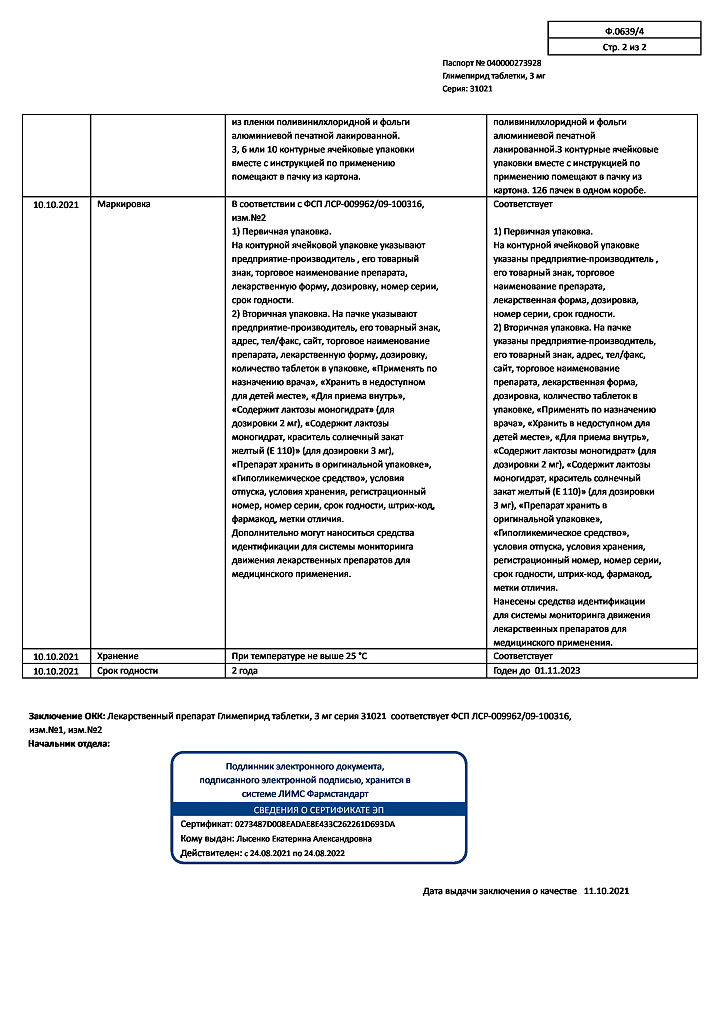
No products in the cart.
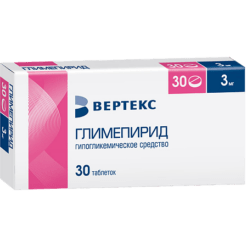
Glimepiride, tablets 3 mg 30 pcs
€7.06 €5.89
Description
Glimepiride is an oral hypoglycemic drug – a sulfonylurea derivative of new (third) generation. Glimepiride acts mainly by stimulating the secretion and release of insulin from the beta cells of the pancreas (pancreatic action). As with other sulfonylurea derivatives, this effect is based on the increased response of pancreatic beta cells to physiological glucose stimulation, and the amount of insulin secreted is significantly lower than that of traditional sulfonylurea derivatives. The smallest stimulating effect of glimepiride on insulin secretion also reduces the risk of hypoglycemia. In addition, glimepiride has extrapancreatic action – the ability to improve the sensitivity of peripheral tissues (muscle, fat) to the action of its own insulin, reduce insulin uptake by the liver; it inhibits the production of glucose in the liver.
Glimepiride selectively inhibits cyclooxygenase and reduces the conversion of arachidonic acid to thromboxane A2, which promotes platelet aggregation, thus having an antithrombotic effect.
Glimepiride promotes normalization of lipids, reduces the level of low aldehyde in the blood, which leads to a significant reduction in lipid peroxidation, this promotes the anti-atherogenic effect of the drug.
Glimepiride increases the level of endogenous α-tocopherol, catalase activity, glutathione peroxidase and superoxide dismutase that helps to decrease the severity of oxidative stress in patient’s body which is constantly present in type 2 diabetes.
Indications
Indications
Type 2 diabetes
Type 2 diabetes mellitus.
Active ingredient
Active ingredient
Glimepiride
Composition
Composition
Active ingredient:
glimepiride 3 mg;
Associates:
Lactose monohydrate 150.80 mg,
Corn starch 4.66 mg,
sodium carboxymethyl starch 10.00 mg,
povidone 6.00 mg,
polysorbate 1.34 mg,
2.00 mg talc,
magnesium stearate 1.20 mg,
iron dye (E 172) 0.30 mg
How to take, the dosage
How to take, the dosage
The drug is administered orally. The initial and maintenance doses of Glimepiride are set individually on the basis of the results of regular monitoring of blood glucose concentration.
The initial dose and dose selection
At the beginning of treatment, 1 mg of Glimepiride is prescribed once daily. When the optimal therapeutic effect is achieved, it is recommended to take this dose as a maintenance dose. In the absence of glycemic control, the daily dose should be gradually increased under regular monitoring of blood glucose concentration (at intervals of 1-2 weeks) to 2 mg, 3 mg or 4 mg daily. Doses over 4 per day are effective only in exceptional cases. The maximum recommended daily dose is 6 mg.
Perhaps in combination with metformin
In the absence of glycemic control in patients taking metformin, concomitant therapy with Glimepiride may be initiated. If the dose of metformin is maintained at the same level, treatment with Glimepiride begins with the minimum dose, and then the dose is gradually increased depending on the desired level of glycemic control, up to the maximum daily dose. Combination therapy should be carried out under close medical supervision.
In combination with insulin
In cases when it is not possible to achieve glycemic control by taking the maximum dose of Glimepiride, in monotherapy or in combination with the maximum dose of metformin, a combination of Glimepiride with insulin is possible. In this case, the last dose of Glimepiride prescribed to the patient remains unchanged. In this case, treatment with insulin begins with a minimum dose, with a possible subsequent gradual increase in its dose under the control of blood glucose concentrations. Combined treatment requires close monitoring by a physician.
The time and frequency of administration of the daily dose is determined by the physician, taking into account the patient’s lifestyle. As a rule, it is sufficient to prescribe a daily dose in one sitting immediately before or during a solid breakfast or the first main meal. Glimepiride tablets are taken whole, without chewing, with enough fluid (about 0.5 glasses). It is very important not to skip meals after taking Glimepiride.
The duration of treatment
As a rule, treatment with Glimepiride, is long.
The transfer of a patient from another oral hypoglycemic drug to Glimepiride.
When transferring a patient from another oral hypoglycemic drug to Glimepiride, the initial daily dose of the latter should be 1 mg (even if the patient is transferred to Glimepiride from the maximum dose of another oral hypoglycemic drug). Any increase in the dose of Glimepiride should be made in stages according to the above recommendations. It is necessary to take into account the effectiveness, dose and duration of action of the used hypoglycemic agent. In some cases, especially when taking hypoglycemic drugs with a long elimination half-life (e.g., chlorpropamide), it may be necessary to temporarily (for several days) discontinue treatment to avoid additive effects that increase the risk of hypoglycemia.
Transfer of a patient from insulin to Glimepiride
In exceptional cases, when insulin therapy is performed in patients with type 2 diabetes, with compensation of the disease and with preserved secretory function (pancreatic 3-cells), insulin replacement with Glimepiride is possible. The transfer should be carried out under the close supervision of a physician. In this case, transfer of the patient to Glimepiride is started with a minimum dose of 1 mg.
Interaction
Interaction
Increased hypoglycemic effect of glimepiride is possible with concomitant use with insulin or other hypoglycemic drugs, ACE inhibitors, allopurinol, anabolic steroids and male sex hormones, chloramphenicol, coumarin derivatives, cyclophosphamide, disopyramide, fenfluramine, pheniramidol, fibrates, fluoxetine, guanethidine, isophosphamides, MAO inhibitors, miconazole, PASC, pentoxifylline (when injected in high doses), phenylbutazone, azapropazone, oxyphenbutazone, probenecid, quinolones, salicylates, sulfinpyrazone, sulfonamides, tetracyclines.
. Weakening of the hypoglycemic effect of glimepiride is possible with concomitant use with acetazolamide, barbiturates, corticosteroids, diazoxide, diuretics, epinephrine (adrenaline) and other sympathomimetics, glucagon, laxatives (after long-term use), nicotinic acid (in high doses), estrogens and progestogens, phenothiazine, phenytoin, rifampicin, thyroid hormones.
When used concomitantly, histamine H2-receptor blockers, clonidine and reserpine can both potentiate and decrease the hypoglycemic effects of glimepiride.
The effects of coumarin derivatives may be enhanced or weakened with glimepiride.
Ethanol may increase or decrease the hypoglycemic effect of glimepiride.
Contraindications
Contraindications
Hypersensitivity, type 1 diabetes, diabetic ketoacidosis, diabetic precoma and coma, severe hepatic and renal dysfunction, leukopenia, pregnancy, lactation.
Side effects
Side effects
Cardiovascular system and blood (hematopoiesis, hemostasis): rarely – BP decrease, thrombocytopenia, leukopenia, granulocytopenia, agranulocytosis, erythropenia, pancytopenia, hemolytic and aplastic anemia.
Nervous system and sensory organs: dizziness, headache, transient visual impairment.
Gastrointestinal system disorders: nausea, vomiting, abdominal pain, feeling of heaviness in the epigastric region, diarrhea, intrahepatic cholestasis.
Metabolism disorders: hypoglycemia.
Others: increase in transaminases, hyponatremia, skin allergic reactions, cutaneous porphyria, asthenia; rarely – shortness of breath, hepatitis, allergic vasculitis, photosensitization.
Overdose
Overdose
Symptoms: hypoglycemia (nausea, vomiting and epigastric pain, restlessness, tremors, visual disturbances, coordination disorders, drowsiness, coma and seizures).
Treatment: if the patient is conscious – induction of vomiting, plenty of fluids, activated charcoal and laxatives. In case of severe overdose – intravenous bolus injection of dextrose solution (50 ml of 40% solution), then – infusion of 10% solution. Continuous monitoring of the patient, maintenance of vital functions and control of blood glucose concentration (recurrent episodes of hypoglycemia are possible). Further treatment is symptomatic.
Similarities
Similarities
Amaril, Glimepiride, Glimepiride
Additional information
| Weight | 0.013 kg |
|---|---|
| Shelf life | 5 years |
| Conditions of storage | In a light-protected place at a temperature not exceeding 25 °C, in a tightly closed container. |
| Manufacturer | Pharmstandard-Leksredstva, Russia |
| Medication form | pills |
| Brand | Pharmstandard-Leksredstva |
Other forms…
Related products
Buy Glimepiride, tablets 3 mg 30 pcs with delivery to USA, UK, Europe and over 120 other countries.