Subtotal: €2.65
Anti-allergic agent – H1-histamine receptor blocker.
ATC code:
Indications
Allergies, Sneezing, Pollinosis, Dermatosis, Neurodermatitis, Psoriasis, Allergic Rhinitis, UrticariaPollinosis, acute and chronic urticaria, angioneurotic edema, allergic rhinitis, dermatoses including eczema, neurodermatitis, skin itching.
Active ingredient
Hyphenadine
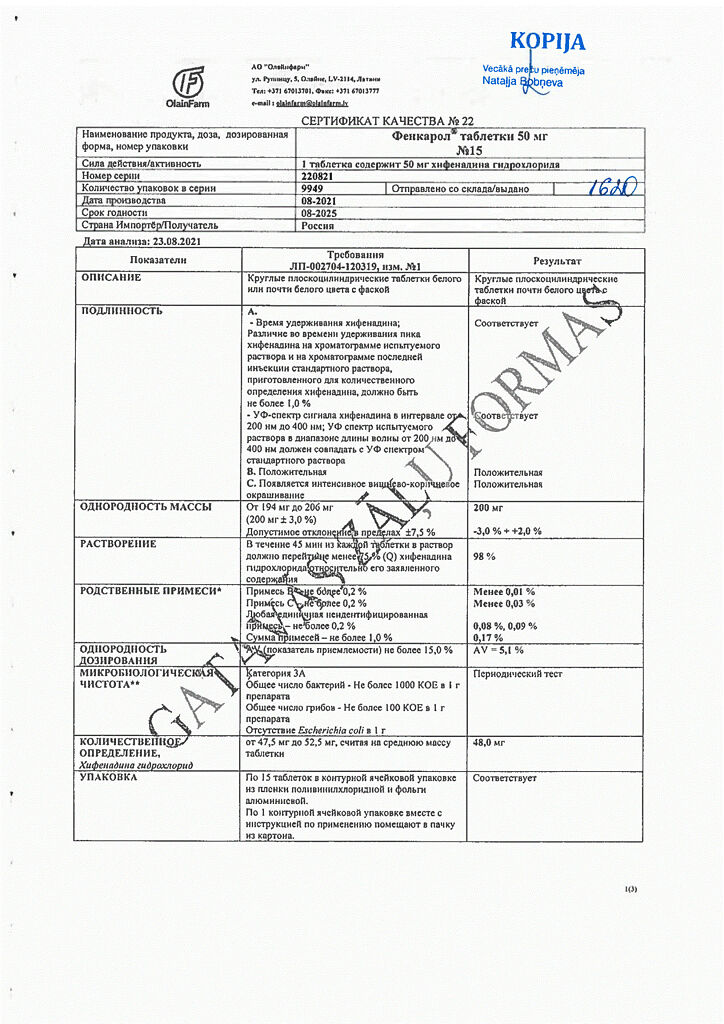
Composition
One tablet:
active ingredient: quiphenadine hydrochloride 50.0 mg;
excipients: potato starch 74.0 mg, sucrose 55.0 mg, modified corn starch 20.0 mg, calcium stearate 1.0 mg.
How to take, the dosage
The drug is taken orally after meals.
The dosing regimen of the drug is the same for all indications for use.
The dosing of quiphenadine may be influenced by the severity of allergic reactions, individual sensitivity of the patient, and the severity of possible side effects.
Adults are prescribed 25-50 mg 3-4 times per day.
The maximum daily dose is 200 mg.
The course of treatment is 10-20 days.
If necessary the course of treatment is repeated.
For children aged 3 to 7 years – 10 mg 2 times a day; aged 7 to 12 years – 10-15 mg 2-3 times a day; over 12 years – 25 mg 2-3 times a day.
The course of treatment is 10-15 days.
Interaction
With weak m-cholin-blocking properties, the drug may decrease gastrointestinal motility, which contributes to increased absorption of slowly absorbed drugs (for example, coumarin indirect-acting anticoagulants).
Special Instructions
The drug should be prescribed with caution in gastrointestinal diseases, liver and kidney diseases.
The absence of a pronounced m-cholin-blocking effect allows the drug to be prescribed to patients who are contraindicated in antihistamines with m-cholin-blocking activity.
Contraindications
- Hypersensitivity to any of the ingredients.
- Pregnancy, lactation.
- Deficiency of sucrose/isomaltase, fructose intolerance, glucose/galactose malabsorption, because the drug contains sucrose.
- Children under 18 years of age (for 50 mg dosage).
With caution
With caution for gastrointestinal, hepatic and renal diseases.
Side effects
Dry mouth, nausea, vomiting, drowsiness, allergic reactions, headache.
Overdose
Symptoms: dry mucous membranes, headache, vomiting, abdominal pain and other symptoms of dyspepsia.
Treatment: gastric lavage, administration of activated charcoal, symptomatic therapy.
In case of overdose the patient should consult a physician.
Pregnancy use
The drug is contraindicated in pregnancy.
If you need to treat the drug, breastfeeding should be discontinued.
| Weight | 0.050 kg |
|---|---|
| Shelf life | 4 years. |
| Conditions of storage | In a light-protected place at a temperature not exceeding 25 °C. |
| Manufacturer | Olinefarm, Latvia |
| Medication form | pills |
| Brand | Olinefarm |
Other forms…
Related products
Buy Fencarol, tablets 50 mg 15 pcs with delivery to USA, UK, Europe and over 120 other countries.