No products in the cart.
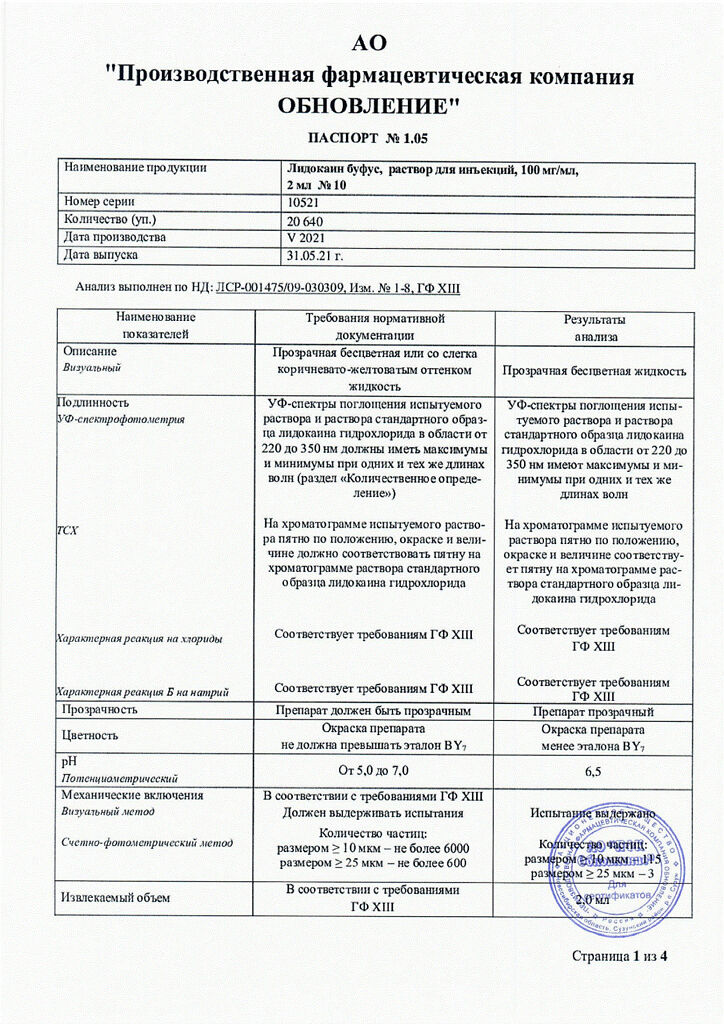
Lidocaine bufus, 100 mg/ml 2 ml 10 pcs
€4.42 €3.93
Description
It has antiarrhythmic (class Ib) action. Stabilizes cell membranes, blocks sodium channels, increases membrane permeability to potassium ions.
With almost no effect on the electrophysiological state of the atria, lidocaine accelerates repolarization in the ventricles, inhibits phase IV depolarization in Purkinje fibers (especially in ischemic myocardium), reducing their automaticity and duration of action potential, increases the minimum potential difference at which myofibrils respond to premature stimulation.
Shortens the duration of the action potential and the effective refractory period. It has no significant effect on myocardial conduction and contractility (suppression of conduction is observed only when administered in large doses, close to toxic) – the duration of intervals PQ , QT and the width of the QRS complex on the electrocardiogram does not change. Negative inotropic effect is also expressed insignificantly and appears transiently only when administered in high doses.
Pharmacokinetics
Absorption
The main factor determining the absorption rate and concentration in the blood is the total dose administered, regardless of the site of administration. There is a linear relationship between the amount of lidocaine administered and the resulting maximum concentration of the drug in the blood.
Distribution
Lidocaine binds to plasma proteins, including α1-acid glycoprotein (ACG) and albumin. The degree of binding is variable and is approximately 66%. Plasma concentration of AKG in newborns is low, so they have relatively high content of free bioactive fraction of lidocaine.
Lidocaine penetrates through the blood-brain and placental barriers, probably by passive diffusion.
Metabolism
Lidocaine is metabolized in the liver, with about 90% of the administered dose undergoing N-dealkylation to form monoethylglycincylidide (MEGX) and glycincylidide (GX), both contributing to the therapeutic and toxic effects of lidocaine. The pharmacological and toxic effects of MEGX and GX are comparable to those of lidocaine, but are weaker. GX has a longer half-life than lidocaine (about 10 hours) and may cumulate with repeated administration.
Metabolites resulting from subsequent metabolism are excreted with urine, the content of unchanged lidocaine in urine does not exceed 10%.
Excretion
The terminal elimination half-life of lidocaine after intravenous bolus injection in healthy adult volunteers is 1-2 hours. The terminal half-life of GX is approximately 10 hours, MEGX 2 hours.
Special patient groups
Due to rapid metabolism, the pharmacokinetics of lidocaine may be affected by conditions that impair hepatic function. In patients with hepatic dysfunction, the elimination half-life of lidocaine may increase two or more times.
Impaired renal function does not affect the pharmacokinetics of lidocaine, but may lead to cumulation of its metabolites.
Newborns have a low concentration of ACG, so binding to plasma proteins may be reduced. Due to the potentially high concentration of the free fraction, the use of lidocaine in neonates is not recommended.
Indications
Indications
Treatment of persistent paroxysms of ventricular tachycardia, including myocardial infarction and cardiac surgery. Prevention of recurrent ventricular fibrillation in acute coronary syndrome and recurrent paroxysms of ventricular tachycardia (usually within 12-24 hours).
Ventricular arrhythmias due to glycoside intoxication.
Active ingredient
Active ingredient
Lidocaine
Composition
Composition
Active ingredient:
Lidocaine hydrochloride
(in terms of anhydrous substance) – 100 mg
Excipients:
Sodium chloride – 6 mg
Sodium hydroxide
(0.1 M sodium hydroxide solution) – to pH 5.0-7.0
Water for injection – to 1 ml
How to take, the dosage
How to take, the dosage
- For infiltration anesthesia: intradermal, subcutaneous, intramuscular. Lidocaine solution 5 mg/ml (maximum dose 400 mg) is used.
- For blockade of peripheral nerves and nerve plexuses: perineurally, 10-20 ml of 10 mg/ml solution or 5-10 ml of 20 mg/ml solution (max 400 mg).
- For perineural anaesthesia: perineural 10 mg/ml and 20 mg/ml solutions (max. 400 mg).
- For epidural anaesthesia: epidural, 10 mg/ml or 20 mg/ml solutions (max. 300 mg).
Interaction
Interaction
It is undesirable to combine lidocaine with the following drugs:
With beta-adrenoblockers because of increased toxic properties of lidocaine, with digitoxin because of weakened cardiotonic effect, with curare-like drugs – increased muscle relaxation.
Lidocaine should not be administered together with aymalin, amiodarone, verapamil or quinidine due to increased cardiodepressant effect.
Concomitant use of lidocaine and novocainamide may cause CNS agitation and hallucinations.
In intravenous administration of hexenal or thiopental sodium with lidocaine may cause respiratory depression.
MoA inhibitors may increase the local anesthetic effect of lidocaine. Patients taking MAO inhibitors should not administer lidocaine parenterally.
The simultaneous administration of lidocaine and polymyxin-B may increase the inhibitory effect on neuromuscular transmission, so the respiratory function of the patient should be monitored in this case.
The simultaneous use of lidocaine with hypnotics or sedatives may increase their CNS depressant effect. When lidocaine is administered intravenously to patients taking cimetidine such unwanted effects as stunned state, somnolence, bradycardia, parasthesias and others are possible. This is associated with increased plasma levels of lidocaine, which is explained by the release of lidocaine from bonding with blood proteins, as well as a slowdown of its inactivation in the liver. If combination therapy with these drugs is necessary, the dose of lidocaine should be reduced.
Pharmaceutical interaction
When used concomitantly, the following drugs increase the concentration of lidocaine in blood serum: aminazin, cimetidine, propranolol, pethidine, bupivacaine, quinidine, disopyramide, amitriptyline, imipramine, nortriptyline.
Special Instructions
Special Instructions
Lidocaine should be used with caution in patients with myasthenia gravis, epilepsy, chronic heart failure, bradycardia and respiratory depression, and in combination with drugs that interact with lidocaine and lead to increased bioavailability, potentiation of its effects (eg, phenytoin) or prolonged excretion (eg, in hepatic or terminal renal failure, where lidocaine metabolites may cumulate).
Patients receiving class III antiarrhythmic drugs (e.g., amiodarone) should be monitored closely and ECG monitoring should be established as effects on the heart may potentiate.
It has been shown that lidocaine can cause porphyria in animals; its use in persons with porphyria should be avoided.
When injected into inflammatory or infected tissues, the effect of lidocaine may decrease.
Hypokalemia, hypoxia, and acid-base imbalance should be addressed before starting intravenous lidocaine administration.
The lidocaine injection solution is not recommended for use in infants. The optimal serum concentration of lidocaine to avoid toxicity such as seizures and arrhythmias has not been established in neonates.
Effect of the drug for medical use on the ability to drive vehicles, mechanisms
Depending on the dose and route of administration, lidocaine may have a temporary effect on motor ability and coordination. During treatment, caution should be exercised when driving vehicles and engaging in other potentially dangerous activities that require increased concentration and speed of psychomotor reactions.
Contraindications
Contraindications
Hypersensitivity to the drug components; syndrome of sinus node weakness, expressed bradycardia (heart rate less than 50 beats per min), grade II and III atrioventricular (AV) block (if no pacemaker is installed), sinoatrial block, WPW (Wolf-Parkinson-White) syndrome, acute and chronic heart failure (functional class III-IV according to NYHA classification), cardiogenic shock, intraventricular conduction disorders; severe arterial hypotension, Morgania Adams-Stokes syndrome.
With caution
Lidocaine should be administered with caution in patients with myasthenia gravis, epilepsy, chronic heart failure of degree II and III, hypovolemia, atrioventricular block of degree I, sinus bradycardia, severe hepatic and/or renal insufficiency, coagulopathy Complete and incomplete block of intracardiac conduction, seizure disorders, Melkerson-Rosenthal syndrome, porphyria, reduced hepatic blood flow, weakened or elderly patients (over 65 years), children under 18 years (due to delayed metabolism the drug may accumulate) and the third trimester of pregnancy.
Side effects
Side effects
Adverse reactions are described according to the MedDRA systemic organ classes.
Immune system disorders
Hypersensitivity reactions (allergic or anaphylactoid reactions, anaphylactic shock) – see also skin and subcutaneous tissue disorders. Skin allergy test for lidocaine is considered unreliable.
Nervous system disorders and mental disorders
Neurological signs of systemic toxicity include dizziness, nervousness, tremors, paresthesias around the mouth, tongue numbness, drowsiness, seizures, coma.
Reactions from the nervous system may manifest as its excitation or oppression. Signs of central nervous system stimulation may be short-lived or not occur at all, so that the first manifestations of toxicity may be confusion and drowsiness, followed by coma and respiratory failure.
Visual disturbances
Signs of lidocaine toxicity may include blurred vision, diplopia, and transient amaurosis.
Hearing and labyrinth disturbances
Tinnitus, hyperacusis.
Cardiovascular system disorders
Cardiovascular reactions are manifested by arterial hypotension, bradycardia, inhibition of myocardial contractile function (negative inotropic effect), arrhythmias; cardiac arrest or circulatory failure are possible.
Respiratory system, thorax and mediastinum disorders.
Dyspnea, bronchospasm, respiratory depression, respiratory arrest.
Gastrointestinal tract disorders
Nausea, vomiting.
Skin and subcutaneous tissue disorders
Rash, urticaria, angioedema, facial edema.
Overdose
Overdose
Symptoms. Central nervous system toxicity is manifested by symptoms of increasing severity. Paresthesias around the mouth, numbness of the tongue, dizziness, hyperacusis, and tinnitus may first develop. Visual disturbance and muscle tremor or muscle twitching are indicative of more severe toxicity and precede generalized seizures.
Loss of consciousness and grand convulsive seizures lasting from a few seconds to several minutes may then occur. Seizures lead to a rapid increase in hypoxia and hypercapnia due to increased muscle activity and respiratory disturbances. Apnea may develop. In severe cases there are disorders of the cardiovascular system.
At high systemic concentration arterial hypotension, bradycardia, arrhythmia and cardiac arrest may develop which may be fatal.
Overdose resolution is due to redistribution of the drug from the central nervous system and its metabolism, it can proceed quite rapidly (unless a very large dose of the drug was administered).
Treatment. In case of signs of overdose the drug administration should be stopped immediately.
Seizures, central nervous system depression and cardiotoxicity require medical intervention. The main goals of therapy are to maintain oxygenation, stop seizures, maintain blood circulation and arrest acidosis (if it develops).
In appropriate cases, it is necessary to ensure airway patency and administer oxygen and assisted lung ventilation (masked or with Ambu bag). Circulation is maintained by infusion of plasma or infusion solutions. If prolonged circulatory support is necessary, vasopressors should be considered, but they increase the risk of central nervous system excitation.
Seizure control can be achieved by intravenous administration of diazepam (0.1 mg/kg) or sodium thiopental (1-3 mg/kg), but note that anticonvulsants may also depress respiration and circulation. Prolonged seizures may interfere with ventilation and oxygenation of the patient, so early endotracheal intubation should be considered. If the heart stops, standard cardiopulmonary resuscitation is initiated.
The effectiveness of dialysis in the treatment of acute lidocaine overdose is very low.
Pregnancy use
Pregnancy use
Fertility
There are no data on the effect of lidocaine on human fertility.
Pregnancy
Use is possible if the expected benefits to the mother exceed the potential risk to the fetus and child.
Breastfeeding
Lidocaine penetrates breast milk in small amounts, its oral bioavailability is very low. Thus, the expected amount taken with breast milk is very small, therefore, the potential harm to the baby is very low. The decision whether to use lidocaine during the breastfeeding period is up to the physician.
Similarities
Similarities
Lidocaine, Versatis, Lidocaine-Vial
Additional information
| Weight | 0.050 kg |
|---|---|
| Shelf life | 3 years. |
| Conditions of storage | In a dry place protected from light. |
| Manufacturer | Update PFC AO, Russia |
| Medication form | solution for injection |
| Brand | Update PFC AO |
Other forms…
Related products
Buy Lidocaine bufus, 100 mg/ml 2 ml 10 pcs with delivery to USA, UK, Europe and over 120 other countries.