 other countries.